Translate this page into:
Angiogenesis-modulating properties of ethanolic extract of Ferula assa-foetida oleo-gum-resin
*Corresponding author: Gholamreza Anani Sarab, Cellular and Molecular Research Center, Birjand University of Medical Sciences, Birjand, Iran ghansa@yahoo.com
-
Received: ,
Accepted: ,
How to cite this article: Yazdanipour N, Khorashadizadeh M, Sarab GA. Angiogenesis-modulating properties of ethanolic extract of Ferula assa-foetida oleo-gum-resin. Indian J Physiol Pharmacol 2021;65:177-87.
Abstract
Objectives:
Angiogenesis has been known to have a critical role in the tumour growth. Different studies indicated that angiogenesis was stimulated by hypoxia. In the present study, we assessed the anti-angiogenesis activity of ethanolic extract of Ferula assa-foetida (EEFA) on hypoxic-induced human umbilical vein endothelial cells (HUVECs).
Materials and Methods:
The F. assa-foetida gum extract was characterised by total phenolic contents (TPC) and total flavonoids content (TFC). The active compounds of EEFA were determined by high-performance liquid chromatography (HPLC). Then, cytotoxic effects of EEFA on the growth of HUVECs were assessed using MTT assay, wound healing and cell cycle analysis. The expression of Vascular endothelial growth factor (VEGF), Akt,HIF-1, VEGF receptor 1 (VEGFR-1) and VEGFR-2 genes was also quantified by Real-Time PCR. GeneMANIA and EnrichR databases were used to predict gene network interactions for the studied genes and their mechanism.
Results:
The TFC and TPC of the extract were 26 mg gallic acid equivalent per gram of extract and 5.45 mg quercetin/g, respectively. HPLC analysis revealed the presence of anti-angiogenic components in EEFA. Our data showed that EEFA had no cytotoxicity effect on HUVECs. The obtained results also indicated that EEFA prevented the proliferation and migration of HUVECs. Expression analysis showed that EEFA significantly decreased the VEGF-A mRNA level in the hypoxia-induced HUVECs. No change was found in the VEGFR-2 gene expression following treatment with EEFA in the HUVECs. However, the significantly upregulation of the VEGFR-1 gene expression was observed in the EEFA-treated HUVECs. The bioinformatics analysis of gene-gene interaction network also showed that the studied genes play an essential role in the regulatory pathways of angiogenesis and cancer.
Conclusion:
These findings provided evidence about the anti-angiogenesis role of EEFA, suggesting that this could be considered in the cancer therapy.
Keywords
Hypoxic-induced human umbilical vein endothelial cell
Vascular endothelial growth factor
Vascular endothelial growth factor receptor-1
Ferula assa-foetida
Anti-angiogenesis
Hypoxia
INTRODUCTION
Angiogenesis is a critical biological mechanism in the body. It is necessary to perform a precisely balanced interaction between angiogenic stimulators and angiogenesis-regulating inhibitors. This interaction was associated with a wide range of angiogenesis-dependent diseases including cancer and non-neoplastic disorders such as rheumatoid arthritis atherosclerosis.[1] Angiogenesis relies on the endothelial cells. These cells are the essential and permanent elements of the vascular system. In angiogenesis, the capillary basement membrane is enzymatically compromised to allow endothelial cells to spread. Then, through the proliferation of endothelial cells and migration toward the source signal, these processes reorganised the formation of endothelial cell tubes (tubulogenesis).[2]
Hypoxia, due to poor oxygen delivery, stimulated angiogenesis.[3] Hypoxia is well established as the major factor driving tumour angiogenesis. Under hypoxic conditions, Heterodimer transcription factor hypoxiainducible factor 1 alpha (HIF-1α) degradation was inhibited and vascular endothelial growth factor (VEGF) expression was induced through the binding of HIF-1 to the hypoxia response element located on the upstream region of the VEGF gene.[4,5] This upregulation induced the angiogenesis, leading to neovascularisation.[6] VEGF mainly exerts its biological effects by interacting with its unique VEGFR receptors. VEGF receptors on the membrane of endothelial cells consist of two different receptor groups, VEGF receptor 1 (VEGFR1) and VEGFR2. These receptors have been known to be active in the angiogenesis.[7] VEGFR-2 mediated the endothelial cells development and their permeability to cells and molecules through the binding of VEGFs. VEGFR-1 has been identified to have a regulatory effect in the angiogenesis.[8,9] Regulation of VEGF/VEGFR was suggested as a crucial technique for cancer anti-angiogenic therapy.[10-12]
Different studies indicated that the intake of natural products prevented the production and growth of cancer.[13] The natural products were known to be a suitable source of angiogenesis regulation molecules.[14] For instance, some angiogenesis regulators including farnesiferol C and galbanic acid have been previously isolated from Ferula assa-foetida.[15,16] Old conventional phytomedicine assa-foetida is an oleo-gum-resin derived from the root exudate of the endemic medicinal plant of Iran.[17] F. assafoetida was also regarded as the primary source of assafoetida.[18] Assa-foetida is commonly used to treat various illnesses including allergies, intestinal parasites and gastrointestinal disorders.[17] The biological effects of this plant are due to the presence of phenols and flavonoids in its extract.[19]
The combination of multiple anti-angiogenic agents has been considered as the novel approach in the treatment of the diseases.[20] Hence, we aimed to investigate the potency of the oleo-gum-resin cocktail ingredients F. assa-foetida, a native plant to South Khorasan, in targeting angiogenesis. In this study, the anti-angiogenic ability of the ethanolic extract of F. assa-foetida was investigated in human umbilical vein endothelial cells (HUVECs). Furthermore, the molecular behaviour of the endothelial cells under hypoxic conditions was evaluated. The present study results could increase our understanding about the possible anti-angiogenic effect of the extract of F. assa-foetida under hypoxic conditions.
