Translate this page into:
Potential of using JNK and p53 as novel drug targets for the treatment of alcoholic encephalopathy
-
Received: ,
Accepted: ,
How to cite this article: Zyuz`kov G, Miroshnichenko L, Polyakova T, Simanina E. Potential of using JNK and p53 as novel drug targets for the treatment of alcoholic encephalopathy. Indian J Physiol Pharmacol 2022;66:233-40.
Abstract
Objectives:
Investigating novel therapies for alcoholic encephalopathy (AE) would be part of the implementation of the concept of targeted pharmacological control of intracellular signalisation in regeneration-competent cells. This study aimed to explore the involvement of JNK and p53 in the implementation of the functions of different types of regeneration-competent cells of nervous tissue in alcoholic neurodegeneration (AN).
Materials and Methods:
The studies were conducted on C57B1/6 mice. AN was modelled in vitro and in vivo. The effects of the JNK and p53 inhibitors on the realisation of neural stem cell (NSC) and neuronal-committed progenitor (NCP) functions (their colony-forming ability, proliferative activity and intensity of specialisation), as well as on the secretion of neurotrophins by astrocytes, oligodendrocytes and microglial cells were studied. Individual cell fractions were prepared using an immunomagnetic separation method.
Results:
We showed that JNK and p53 stimulate the proliferation and specialisation of intact NSCs. An inversion of the role of these signalling molecules in the regulation of NSC proliferation in the conditions of modelling AN was revealed. It has been found that JNK and p53 are not involved in regulating the functions of NCP. The ambiguous role of JNK and p53 in the production of neurotrophic growth factors by different types of neuroglia cells was also found. Increased secretion of neurotrophins by oligodendrocytes and microglia during the blockade of JNK and p53 under conditions of exposure to ethanol cells was revealed.
Conclusion:
The results suggest the prospect of exploring the possibility of using JNK and/or p53 inhibitors as novel drugs to treat AE.
Keywords
Neural stem cells
JNK
p53
Neurodegenerative diseases
Targeted therapy
INTRODUCTION
In recent decades, the possibility of using individual intracellular signalling molecules as pharmacological targets has been actively explored. For example, in oncopharmacology, this direction is one of the main modern trends in the development of antitumor drugs.[1] Large pharmaceutical companies have developed several dozen antiblastic drugs based on intracellular signalling molecule inhibitors responsible for the growth and development of transformed cells. One of the advantages of these drugs is their selectivity, including not only the tissue affected by the pathological process but also in some cases –the type of tumour.[2,3]
A promising direction for solving the problems of regenerative medicine is the concept of targeted pharmacological control of intracellular signal transduction in regeneration-competent cells.[4-7] This approach involves the use as targets of individual signalling molecules of progenitors and microenvironment cells of tissues (cells regulators of reparation) to create drugs with selective regenerative activity.[8,9] However, the development of this concept of pharmacotherapy requires a detailed understanding of the peculiarities of intracellular signalling in cells of different tissues to determine the optimal targets for selective tissue-specific effects.
This is especially relevant when considering the problem within the framework of neurological practice for the treatment of neurodegenerative diseases.[10,11] Alcoholic encephalopathy (AE) is one of the common neurodegenerative diseases that are difficult to treat with existing drugs.[9] Therefore, it is expedient to develop novel effective approaches to its treatment. It is known that dysfunction of the central nervous system (CNS) in AE is observed against the background of the loss of the ability of the nervous tissue to neurogenesis.[8-10] In this regard, it is advisable to develop methods for pharmacological stimulation of the processes of cellular renewal of the brain through the activation of resident progenitor cells.
Promising is the development of approaches to stimulation of neurogenesis by activation of the functions of progenitor cells of nerve tissue – multipotent neural stem cells (NSCs) and neuronal-committed progenitors (NCPs).[6,12] It is known the importance of JNK- and p53-pathways (interacting with each other [having some ‘crossroads’]), in regulating the processes of proliferation and differentiation of progenitors,[13,14] as well as in the implementation of the functions of neuroglial cells (astrocytes, oligodendrocytes and microglial cells) producing a wide range of growth factors.[15] However, there is no detailed understanding of their role in the functioning of nerve tissue regeneration-competent cells.
The work aimed to identify the role of JNK and p53 in realising the growth potential of nervous tissue precursors (NSC and NCP) and the production of neurotrophic growth factors by neuroglial cells to determine the potential for their use as targets of agents for the treatment of AE.
