Translate this page into:
Blood pressure trend of lowlanders during first 6 days of acclimatisation to high altitude
-
Received: ,
Accepted: ,
How to cite this article: Singh K, Shekhar A, Singh SK, Kumar R, Yadav AK, Manimaran P, et al. Blood pressure trend of lowlanders during first 6 days of acclimatisation to high altitude. Indian J Physiol Pharmacol 2022;66:126-30.
Abstract
Objectives:
The present study was planned to delineate the blood pressure (BP) response of the lowlanders during initial 6 days of acclimatisation to high altitude.
Materials and Methods:
398 normotensive sojourners at sea level ascended to HA (3,500 m). Blood pressure response of these subjects were analysed for 6 consecutive days at HA. Based on the BP response of these subjects on the 6th day at HA they were divided into two groups: HBP-high BP group and NBP- normal BP group.
Results:
It showed that on 6th day at HA, 51 out of 398 subjects presented with BP≥140/90 mmHg (HBP) while others remained normotensive (NBP). Comparative systolic BP of HBP and NBP groups during successive days at HA were as follows: HA day 1 (146.8 ± 13.7 mmHg vs 146.3 ± 8.8 mmHg, p>0.05), HA day 4 (144.5 ± 8.7 mmHg vs 135.0 ± 7.0 mmHg, p<0.05) and HA day 6 (148.1 ± 7.9 mmHg vs 131.7 ± 5.3 mmHg, p<0.05). Whereas, diastolic BP of HBP and NBP groups were: HA day 1 (94.0 ± 8.1 mmHg vs 93.2 ± 6.6 mmHg, p>0.05), HA day 4 (88.9 ± 6.0 mmHg vs 85.3 ± 5.1 mmHg, p<0.05) and HA day 6 (93.8 ± 6.3 mmHg vs 83.7 ± 3.8 mmHg, p<0.05).
Conclusion:
It was found that NBP had initially raised BP which returned to normal levels after acclimatisation period of 6 days, but in HBP, it started rising after 4th day. It can be concluded that those individuals whose BP on 6th day at HA does not return to normal after acclimatisation period needs regular follow up as these individuals can have a delayed acclimatisation normalising BP later on or can become hypertensive subsequently.
Keywords
High altitude
Hypoxia
High altitude systemic hypertension
Acclimatisation
Lowlanders
INTRODUCTION
Low barometric pressure at high altitude (HA) lowers the alveolar oxygen in human sojourns in these areas. This hypobaric hypoxia stimulates oxygen sensitive chemoreceptors in the carotid body and the brainstem resulting in an increased efferent sympathetic outflow.[1,2] The sustained activation of sympathetic nervous system ensures oxygen supply to cells and tissues by increasing heart rate and cardiac output.[3] Subsequently, the acclimatisation process partially restores arterial oxygen content thereby reducing sympathoexcitatory chemoreceptor drive. Despite this normalisation, some healthy humans exhibit increased systemic arterial pressure irrespective of the hypoxia exposure duration.[4]
It is observed that the normotensive individuals at sea level frequently end up with hypertension when exposed to HA stress. This hypoxic stress aggravates the 24-h ambulatory systolic, diastolic, day time and night time blood pressure of individuals, in both normotensive as well as hypertensive.[5] This augmentation of BP in hypertensive individuals is more pronounced; hence, poorly controlled hypertensives are advised to refrain from travel to HA. Various investigations have demonstrated differing impacts of BP in response to HA in normotensive subjects. Studies in healthy individuals without hypertension have either shown increase, decrease, or no change in BP at HA.[5-7] It is important to note that effect of HA on BP is inadequately characterised. Some studies have associated modified sympathoadrenal activity for systemic hypertension at HA.[8,9]
One of the intriguing findings is that normotensives who develop hypertension at HA revert to normal values on returning to sea level. Such type of hypertension has been arbitrarily labelled as HA systemic hypertension. This is of significance in case of people who spend a longer tenure at HA like soldiers and mine workers. Hypertension in such individuals regularly faces the repercussion of increased BP and is frequently put on antihypertensive medication.
It has been seen that hypoxia of HA increases BP and also an appreciable number of lowlanders who stay at HA for longer time develop systemic hypertension. The present study was hence planned to delineate the BP response of the lowlanders during initial days of acclimatisation to HA.