MATERIALS AND METHODS
Cell culture and hypoxic conditions preparation
HUVECs were used as an in vitro model to investigate the mechanisms influencing on the angiogenesis during hypoxia. HUVECs were donated by Dr. Abdolreza Ardeshirylajimi (Shahid Beheshti University of Medical Sciences, Tehran, Iran). These cells were grown in the DMEM medium including 10% heat-inactivated bovine fetal serum (FBS), 100 U/ml penicillin and 100 mg/ml streptomycin. These cells were cultured at a humidified atmospheric incubator containing 37°C and 5% CO2 for 3–5 passages. HUVECs were serum-starved for 4 h and then, they were exposed to 1% O2 for 24 h using modular gas controller incubator (Cytation 3, BioTeck, USA).
Isolation of extracts
F. assa-foetida was collected during the summer from Tabas region (Southern Khorasan Province, Iran). The plant species was certified by an experienced applied medicinal plant researcher, Mr. Mohsen Pouyan, at the Birjand Agricultural Research Center (Birjand, Iran) and voucher number of the specimen was 2365.
In this method, about 35 g of dried asafetida, the oleo-gum-resin of the herbaceous plant F. assa-foetida root exudate, was macerated by soaking assa-foetida in absolute ethanol and distilled water at a ratio of 1:1. It was then homogenised with a mixer and kept in a shaker incubator (28℃, 145 rpm) for 2 days. After maceration, the solution was filtered through the Whatman filter No.4 and centrifuged at 4000 g for 10 min and the supernatant was collected. The filtrate was concentrated using a rotary evaporator at 38℃, the remaining solution was then dried at 37℃ weighted and maintained at −80℃ throughout the study.
Quantification of total phenolic contents (TPCs)
TPCs of the oleo-gum-resin extract were measured through Folin-Ciocalteu test. First, 2.8 ml of DW, 2 ml of Na2CO3 and 0.1 ml of Folin- Ciocalteu (50%) were added to 0.2 ml of the extract (10 mg/ml). Then, the solution was incubated for half an hour and its absorbance was calculated (750 nm) using spectrophotometer. Finally, to measure the TPC, Gallic acid was utilised as a standard and this content was calculated based on mg gallic acid equivalents (GAE) per gram of sample in dry weight (mg/g).[1]
Quantification of total flavonoids content (TFC)
TFC was estimated by diluting the extract to 100 μg/ml, using 95% alcohol and 0.5 ml of that was mixed with 0.1 ml of aluminium chloride (10%) and 0.1 ml of potassium acetate (0.1 mM). Then, the solution remained at RT for 30 min. The flavonoid content was calculated as mg quercetin equivalent (QE/g) using the linear equation based on the quercetin standard calibration curve.[2]
Phytochemical analysis
The preliminary phytochemical screening of the ethanolic extract of F. assa-foetida oleo gum resin was performed using Spectrophotometry and Fourier Transform Infrared Spectroscopy (FTIR) Analysis, gas chromatography mass spectrometry (GC-MS) and high-performance liquid chromatography (HPLC).
Spectrophotometric and FTIR analysis
Ultraviolet-Visible (UV-Vis) spectra were recorded within the wavelength range between 190 and 800 nm using a UV-Vis spectrophotometer (Shimadzu UV-24001 PC) to analyse the quantification of the chemical structure of the compound.
Infrared (IR) spectra were recorded within the wavelength range between 4000 and 600 nm to identify the functional groups presented in the extract using a FTIR Nicolet 6700 FT-IR (Thermo scientific).
HPLC analysis
HPLC chromatograms obtained on an Agilent 1200 series HPLC system (Agilent, United States). The stationary phase was Agilent C18 column (250 mm–4.6 mm id, 5 mm). The micro-fractions were collected using Agilent 1200 series fraction collector and then, they were processed with the Agilent OpenLab CDS Chemstation Edition software (Agilent).