MATERIALS AND METHODS
Chemicals and drugs
MACS, Neuro Medium; anti-PSA-NCAM MicroBeads; anti-ACSA-2 MicroBead Kit; Anti-O4 MicroBeads; Anti-CD11b (Microglia) MicroBeads; autoMACS Rinsing Solution; MACS BSA Stock Solution (all manufactured by Miltenyi Biotec, Germany); JNK inhibitor «SP600125» (InvivoGen, USA); p53 inhibitor «Pifithrin-a, Cyclic» (Santa Cruz Biotechnology, Inc. USA); hydroxyurea (Calbiochem, USA) and ethanol (Kemerovo Pharmaceutical Factory LLC, Russia) were used.
Animals and experimental design
All animal experiments were performed in accordance with the U.K. Animals (Scientific Procedures) Act, 1986 and the related guidelines of Directive 2010/63/EU for animal experiments. The study was approved by the Institute’s Local Ethics Committee (protocol GRIPh&RM-2020-010/02). Experiments were conducted using C57B1/6 male mice (n = 84) at the age of 2–2.5 months, weighing 22–24 g. Animals (1st category – conventional mice) were obtained from the Experimental Biological Models Department of the Goldberg Research Institute of Pharmacology and Regenerative Medicine (Tomsk, Russia) (certificate available). The euthanasia of the animals was carried out using a CO2 chamber.
Using cultural methods, we studied the direct effect of the JNK and p53 inhibitors (at a concentration of 10 mM and 5 mM, respectively) on the realisation of the growth potential of neural tissue precursor cells (NSC and NCP) and the secretion of neurotrophic growth factors (neurotrophins) by glial cells (astrocytes, oligodendrocytes and microglia) in the conditions of modelling alcoholic neurodegeneration (AN) in vitro and in vivo. The concentrations of the inhibitors of signalling molecules were determined according to the instructions of the company’s developers of these reagents.
In vitro ethanol-induced neurodegeneration was obtained by adding ethanol to the culture medium at a concentration (65 mM) (n = 18, each experimental and control group n = 6). In vivo modelling of the AN,[6] accompanied by the development of AE,[12] was carried out by daily oral administration (through probe) of 30% ethanol at a dose of 3 g/kg/day for 8 weeks to mice (n = 60, each experimental and control group n = 22). In addition, a 5% ethyl alcohol solution was used instead of free drinking water in the AN simulation. Cellular materials for the study were taken 10 days after the end of the introduction of ethanol in vivo. The control group of mice was injected with distilled water in an equivalent volume (mice also had free access to clean drinking water).
Determination of progenitors functional activity
The progenitors were sampled from the subventricular zone (SVZ) of the cerebral hemispheres. The NSC was investigated during the cultivation of unfractionated cells. The neuronal-committed progenitors (NCP and CD56+ cells) were isolated from SVZ cells using the immunomagnetic separator ‘MIniMACS Cell Separator’ (Miltenyi Biotec, Germany). CD56+ cells were obtained by positive selection (using suitable antibody kits and MS columns).[16] The obtained unfractionated and PSA-NCAM+ cells at a concentration of 105/ml were incubated in MACS Neuro Medium for 5 days in a CO2 incubator at 37°C, 5% CO2 and 100% air humidity.