MATERIALS AND METHODS
Subjects
An observational and cohort study was conducted at two transit camps located at sea level (altitude 321 m) and at an altitude of 3500 m, during December 2018–April 2019. A total of 500 Indian male soldiers initially became a part of the study but data collection could be completed for 398 subjects. Inclusion criteria were age more than 20 years and BP <140/90 mmHg at sea level. Exclusion criteria were: (1) Smoking (2) chronic diseases such as diabetes, chronic kidney or liver disease and (3) on any long-term medication. Sea level data served as baseline values of these subjects. The approval of study protocols was taken from the Institutional Ethics Committee and informed written consent was obtained from all the subjects.
BP measurements
BP measurements were made on the right-hand supine using calibrated mercury sphygmomanometers by trained nurses. Three readings were taken at an interval of 5 min after relaxing for 5 min and subject was lying quietly and arm at the heart level. Average of three readings was taken as the BP value. Every time, one dedicated nurse use to measure BP at sea level and HA. Systolic BP (SBP) was defined by the appearance of first Korotkoff sound whereas diastolic BP (DBP) by fifth Korotkoff sound (disappearance of sound). Mercury manometers were chosen as they are not influenced by altitude. Normal BP (NBP) was considered BP <140/90 mm Hg called NBP group and high BP (HBP) was > 140/90 mm Hg – HBP group.[10,11]
HA measurements
For HA, subjects were taken to Leh, Ladakh (3500 m), India by air induction and BP of these subjects was monitored for 6 consecutive days. All the subjects received same fixed diet and performed comparable exercise during the whole investigation time frame. The subjects were refrained from taking any medication of their own like acetazolamide or dexamethasone.
Statistical analysis
Continuous variable was presented as mean and standard deviation. Categorical was described as number and percentage. Repeated measure ANOVA was done for repeated SBP, DBP and mean BP (MBP) over a period of days. Repeated measures mixed model using REML was used to adjust for age and body mass index (BMI). StataCorp. 2019. Stata Statistical Software: Release 16. College Station, TX: StataCorp LLC. P < 0.05 was considered significant.
RESULTS
Data were collected from 398 participants out of 500 participants eligible during the study period. The physical characteristics of the participants are shown in [Table 1].
| Age | 31.5±6.4 Yrs |
|---|---|
| Weight | 68.8±8.4 Kg |
| Height | 1.7±0.1 m |
| BMI | 23.2±2.5 kg/m2 |
| SBP at Sea Level | 123.3±8.5 |
| BP at Sea Level | 79.2±5.8 |
Values are presented as Mean±SD
At HA out of 398 participants on 6th day, 51 (12.8% [95% CI: 9.6–16.5]) had BP more than 140/90 mmHg and 347 had BP <140/90 mmHg. The pattern of change of BP over time is shown in [Figure 1].

- Pattern of change in blood pressure at different days at high altitude.
On HA day 1, both the groups exhibited significantly increased BP from sea level (SL) and on HA day 4, HBP group showed significantly higher BP than NBP group SBP (144.5 ± 8.7 mmHg vs. 135.0 ± 7.0 mmHg, P < 0.05) and DBP (88.9 ± 6.0 mmHg vs. 85.3 ± 5.1 mmHg, P < 0.05). Similarly, on HA day 6, HBP group showed significantly higher BP than NBP group SBP (148.1 ± 7.9 mmHg vs. 131.7 ± 5.3 mmHg, P < 0.05) and DBP (93.8 ± 6.3 mmHg vs. 83.7 ± 3.8 mmHg, P < 0.05) [Table 2].