GC-MS analysis
GC-MS was used to identify the compounds in the F. assa-foetida extracts. The GC-MS analysis of F. assa-foetida was performed using a GC-MS equipment. Experimental conditions of GC-MS system were as followed: Elite-5 MS (5% diphenyl/95% dimethyl poly siloxane). The dimension of 30 × 0.25 mm ×0.25 mm df was used and flow rate of mobile phase (carrier gas: He) was set at 1.0 ml/min and an injection volume of 2 ml was employed (split ratio of 10:1 injector temperature 250°).[21]
The temperature of the oven was programmed from 110°C (2 min isothermal) with a rise of 10°C/min to 200°C, then 5°C/min to 280°C and finished with a 9 min isothermal temperature of 280°C.
A mass spectrum, scan interval of 0.5 s and fragments of 45–450 Da were collected at 70 eV. The temperature of the source and inlet line was 200°C and the mass spectrum was observed at 36 min. Identification was performed according to a custom database created from the dictionary of Natural Products. We identified the unknown individual constituents based on the direct comparison of the retention time (RT) and their mass spectra of known compounds stored in the database library.
Cell viability using MTT assay
The MTT test was performed to determine the in vitro cytotoxicity of the ethanolic extract of Ferula assa-foetida (EEFA). HUVECs (1 × 104 cells/well) were seeded in 96 well-cultivated plates and incubated for 1 day. Then, the cells were treated with a 200 ml serum-free culture medium containing different concentrations of EEFA (0, 125, 250, 500 and 1000 μg/ml) for 24 h at the hypoxic conditions. After that, 20 ml of MTT reagent (5 mg/ml) was added to each well and incubated at 37° for 4 h. The media were then removed and formazan crystals were dissolved in 100 ml of Dimethyl sulfoxide. Solubilised formazan absorbance was measured at 570 and 630 nm using the Epoch Microplate Spectrophotometer (Biotek Instrument, USA). The untreated cells were considered to have 100% viability.[22]
Scratch wound healing assay
Proliferation/migration capabilities of HUVECs were assessed using a scratch wound-healing assay.[23] The HUVECs were seeded in a 24 well plate pre-coated with gelatin in a humidified atmosphere containing 5% CO2 for 1 h at a concentration of 2 × 105 cells/ml. Then, they were cultivated in a medium containing 10% FBS to form a confluent monolayer. The cell monolayers were scratched in a straight line pattern using a strile 100 ml pipette-tip to create a wound. The cell debris was removed with PBS. Afterward, the edge of the scratch was smoothed by washing through a serum-free medium. Images were immediately collected, labelled (0 h) and incubated with 250 μg/ml and without EEFA for 24 h at 1% O2 and 5% CO2. Cells without treatment were used as the control group. A reduction in the scraped region was regarded to indicate the wound healing and cells migration.[24,25] To quantify the closure of the scratch, the difference between wound width at time 0 and time 24 h was determined. Each well was marked below the plate surface by drawing a vertical line. This process identified the same scratched area to take consistent pictures. Scratch area was measured using Image J software (National Institutes of Health, Bethesda, MD, USA). Migration rate was calculated according to the following equation:
The scratch closure rate = (scratch width at 0 h−the remaining scratch width at 24 h)/scratch width in 0 h × 100
Cell cycle analysis
After incubation of HUVECs under hypoxic conditions, we observed no cytotoxicity at an optimum dose of EEFA (250 μg/ml) for 24 h. These cells were prepared as a single cell suspension by trypsinisation. Then, 4 ml of 70% cold ethanol was added to the cells to fix cells and then, they were incubated at −20°C for overnight. After that, these cells were washed twice with cold PBS to remove residual ethanol. To analyse the cell cycle, the cell pellets were resuspended at 0.5 ml of PBS containing 50 mg/ml of propodeum iodide and 100 mg/ml of RNase. Afterward, they were incubated and studied using the FACS Scan flow cytometer (Becton Dickinson, San Jose, CA, USA). For each sample, data from 10,000 cells were measured and the cell percentages in different populations were calculated. Then, data were analysed using FSC express software (Ver 5).
Analysis of gene expression by real-time PCR
HUVECs were seeded at a concentration of 2 × 105 cells in a 6-well plate and treated with EEFA (250 μg/ml) for 24 h. Total RNAs were isolated from the TRIzol reagent (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. Complementary DNA (cDNA) was prepared using the Revert Aid First-Strand cDNA Synthesis Kit (K1621, Thermo Fisher Scientific, CA, USA). Real-time PCR reactions were performed to determine the expression level of HIF-1a, VEGF-A, VEGFR-1, VEGFR-2 and Protein kinase B (Akt) using SYBR Green qPCR Master Mix (A325402, Ampliqon, Denmark) on the Step-One Real-time PCR system. The primer sequences are presented in [Table 1]. Glyceraldehyde 3-phosphate dehydrogenase gene level was used as endogenous control and analysed under the same conditions. The relative abundance of detected RNAs was calculated using the 2−ΔCt method.