After incubation in both cases (during the cultivation of unfractionated cells and CD56+ cells), the content of colony-forming units (CFU, neurospheres with more than 100 cells), cluster-forming units (ClFU, neurospheres from 30 to 100 cells), mitotic activity of CFU and their intensity of specialisation were calculated. The proliferative activity of the progenitors was assessed by the method of cell suicide technic using hydroxyurea (1 mM).[6] The CFU pool in phase S of the cell cycle was determined based on the formula: N = [(a-b)/a] × 100%, where a is the average for the group the number of CFU from cells not treated with hydroxyurea; b – the group average of the number of CFU from cells treated by hydroxyurea. The intensity of the processes of progenitor specialisation (differentiation index) was determined by calculating the ratio of the ClFU to CFU.[6,9]
Study of neurotrophic growth factors secretion by neuroglial cells
Individual fractions of astrocytes (ACSA-2+ cells),[17] oligodendrocytes (O4+ cells)[18] and microglial cells (CD11b+ cells)[19]) were also obtained from the SVZ using immunomagnetic positive selection (using the immunomagnetic separator ‘MIniMACS Cell Separator’ and suitable antibody kits and MS columns). The isolated cells at a concentration of 2 × 106/ml were incubated in MACS Neuro Medium for 2 days in a CO2 incubator at 37°C, 5% CO2 and 100% air humidity to obtain conditioned media from cells. To determine their secretory activity (neurotrophic growth factor production), the effect of supernatants on the level of CFU formation in the test system was studied.[5,6]
Statistical analysis
The results were analysed with one-way ANOVA followed by the Mann–Whitney test for independent samples. The data are expressed as arithmetic means. The significance level was P < 0.05.[20]
RESULTS
Changes in the functions of nervous tissue regeneration-competent cells under the influence of alcohol
The addition of toxicant (ethanol) to the culture medium did not cause a change in the formation of CFU and ClFU from both unfractionated and CD56+ cells. However, there was a decrease in the mitotic activity of NSC (CFUNSC) and NCP (CFUCD56+) (up to 77.3% and 80.8% of background values, respectively) with no change in their specialisation intensity [Figures 1a-d and 2a-d].

- Number of CFUNSC (a), ClFUNSC (b); NSC proliferative activity (c) and their differentiation index (d). Here and in Figures 2 and 3: Cell culture without alcohol (intact); with alcohol (in vitro); and mice after prolonged administration of ethanol per os (in vivo). White bars – without inhibitors of signalling molecule (white bars); gray bars – with the JNK inhibitor; black bars – with the p53 inhibitor; * – the significance of differences with intact at P < 0.05 and # – the significance of differences with the group without inhibitors of signalling molecule at P < 0.05.

- Number of CFUCD56+ (a), ClFUCD56+ (b); NCP proliferative activity (c) and their differentiation index (d).
The modelling AN by feeding ethyl alcohol to mice through the probe was accompanied by the development of similar changes in the functioning of the precursors. However, in this case, a decrease in CFUCD56+ and NSC specialisation (differentiation/maturation) intensity was observed.
In vitro ethanol addition was not accompanied by a change in neurotrophin secretion by astrocytes and oligodendrocytes [Figures 3a and 3b]. However, there was a decrease in the value of this parameter in the supernatants from microglial cells (up to 81.8% of the background value – a similar indicator in cultures without alcohol) [Figure 3c].

- Effect of conditioned media of ACSA-2+ cells (a), O4+ cells (b) and CD11b+ (c) on the level of neurosphere formation in the test system.
Other patterns of change in the secretion of humoral regulators’ have been reported in cells of mice subjected to chronic alcohol intoxication in vivo. There was an increase in the production of growth factors by astrocytes and oligodendrocytes (up to 121.7% and 153.7%, respectively) against the background of a significant drop in the level of formation of CFU under the influence of the supernatants from microglial cells (up to 75.8% of the control).
Abstracts and some oligodendrocyte populations (NG-2, OPC and others) are known to secrete many neurotrophic factors (NGF, BDNF, NT3, NT4, etc.) and do not produce inhibitors of nervous tissue progenitor functions.[21,22] While, microglia secrete cytokines that have both stimulating effects on NSC (LIF and CNTF) and inhibit the realisation of their growth potential (IL-1, IL-6, IL-15 and NF-α).[23,24] Therefore, it is obvious that the detected changes on the part of microglia largely reflect the production of precursor inhibitors – pro-inflammatory cytokines.[25]
These findings correspond to previously obtained data on the violation of proliferative capacities of nervous tissue precursors under the influence of alcohol.[6,12] In this regard, progenitors determined in the neuronal direction of cells should be considered the most vulnerable when exposed to alcohol in the body. At the same time, the obtained results indicate the development of compensatory responses from macroglia aimed at stimulation of neurogenesis in conditions of the impaired proliferation of NSC under the influence of ethanol,[12,23] while the effect of microglia in AN is not unequivocal. The intensity of the inflammatory response in nervous tissue in alcohol abuse largely determines the degree of progression of the pathological process.[26,27]
Effect of JNK and p53 inhibitors on the functioning of nervous tissue progenitors under conditions of their optimal vital activity
The addition of signalling molecule inhibitors to the culture of unfractionated SVZ cells was accompanied by a decrease in their clonogenicity. The number of CFUNSC was 54.6% and 58.5% of the background value under the blockade of JNK and p53, respectively [Figure 1a]. The state of the ClFUNSC (which are precursors with a lower self-renewal capacity and proliferating potential)[6,12] was characterised by less unambiguous changes. Violation of JNK phosphorylation led to an increase in the amount of ClFUNSC in the culture (up to 166.7% of the background) and inactivation of p53 did not affect the value of this parameter [Figure 1b]. In both cases, however, there was a significant increase in the intensity of progenitor specialisation processes. The differentiation index was 327.5% and 185.5% of controls with the JNK and p53 inhibitors, respectively [Figure 1d].