| SBP1 | SBP2 | SBP3 | SBP4 | SBP5 | SBP6 | P-value | |
|---|---|---|---|---|---|---|---|
| NBP group (347) | 146.3 (8.8) | 141.5 (8.8) | 137.4 (9.1) | 135 (7) | 132.8 (8.5) | 131.7 (5.3) | <0.001 |
| HBP group (51) | 146.8 (13.7) | 144.2 (8.9) | 142.9 (9.6) | 144.5 (8.7) | 146.6 (11) | 148.1 (7.9) | |
| DBP1 | DBP2 | DBP3 | DBP4 | DBP5 | DBP6 | ||
| NBP group (347) | 93.2 (6.6) | 89.2 (8) | 87.4 (5.4) | 85.3 (5.1) | 84.9 (4.5) | 83.7 (3.8) | <0.001 |
| HBP group (51) | 94 (8) | 91.3 (6.8) | 90.3 (5.8) | 88.9 (6) | 90.9 (5.8) | 93.8 (6.3) | |
| MBP1 | MBP2 | MBP3 | MBP4 | MBP5 | MBP6 | ||
| NBP group (347) | 109.8 (6) | 105.6 (7) | 103 (5.5) | 100.8 (5) | 99.8 (4.5) | 98.7 (3.7) | <0.001 |
| HBP group (51) | 110.5 (9.1) | 107.8 (6.6) | 106.8 (6) | 106.4 (5.9) | 108.4 (6.1) | 110.8 (5.4) |
Values are mean (SD), SBP1 to SBP6 - Systolic BP at day 1 to day 6 HA, DBP1 to DBP6 - Diastolic BP at day 1 to day 6 HA, MBP1 to MBP6 - Mean BP at day 1 to day 6 HA, HA: High altitude, BP: Blood pressure, SBP: Systolic blood pressure, NBP: Normal blood pressure, HBP: High blood pressure, DBP: Diastolic blood pressure
There was a significant difference in BP values through the extent of time as well as between HBP and NBP group for SBP, DBP and MBP (Repeated Measurement ANOVA: P < 0.001 for all three measurements). Repeated measurement model using restricted maximum likelihood method was done for adjustment of the age and BMI. However, the difference among and within group remains significant [Table 3] as regard to systematic BP, similar findings have been shown for diastolic and MBP (Not Shown). In the two groups, the difference in blood pressure became statistically significant from fourth day onwards [Table 3].
| Characteristic | Coefficient | 95% Confidence Interval | P-value |
|---|---|---|---|
| Time | |||
| Day 1 | Reference | ||
| Day 2 | −3.99 | −4.77-(−3.22) | <0.001 |
| Day 3 | −5.80 | −6.5–(−5.03) | <0.001 |
| Day 4 | −7.90 | −8.6–(−7.13) | <0.001 |
| Day 5 | −8.33 | −9.1–(−7.55) | <0.001 |
| Day 6 | −9.48 | −10.25–(−8.7) | <0.001 |
| Groups | |||
| Day 1 | Reference | ||
| Day 2 | 1.27 | −0.90– (3.43) | 0.25 |
| Day 3 | 2.12 | −0.47–4.3 | 0.06 |
| Day 4 | 2.84 | 0.68–5.01 | 0.01 |
| Day 5 | 5.22 | 3.06–7.40 | <0.001 |
| Day 6 | 9.34 | 7.18–11.51 | <0.001 |
| Age | 0.04 | −0.018–0.09 | 0.19 |
| BMI | −0.067 | −0.21–0.08 | 0.19 |
BMI: Body mass index
Mean MBP increased on 1st day of HA. The mean MBP decreased gradually for both HBP and NBP groups; however, for HBP, it started increasing on 4th day onwards at HA [Figure 2].
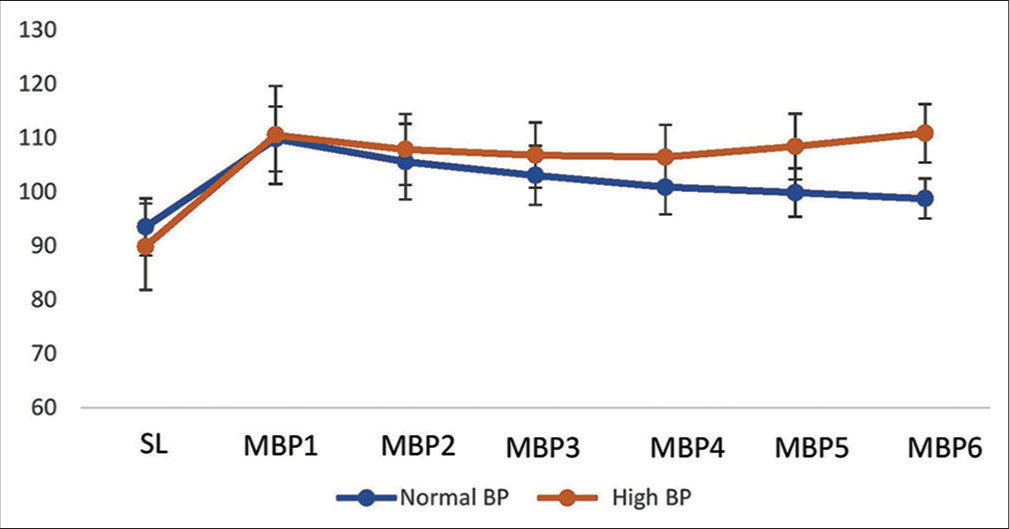
- Change in mean blood pressure on different days at high altitude.