| Gene | Forward primer | Reverse primer | Product (bp) |
|---|---|---|---|
| HIF-1a | TGAGGAAATGAGAGAAATGCTTACA | ACACTGAGGTTGGTTACTGTTGGT | 207 |
| VEGF-A | CTACCTCCACCATGCCAAGT | CACACAGGATGGCTTGAAGA | 186 |
| VEGFR-1 | CTGGGCAGCAGACAAATCCT | AAAGTCACACCTTGCTTCGGA | 120 |
| VEGFR-2 | GTGATCGGAAATGACACTGGAG | CATGTTGGTCACTAACAGAAGCA | 123 |
| Akt | TCCTCCTCAAGAATGATGGCA | GTGCGTTCGATGACAGTGGT | 180 |
| GAPDH | AGATCATCAGCAATGCCTCCT | GCCATCCACAGTCTTCTGGG | 136 |
HIF-1:Heterodimer transcription factor hypoxia-inducible factor 1, VEGF:Vascular endothelial growth factor, VEGFR: Vascular endothelial growth factor receptor, Akt: Protein kinase B, GAPDH: Glyceraldehyde 3-phosphate dehydrogenase
Bioinformatics data mining
The molecular functional network maps of canonical pathways including co-expression, physical interaction, genetic interaction and predicted networks of VEGF, Akt, HIF-1α, VEGFR-1 and VEGFR-2 genes were analysed by GeneMANIA (http://genemania.org/). The gene network was then analysed using the Kyoto Encyclopedia of Genes and Genomes pathway enrichment to compare the specific gene with the whole reference gene background using the Database for Annotation, Visualisation and Integrated Discovery (DAVID; version 6.7, http://david.abcc.ncifcrf.gov). The number >2 and P < 0.05 were selected as the cutoff criteria. The selected genes analyses were performed through the bioinformatics resource EnrichR,[26] which was chosen using a complete list of angiogenesis-related signalling pathways.
Statistical analysis
All values were analysed using the SPSS software version 16.0 (SPSS Inc., Chicago, IL, USA). The results obtained from each experiment were expressed as mean ± SD. Each of the conditions was performed at least for three independent experiments. The individual scratch width (micrometre, mean and standard deviation) was measured using the Image J software for quantification and statistical analysis. Flow Cytometry and Real-Time PCR were evaluated by FlowJo V10 and Rest 2009 software, respectively. The group differences were then analysed by one-way analysis of variance and a Tukey test. P < 0.05 was considered statistically significant.
RESULTS
TPC and TFC of the extract
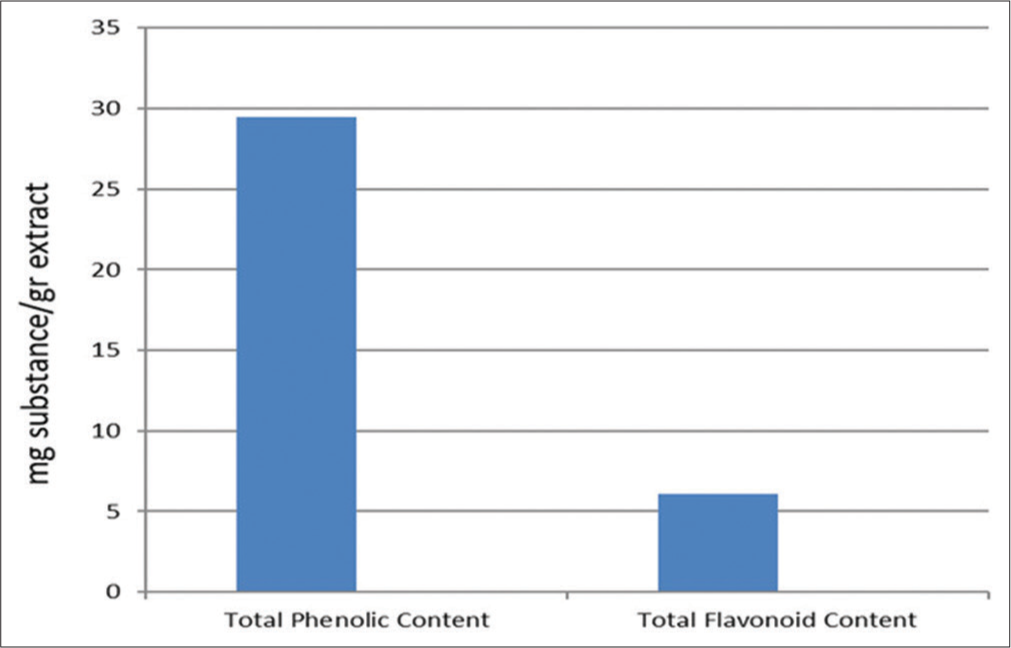
The TFC and TPC of 1 g of the ethanolic extract of F. assa-foetida oleo gum resin are illustrated in [Figure 1]. The TPC value of the extract, calculated from the calibration curve (R2 = 0.998), was 29.5 mg of GAE/g and the total flavonoid content (R2 = 0.999) was 6.1 mg QE/g.