Other phenomena were observed in the study of the role of JNK and p53 in the functioning of neuronal-committed progenitors. The blockade of signal transduction through JNK and p53 did not change the values of the studied indicators in the culture of CD56+ cells [Figure 2].
Thus, JNK- and p53-pathways play an important role in the regulation of the NSC-only cell cycle. They are responsible for maintaining multipotency and self-renewal of cells – maintaining a ‘deep reserve’ of CNS regeneration.[5,28]
Effect of JNK and p53 inhibitors on the secretion of growth factors by neuroglial cells under conditions of their optimal vital activity
The study of the involvement of JNK and p53 in the production of growth factors by different fractions of glial cells revealed ambiguous changes. The JNK and p53 inhibitors caused a significant decrease in neurotrophin secretion by ACSA-2+ cells (astrocytes) [Figure 3a], reaching 54.3% and 76.1% of the background, respectively.
The stimulating effect of CD11b+ cells (microglial cells) on the realisation of the growth potential of progenitors during the blockade of JNK and p53, in contrast, increased (to 116.7% and 122.7% of the baseline, respectively) [Figure 3c]. However, inhibitors of signalling molecules did not cause changes in the functioning of O4+ cells (oligodendrocytes) in any of the cases [Figure 3b].
Effect of JNK and p53 inhibitors on nervous tissue progenitor function in AN
The study of the effect of alcohol on the participation of JNK- and p53-pathways in the functioning of various types of nervous tissue progenitors has revealed several important phenomena. First, the addition of JNK and p53 inhibitors to ethanol culture media resulted in a significant increase in the ability of unfractionated SVZ cells to neurosphere formation and proliferative activity of NSC [Figures 1a and c]. Furthermore, the JNK blockade accelerated the NSC specialisation process to 123.6% of the control (ethanol medium without signalling molecule inhibitors) [Figure 1d]. There were no changes in CNP function during the inactivation of JNK and p53 in vitro [Figure 2].
Similar patterns were revealed in the cultivation of nervous tissue cells in alcoholised mice. The inactivation of JNK and p53 resulted in an increase in CFUNSC content and their proliferative activity in the culture of unfractionated SVZ cells [Figures 1a and c]. Besides, the JNK inhibitor also caused an increase in the NSC specialisation index [Figure 1d]. However, there were no changes in the regulation of NCP functions. The blockage of these signalling molecules in NCP of mice, which had long been treated with ethanol, did not affect the realisation of their growth potential [Figure 2].
The results indicate an inversion of the role of JNK and p53 in the regulation of NSC proliferation in AN.
Effect of JNK and p53 inhibitors on the secretion of growth factors by neuroglial cells in AN
Changes in the functioning of different types of neuroglia cells depending on their living conditions under JNK and p53 blockade were ambiguous. The inactivation of JNK and p53 of astrocytes in the presence of ethanol in vitro was accompanied by a drop in neurotrophins secretion (up to 45.1% and 78.4% of the control values, respectively). However, in mice that were long-term injected with ethyl alcohol, blockade of JNK and p53 in ACSA-2+ cells did not affect the secretion of humoral factors [Figure 1a].
Another phenomenology was observed in the study of the functioning of oligodendrocytes. The blockade of signal transduction through JNK and p53 in oligodendrocytes when exposed to ethanol in vitro, in contrast, led to increased production of neurotrophins (especially when using a JNK inhibitor) [Figure 3b]. While inactivation of JNK in oligodendrocytes of alcoholised animals caused a decrease in the secretion of neurotrophins (up to 84.1% of similar parameters in the control). The p53 inhibitor, in this case, did not affect the production of growth factors by oligodendrocytes.
Unlike macroglial cells, the microglial cell’s response was in all cases the same. The inactivation of signalling molecules by exposure to alcohol (both in vitro and in vivo) resulted in increased secretion of growth factors by CD11b+ cells. A particularly pronounced increase in this indicator was with the use of the JNK inhibitor (up to 150.0% and 156.2% of the control values, respectively) [Figure 3c].