DISCUSSION
The main finding of the present study is that the majority of the subjects had a continuous fall of BP during the days of acclimatisation making them normotensive whereas in others BP continuously increased 4th day onwards. The immediate effect of HA ascent is a decrease in BP by endothelium-dependent and endothelium-independent systemic vasodilation[12] but curiously our subjects did not show such instantaneous hypotensive impact of HA. This physiological response is counteracted by sympathetic vasoconstriction essentially by arterial hypoxemia after few hours of stay at HA. Peripheral chemoreceptors located in the carotid bodies and cardiovascular control regions of the mid-brain mediate hypoxia-induced vasoconstriction resulting in critical and steady increase in arterial BP.[13] In our study, there was increased BP on the 1st day at HA and this change was sustained over the first 3 days after which NBP group starts to come back to normal as acclimatisation sets in. In the HBP group, the increase was sustained and there was further increase. This was distinctly different from a study by Roach et al.,[14] in which the MBP of 64 normotensive and 33 older hypertensives was higher at 2500 m than recorded home measurements, but gradually diminished, stabilising on day 3. The present study observed this pattern in both the groups initially for 3 days but the HBP participants, started having higher BP from the 4th day onwards at HA and in NBP subjects BP continues to decline. Hence, on the 6th day at HA, BP reverted to normal indices (i.e., ≤140/90 mmHg) in the NBP group but persisted above 140/90 mmHg in the HBP group. The underlying mechanisms through which BP returns to near-normal or even lower values have been generally thought to be a part of the acclimatisation process. Despite the extant of the literature with regard to BP and acclimatisation timing, the period when continually exposed lowlanders achieves that acclimatisation is not well established,[3] and there are no such epidemiological studies.
The sustained high DBP and borderline SBP values observed in many subjects of our study group for a protracted period of time requires further investigation. The phenomenon could be a steady portrayal of sympathetic stimulation, though it is not yet known when it ceases.[3] Another factor playing a role in the elevation of BP in chronic hypoxia could also be an overstated reaction to enhanced chemoreceptor reflex described for hypertension prone individuals.[15] An increased blood viscosity[16] and alteration of the endothelial function[17] have likewise been implicated. Hence, a single factor will be insufficient to explain the phenomenon.
The increase of sympathetic tone might be a characteristic reaction to balance the impacts of hypoxia in case of nonacclimatised subjects. Hypoxia directly causes vasodilation and if acting through peripheral chemoreceptors leads to vasoconstriction through sympathetic stimulation.[18,19] Interactions occur between the hypoxic vasodilatation of systemic arterioles and the chemoreceptor-mediated responses in the systemic circulation.[20-22] This, thus prompts baroreceptor-mediated sympathetic excitation. Other varied reasons could be alterations in baroreflex function, an increase in the ‘set point’ and possibly a decrease in gain. These autonomic adjustments may have a role in an escalation in BP during sustained hypoxia.[22,23] Moreover, few data exist regarding the relative importance of other neurohormones such as angiotensin, aldosterone or vasopressin, all of which may be involved in some degree with raising BP during sustained high-altitude exposure.
Limitations
This study included only male subjects hence the data cannot be generalised to both the genders. Those subjects who had HBP on the 6th day could have been monitored for future development of hypertension.
CONCLUSION
There was a contrasting BP response of individuals during acclimatisation on continual recording. The majority of the subjects could revert the BP back to normal levels after the initial rise while in few, BP remained elevated throughout. Hence, it can be concluded that few percentage of lowlanders remain with high BP after acclimatisation to HA. Such individuals should be monitored regularly after acclimatisation as these individuals can have a delayed acclimatisation normalising BP later on or can become hypertensive subsequently.