-
Ferula assa-foetida gum extract total phenolic content (mg GAE/g extract) and total flavonoid content (mg quercetin/g extract).
Spectrophotometric analysis
F. assa-foetida extracts were analysed using UV-Vis spectrometry. As seen in the UV-VIS spectrophotometry, about 254 nm indicated there was benzene and phenolic ring in the extract and it contained the effective compounds and polyphenol and flavonoid metabolites.
FTIR analysis
The FTIR spectra of the extracts are shown in [Figure 2]. As seen in the IR spectrum (FTIR spectroscopy), the hydroxy and amine functional group (OH, NH2 at 3398 cm−1), the aliphatic group (CH) at 2854, 2924 and 2962 cm−1, the carbonyl group (C = O) at 1738 cm−1, the aromatic group (C = C) at 1664 cm−1 and the ether group (CO) at 1078 cm−1 indicated there were the polyphenol and flavonoid metabolites in the extract.
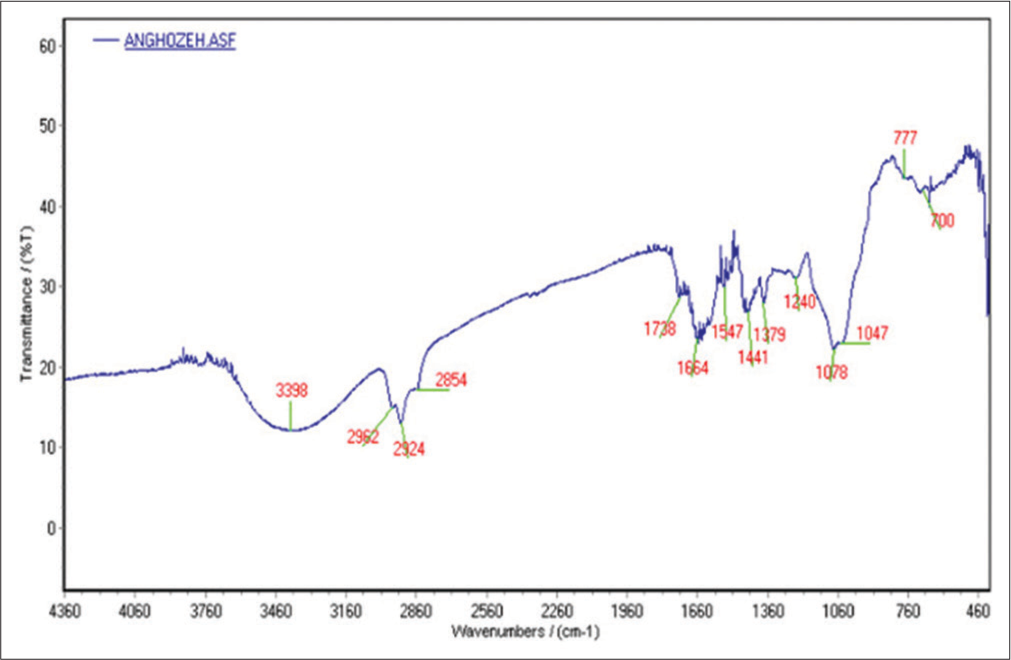
- Fourier transform infrared spectroscopy spectra of Ferula assa-foetida.
HPLC analysis
HPLC profile of F. assa-foetida was carried out using methanol and water (35:65, v/v) solvent system to confirm the presence of various 17 peaks at different tR values. We obtained peak area at 254 nm [Figure 3].

- High-performance liquid chromatography chromatogram of the ethanolic extract of Ferula assa-foetida, detection wavelength at 254 nm.
As can be seen in HPLC at l max = 254 nm, 17 species emerged from the C18 column, with approximately ten species having the highest relative abundance. Compared to the standards, the presence of active compounds of ferulic acid (26.7 min) with relative frequency percentage (21.55%), gummosin (24.917 min) with relative frequency percentage (3.39%), farnesiferol (22.467 min) with relative frequency percentage (6.57%), Galbanic acid (19.533 min) with relative frequency (7.33%), samarcandin (17.7 min) with relative frequency (11.35%), asafoetida (7.25 min) with relative frequency (3.41%) and umbelliprenin (4.283 min) was identified with a relative frequency percentage (6.66%).
GC-MS analysis
GC-MS was used to determine the content of various compounds in the extract. Many peaks were observed in the chromatograms at various RTs. The RTs and chromatogram peaks obtained results indicated that several compounds were within the F. assa-foetida oleo-gum-resin and compared with reference standards and confirmed with National Institute of Standards and Technology library.
GC-MS analysis of the ethanolic extract of the root of F. assa-foetida revealed that the presence of 72 types entered the mass device after leaving the column and the pattern of mass spectrum and molar mass of each combination was obtained. Among them, ten combinations were selected with the highest frequency in its time 24.76–29.03 min.