DISCUSSION
The findings confirm the evidence that there are significant peculiarities in the role of JNK and p53 in the regulation of the cell cycle of different types of regeneration-competent cells.[8,13,14,28] It was found that JNK- and p53-pathways are responsible for the plasticity of the CNS – for maintaining the self-renewal, rapid proliferation and multiple differentiation potential of NSC. At the same time, it was found that these signalling molecules do not take a significant part in the functioning of the NCP (the most mobile part of tissue-specific regeneration).[6,9]
However, it is known that balanced neurogenesis can occur through the implementation of the functions of an exclusively multipotent NSCS (without the participation of NCP).[5,29] Therefore, the important role of JNK and p53 in the progression of the NSC cell cycle indicates the potential for their use as pharmacological targets [Figure 4].
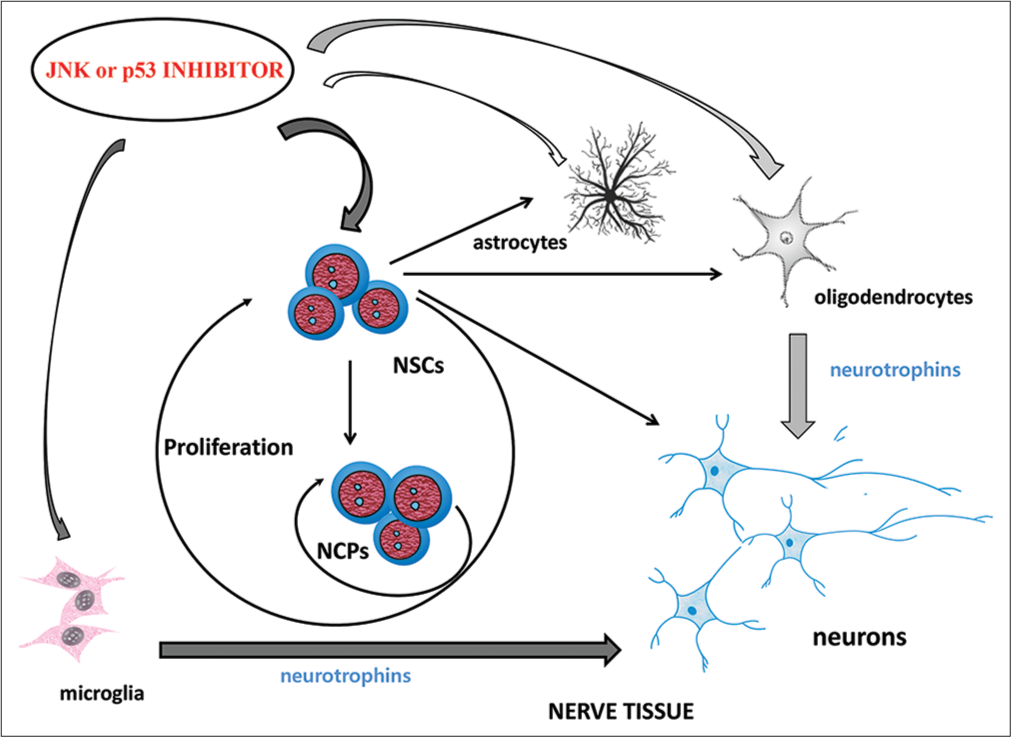
- Effect of JNK or p53 inhibitors on the regeneration-competent cells of the nervous tissue in AN. Gray arrows are a stimulating influence; white arrows are an inhibitory effect.
At the same time, it has been found that AN leads to the inversion of the role of JNK and p53 in the regulation of the proliferation of NSC. The blockage of these signalling molecules in conditions of ethanol intoxication can significantly increase the degree of realisation of the growth potential of NSC and, thus, stimulate neurogenesis. However, it should be borne in mind that such therapy can lead to the transformation of the intracellular signalling pattern characteristic of AN into a ‘starting’ (normal) state in the formed de novo precursors.[6,9,12] That is, to cause a ‘reverse’ inversion of the role of JNK and p53 in the regulation of NSC functions. In such conditions, the JNK and p53 inhibitors will, on the contrary, inhibit the implementation of the functions of these ‘intact’ progenitor cells.