Acknowledgments
The authors acknowledge the support of Indian Army soldiers for giving their supporting in conducting the study.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Sympathetic neural overactivity in healthy humans after prolonged exposure to hypobaric hypoxia. J Physiol. 2003;546:921-9.
- [CrossRef] [PubMed] [Google Scholar]
- Mechanisms and drug therapy of pulmonary hypertension at high altitude. High Alt Med Biol. 2013;14:126-33.
- [CrossRef] [PubMed] [Google Scholar]
- Chronic hypoxia increases blood pressure and noradrenaline spillover in healthy humans. J Physiol. 2003;551:379-86.
- [CrossRef] [PubMed] [Google Scholar]
- Increased arterial pressure after acclimatization to 4300 m: Possible role of norepinephrine. Int J Sports Med. 1992;13(Suppl 1):S18-21.
- [CrossRef] [PubMed] [Google Scholar]
- Changes in 24 h ambulatory blood pressure and effects of angiotensin II receptor blockade during acute and prolonged high-altitude exposure: A randomized clinical trial. Eur Heart J. 2014;35:3113-22.
- [CrossRef] [PubMed] [Google Scholar]
- Should travelers with hypertension adjust their medications when traveling to high altitude? High Alt Med Biol. 2009;10:11-5.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of acetazolamide on central blood pressure, peripheral blood pressure, and arterial distensibility at acute high altitude exposure. Eur Heart J. 2013;34:759-66.
- [CrossRef] [PubMed] [Google Scholar]
- Systemic hypertension at high altitude. Hypertension. 2018;72:567-78.
- [CrossRef] [PubMed] [Google Scholar]
- Systemic hypertension at 4,300 m is related to sympathoadrenal activity. J Appl Physiol (1985). 1994;76:1643-50.
- [CrossRef] [PubMed] [Google Scholar]
- Correlation between blood pressure changes and AMS, sleeping quality and exercise upon high-altitude exposure in young Chinese men. Mil Med Res. 2014;1:19.
- [CrossRef] [PubMed] [Google Scholar]
- Blood pressure and hypertension in people living at high altitude in Nepal. Hypertens Res. 2019;42:284-91.
- [CrossRef] [PubMed] [Google Scholar]
- Blood pressure responses in young adults first exposed to high altitude for 12 months at 3550 m. High Alt Med Biol. 2009;10:329-35.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical recommendations for high altitude exposure of individuals with pre-existing cardiovascular conditions: A joint statement by the European society of cardiology, the council on hypertension of the European society of cardiology, the European society of hypertension, the international society of mountain medicine, the Italian society of hypertension and the Italian society of mountain medicine. Eur Heart J. 2018;39:1546-54.
- [CrossRef] [PubMed] [Google Scholar]
- Autonomic and cardiovascular effects of acute high altitude exposure after myocardial infarction and in normal subjects. Circ J. 2009;73:1485-91.
- [CrossRef] [PubMed] [Google Scholar]
- Potentation of sympathetic nerve responses to hypoxia in borderline hypertensive subjects. Hypertension. 1988;11:608-12.
- [CrossRef] [PubMed] [Google Scholar]
- Invited review: Physiological consequences of intermittent hypoxia: Systemic blood pressure. J Appl Physiol (1985). 2001;90:1600-5.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of altitude on the heart and the lungs. Circulation. 2007;116:2191-202.
- [CrossRef] [PubMed] [Google Scholar]
- Richards lecture: Circulatory adjustments to hypoxia. Circulation. 1980;61:463-70.
- [CrossRef] [PubMed] [Google Scholar]
- Peripheral chemoreceptors and cardiovascular regulation. Physiol Rev. 1994;74:543-94.
- [CrossRef] [PubMed] [Google Scholar]
- Blood pressure changes in young male subjects exposed to a median altitude. Clin Auton Res. 2008;18:84-9.
- [CrossRef] [PubMed] [Google Scholar]
- Sympathetic response during 21 days at high altitude (4,300 m) as determined by urinary and arterial catecholamines. Metabolism. 1994;43:1226-32.
- [CrossRef] [Google Scholar]