The criterion was according to the comparison with the digital library model of the device [Figure 4]. The GC-MS analysis of F. assa-foetida is reported in [Table 2].

- Gas chromatography mass spectrometry chromatogram of the hydroalcoholic extract of Ferula assa-foetida (see also Table 2).
| Peak | RT (min) | Compound name | Area % | Molecular formula |
|---|---|---|---|---|
| 1 | 24.76 | 3,4-Di-O-methyl-L-arabinopyranose | 10.03 | C7H14O5 |
| 2 | 25.933 | Pterin-6-carboxylic acid | 8.73 | C7H5N5O3 |
| 3 | 26.724 | 1,2-Dithiolane | 100 | C7H14O5 |
| 4 | 27.115 | Disulphide, bis (1-methylpropyl) |
10.51 | C8H18S2 |
| 5 | 27.481 | Dimethoxyamphetamine, 2,5- | 60.05 | C11H17NO2 |
| 6 | 27.822 | 1,4-Dithiane-2,5-dione, 3,6-dimethyl | 18.82 | C6H8O2S2 |
| 7 | 28.005 | Cyclohexane, (2-nitro-2-propenyl) |
23.81 | C9H15NO2 |
| 8 | 28.163 | Propionic acid, 2,3-dihydroxy | 34.33 | C10H14O5 |
| 9 | 28.688 | Caryophyllene | 25.87 | C15H24 |
| 10 | 29.037 | α-Caryophyllene α | 7.86 | C15H24 |
GC-MS: Gas chromatography-mass spectrometry, RT: Retention time
The effect of EEFA on the proliferation of HUVECs
Because endothelial cell proliferation is necessary for angiogenesis, we investigate the inhibitory effect of EEFA on HUVECs growth. A viability curve was obtained with the MTT assay. EEFA concentration of 125, 250 and 500 μg/ ml did not significantly change HUVECs viability but with 1000 μg/ml reduced the cell viability compared with the control after incubation in hypoxia for 24 h (p < 0.01); [Figure 5]). To further verify these results, the effect of EEFA on HUVEC confluence was observed through phase-contrast microscopy. 500 μg/ml EEFA treatment led to change in morphology and cause shrinking cells.
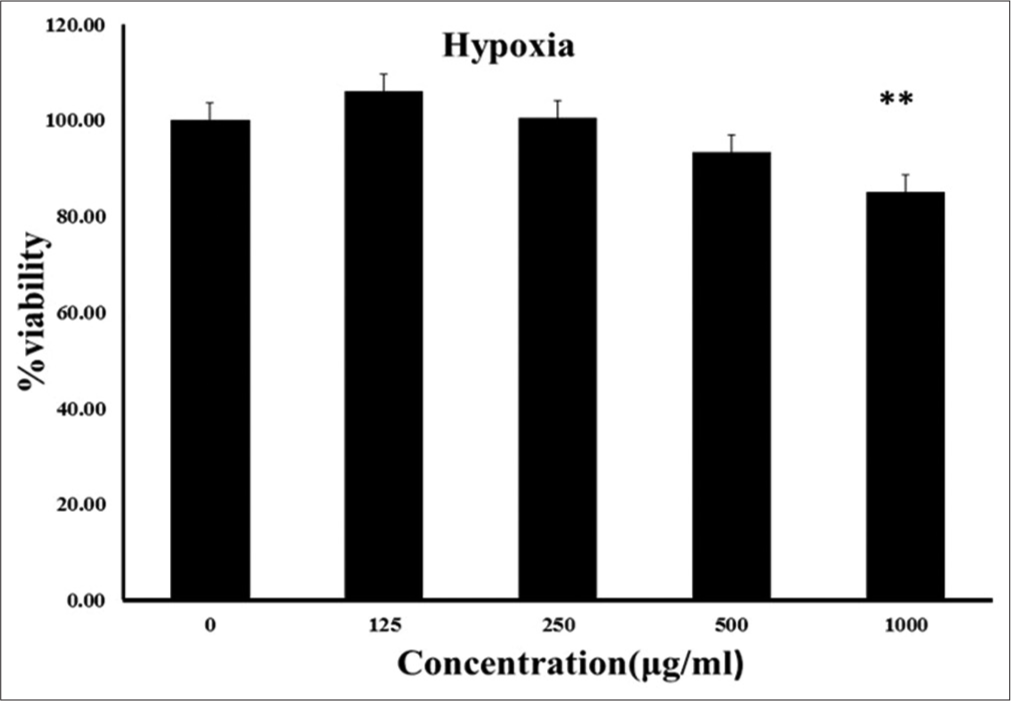
- Human umbilical vein endothelial cells were treated by 0, 125, 250, 500 and 1000 g/ml of ethanolic extract of Ferula assafoetida for 24 h. Cell viability was determined by MTT assay as described in the Methods section. The given values were expressed as the mean ± SD of five independent experiments.