Therefore, it is likely that JNK and p53 inhibitors in AN can have a therapeutic effect when used only during the manifestation of disease signs (and their withdrawal is necessary when clinical signs of recovery appear).
The detected increase in the secretion of neurotrophic growth factors by oligodendrocytes and microglia, when JNK and p53 are blocked in them under the conditions of alcohol exposure, also suggests the possibility of accelerating CNS repair as a result of inactivation of these signalling molecules. However, it should be taken into account that JNK and p53 inhibitors have an ambiguous effect on the functioning of astrocytes in the modelling of ethanol-induced neurodegeneration in vitro and in vivo. Further, research should determine how much this factor can be neglected.
Besides, the anti-mutagenic properties of the p53 protein (p53 – ‘guardian of the genome’)[30] are known. This determines the need to study in detail the potential carcinogenic hazard of using its activity modifiers as drugs. Moreover, it should be borne in mind that p53 can also be targeted through JNK.[13] However, it is only one of the directions of signal transduction through JNK,[31,32] which, in some cases, is not implemented. In addition, even the antiplastic properties of JNK inhibitors are known.[4] Therefore, the potential for drug (carcinogenic) safety of JNK activity/expression inhibitors is higher.
CONCLUSION
Our results reveal the feasibility of further study of the possibility of creating novel effective drugs for the therapy of AE based on JNK or p53 inhibitors. The most promising is the development of pharmacotherapeutic approaches using JNK activity/expression inhibitors.
Availability of data and materials
All data generated or analysed during this study and its supplementary information files are included in this published article.
Acknowledgments
We thank the Director of the Institute V.V. Zhdanov for providing research infrastructure for the work.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
The studies were carried out as part of the execution of the State task of the Ministry of Science and Higher Education of Russia on the topic No. FGWM-2022-0018. This work was supported by the Russian Foundation for Basic Research (RFBR) in the framework of scientific project No. 18-015-00013.
References
- Molecular targeted therapy: Treating cancer with specificity. Eur J Pharmacol. 2018;834:188-96.
- [CrossRef] [PubMed] [Google Scholar]
- Targeted therapies for myeloproliferative neoplasms. Biomark Res. 2019;7:15.
- [CrossRef] [PubMed] [Google Scholar]
- Targeted therapy and personalized medicine in gastrointestinal stromal tumors: Drug resistance, mechanisms, and treatment strategies. Onco Targets Ther. 2019;12:5123.
- [CrossRef] [PubMed] [Google Scholar]
- Targeted regulation of intracellular signal transduction in regeneration-competent cells: A new direction for therapy in regenerative medicine. Biointerface Res Appl Chem. 2021;11:12238-51.
- [CrossRef] [Google Scholar]
- Halogenated (Cl-ion) songorine is a new original agonist of fibroblast growth factor receptors of neuronal-committed progenitors possessing neuroregenerative effect after cerebral ischemia and hypoxia in experimental animals. Biointerface Res Appl Chem. 2019;9:4317-26.
- [CrossRef] [Google Scholar]
- Intracellular signalling molecules of nerve tissue progenitors as pharmacological targets for treatment of ethanol-induced neurodegeneration. J Basic Clin Physiol Pharmacol. 2021;33:305-15.
- [CrossRef] [PubMed] [Google Scholar]
- Wound healing properties of the protein kinase a inhibitor and the mechanisms of their development. Bangladesh J Pharmacol. 2021;16:19-26.
- [CrossRef] [Google Scholar]
- Peculiarities of intracellular signal transduction in the regulation of functions of mesenchymal, neural, and hematopoietic progenitor cells. Bull Exp Biol Med. 2019;167:201-6.
- [CrossRef] [PubMed] [Google Scholar]
- Targeting cAMP-pathway in regeneration-competent cells of nervous tissue: Potential to create a novel drug for treatment of ethanol-induced neurodegeneration. Cent Nerv Syst Agents Med. 2021;21:172-80.
- [CrossRef] [PubMed] [Google Scholar]
- Neurotrophically induced mesenchymal progenitor cells derived from induced pluripotent stem cells enhance neuritogenesis via neurotrophin and cytokine production. Stem Cells Transl Med. 2018;7:45-58.
- [CrossRef] [PubMed] [Google Scholar]
- The neuropathology of the adult cerebellum. Handb Clin Neurol. 2018;154:129-49.