The effect of EEFA on migration rate of hypoxia-induced HUVECs
Wound closure was determined from the 0- to 24-h scratch zone distinction to assess the impact of the EEFA on cell migration. Measuring the distance between the edges showed that EEFA significantly reduced the migration rate at the concentration of 250 μg/ml after 24 h as compared to control (P control vs. EEFA 250 μg/ml). These data added a notion that EEFA could inhibit the angiogenesis through suppressing endothelial cells migration [Figure 6].

- The confluent monolayers of human umbilical vein endothelial cells were Scratched under hypoxia condition and the repair was microscopically monitored after 24 h of treatment with ethanolic extract of Ferula assa-foetida (EEFA) (250 g). The percentage of the scratch area closed = (scratch width at 0 h-the remaining scratch width at 24 h)/scratch width at 0h x100%. Using t-test represent a significant difference between the exposed EEFA cells and the controls (***p<0.002).
The effect of EEFA on cell cycle progression
The results showed that EEFA-treated HUVEC cells were arrested in the G2/M phase [Figure 7]. This was accompanied by a significant decrease in the G1 phase as compared to the untreated control cells.
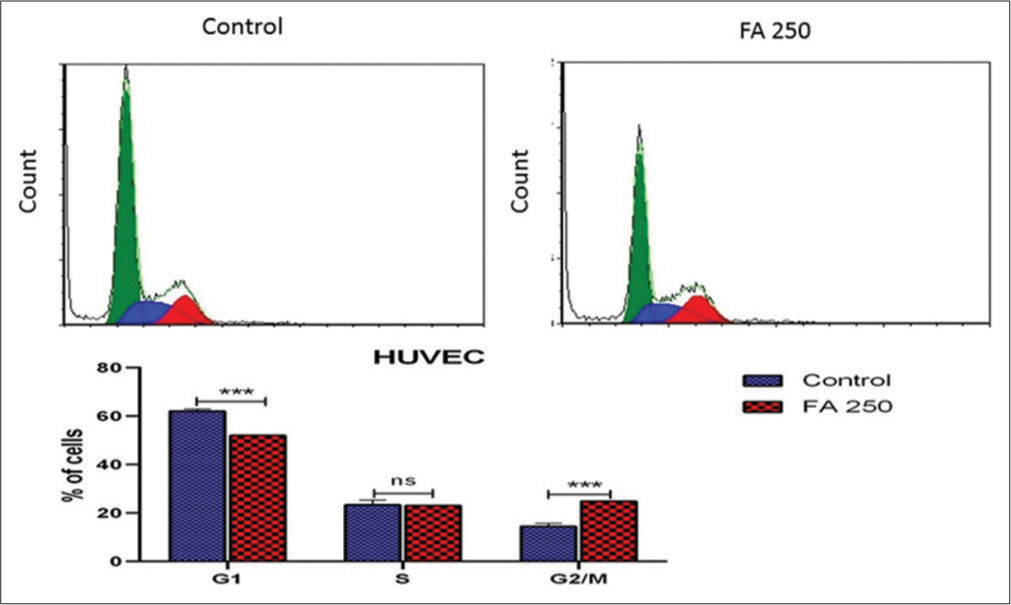
- Effect of the ethanolic extract of Ferula assa-foetida (EEFA) on the progression of hypoxia-induced human umbilical vein endothelial cells (HUVECs) in the cell cycle. The cell cycle was analysed by PI staining for DNA content. EEFA caused G2/M arrest in hypoxic-stimulated HUVECs The DNA histograms are representative of three separate experiments. The data are expressed as a mean ± SD.
Analysis of gene expression by real-time PCR
Real-time PCR was used to measure the expression level of the angiogenesis genes in EEFA-treated HUVECs induced by hypoxia. The obtained results indicated that EEFA significantly decreased the VEGF-A mRNA level as early as 24 h after treatment. However, we observed the non-significantly decreased expression of the HIF-1α and Akt genes. Additional data analysis of the RT-PCR results showed that the transcript level of VEGFR-1 significantly increased in EEFA-treated HUVECs induced by hypoxia. However, EEFA did not show to have the effect on the expression of the VEGFR-2 [Figure 8].

- The mRNA expression of cell growth-associated factors following EEFA treatment in hypoxia condition for 24 h: EEFA impact on (a) VEGF, (b) VEGFR-1, (c) VEGFR-2, (d) HIF-1, (e) Akt expression in HUVEC and (f) Fold changes and p-values of genes; Data are shown as mean ± SEM. EEFA: Ethanolic extract of Ferula assa-foetida, HIF-1: Heterodimer transcription factor hypoxia-inducible factor 1, VEGF: Vascular endothelial growth factor, VEGFR: Vascular endothelial growth factor receptor, Akt: Protein kinase B
Networks construction and analysis
The network of VEGF, Akt, HIF-1α, VEGFR-1 and VEGFR-2 genes was analysed. The co-localisation, co-expression, pathway, physical and predictive interactions of the VEGF, Akt, HIF-1α, VEGFR-1 and VEGFR-2 are shown in [Figure 9a]. The most enriched pathways related to the angiogenesis and cancer were observed for VEGF, HIF-1a, ErbB and PI3K/Akt [Figure 9b]. These gene clusters were corresponding to the top enriched biological pathways.