- [CrossRef] [PubMed] [Google Scholar]
- Prospects for the use of NF-кb inhibitors to stimulate the functions of regeneration-competent cells of nerve tissue and neuroregeneration in ethanol-induced neurodegeneration. Biointerface Res Appl Chem. 2021;11:8065-74.
- [CrossRef] [Google Scholar]
- Targeting p53 via JNK pathway: A novel role of RITA for apoptotic signalling in multiple myeloma. PLoS One. 2012;7:e30215.
- [CrossRef] [PubMed] [Google Scholar]
- Ginsenoside Rh1 alleviates HK-2 apoptosis by inhibiting ROS and the JNK/p53 pathways. Evid Based Complement Alternat Med. 2020;2020:3401067.
- [CrossRef] [PubMed] [Google Scholar]
- p53 predominantly regulates IL-6 production and suppresses synovial inflammation in fibroblast-like synoviocytes and adjuvant-induced arthritis. Arthritis Res Ther. 2016;18:271.
- [CrossRef] [PubMed] [Google Scholar]
- Expression patterns of immature neuronal markers PSA-NCAM, CRMP-4 and NeuroD in the hippocampus of young adult and aged rodents. J Neurosci Res. 2002;70:327-34.
- [CrossRef] [PubMed] [Google Scholar]
- Anti-ACSA-2 defines a novel monoclonal antibody for prospective isolation of living neonatal and adult astrocytes. Glia. 2017;65:990-1004.
- [CrossRef] [PubMed] [Google Scholar]
- Polysialylation at early stages of oligodendrocyte differentiation promotes myelin repair. J Neurosci. 2017;37:8131-41.
- [CrossRef] [PubMed] [Google Scholar]
- Analysis of microglia and monocyte-derived macrophages from the central nervous system by flow cytometry. J Vis Exp. 2017;22:55781.
- [CrossRef] [PubMed] [Google Scholar]
- Experimental design and analysis and theirreporting: New guidance for publication in BJP. Br J Pharmacol. 2015;172:3461-71.
- [CrossRef] [PubMed] [Google Scholar]
- Phenotype overlap in glial cell populations: Astroglia, oligodendroglia and NG-2(+) cells. Front Neuroanat. 2015;9:49.
- [CrossRef] [PubMed] [Google Scholar]
- Neurotrophins and glial cell line-derived neurotrophic factor in the ovary: Physiological and pathophysiological implications. Hum Reprod Update. 2019;25:224-42.
- [CrossRef] [PubMed] [Google Scholar]
- CNTF-STAT3-IL-6 axis mediates neuroinflammatory cascade across schwann cell-neuron-microglia. Cell Rep. 2020;31:107657.
- [CrossRef] [PubMed] [Google Scholar]
- TNF-alpha-sensitive brain pericytes activate microglia by releasing IL-6 through cooperation between IkappaB-NFkappaB and JAK-STAT3 pathways. Brain Res. 2018;1692:34-44.
- [CrossRef] [PubMed] [Google Scholar]
- Neuroimmune and epigenetic mechanisms underlying persistent loss of hippocampal neurogenesis following adolescent intermittent ethanol exposure. Curr Opin Pharmacol. 2020;50:9-16.
- [CrossRef] [PubMed] [Google Scholar]
- Role of MCP-1 and CCR2 in ethanol-induced neuroinflammation and neurodegeneration in the developing brain. J Neuroinflammation. 2018;15:197.
- [CrossRef] [PubMed] [Google Scholar]
- Role of JNK and p53 in implementation of functions of various types of regeneration-competent cells of the nervous tissue. Bull Exp Biol Med. 2021;171:333-7.
- [CrossRef] [PubMed] [Google Scholar]
- Regulation of neurogenesis in the adult and aging brain. Curr Opin Neurobiol. 2018;53:131-8.
- [CrossRef] [PubMed] [Google Scholar]
- Mutant p53 as a guardian of the cancer cell. Cell Death Differ. 2019;26:199-212.
- [CrossRef] [PubMed] [Google Scholar]
- JNK pathway signalling: A novel and smarter therapeutic targets for various biological diseases. Future Med Chem. 2015;7:2065-86.
- [CrossRef] [PubMed] [Google Scholar]
- Cardioprotective effect of erythropoietin in rats with acute myocardial infarction through JNK pathway. Eur Rev Med Pharmacol Sci. 2019;23:153-60.
- [Google Scholar]