- (a) The molecular functional network map of canonical pathways including expression, physical interaction, and predicted networks of VEGF, Akt, HIF-1, VEGFR-1 and VEGFR-2 genes analysed by GeneMANIA (http://genemania.org/) tool. (b) EnrichR Clustergram of the most significant comparisons; clusters of 40 gene targets of miRNAs to the 10 top enriched biological pathway terms are shown. HIF-1: Heterodimer transcription factor hypoxia-inducible factor 1, VEGF: Vascular endothelial growth factor, VEGFR: Vascular endothelial growth factor receptor, Akt: Protein kinase B.
DISCUSSION
Angiogenesis, the formation of new blood vessels, has been demonstrated to play an important role in tumour growth. It has been estimated that more than 90% of deaths from cancer patients are related to angiogenesis, invasion and critical organ cancer metastases.[27] Angiogenesis is a crucial mechanism that mediates metastases through PI3K/Akt and HIF1α/VEGF signal cascades.[28] Hypoxia, by reducing oxygen supply, influenced the genes involving in the angiogenesis.[3-5] Thus, inhibitors of hypoxia-regulated genes, including VEGF, have been revealed to suppress tumour angiogenesis. These inhibitors could be considered as a new class of drugs to manage the cancer. Different studies suggested that certain natural ingredients may have anti-angiogenesis and anti-tumour benefits.[20,21] In the present study, the effect of EEFA on angiogenesis-related gene expression is evaluated in hypoxia-induced HUVECs for the 1st time.
Phenolic and flavonoid compounds have considerable health-improving impacts and are essential elements in pharmaceutical and medicinal applications. This fact is ascribed to their anti-inflammatory, anti-cancer and anti-mutagenic aspects.[1] We measured the total amount of these components and the results were almost in agreement with previous studies. In an investigation, the TPC and TFC of the alcoholic extract of F. Assa-feotida were shown to be 34.2 and 7.2 mg per gram of the extract.[3] Another study that measured the TPC of a species of Ferula, revealed that it contained 36.4 mg of phenolic components per gram of the extract.[4]
The investigation of biologically active compounds of EEFA was performed using LC-MS and GC-MS and HPLC. The HPLC analysis revealed that ferulic acid, farnesiferol and Galbanic acid were present in EEFA.
In the previous studies, these components have been revealed to have anti-angiogenesis and anticancer activities.[15,16,21,29] Although the anti-angiogenic agents have a limited effect individually, the beneficial natural products contained a wide variety of chemicals with a synergistic action. Therefore, HUVECs treated with EEFA in the present study.
The results obtained from the measurement of toxicity effects of EEFA did not show any cytotoxicity in the cell viability of HUVECs. Although 0, 125, 250, 500 and 1000 μg/ ml of EEFA were used up to 24 h, it limited the proliferation of endothelial hypoxia-induced cells [Figure 4]. Besides, the evaluation of wound revealed that EEFA substantially decreased the migration rate of hypoxia-induced HUVECs [Figure 5]. We also observed the effects of EEFA therapy on the development of hypoxia-induced HUVECs in the cell cycle. The results showed that the EEFA-treated HUVEC cells were arrested during the G2/M phase [Figure 6]. The previous studies indicated that Livistona chinensis seed extract (EELC) repressed the migration and proliferation of HUVECs, having an anti-angiogenesis effect.[30] Furthermore, the studies have shown that endothelial migration and proliferation were a main phase in angiogenesis.[31-33] Our study showed that the use of EEFA in hypoxic conditions inhibited the cell proliferation and migration in HUVEC cells. Therefore, the obtained results suggested that the EEFA suppressed the angiogenesis.
The study of angiogenesis-related gene expression revealed that EEFA significantly decreased the mRNA level of VEGF in HUVECs [Figure 7]. These findings were consistent with the results obtained from the previous studies. Cao et al. (2017) demonstrated that EELC has an anti-angiogenesis effect through reducing the expression of VEGF-A protein and mRNA.[30] Moreover, earlier studies have exposed that VEGF was an important mediator of angiogenesis, inducing the tumour formation, metastases and invasion.[34] In fact, our expression analysis results also supported from the role of EEFA in suppression of angiogenesis and tumour invasion.
Although EEFA did not have any effect on the expression of the VEGFR-2 gene, EEFA significantly increased the regulation of the VEGFR-1 gene expression in HUVECs [Figure 7]. These findings supported from the previous studies. Yang et al. (2005) suggested that the anti-angiogenic actions of Silibinin (SM/SB) are consistent with the upregulation of the VEGFR-1 (Flt-1) gene expression while not altering the expression of the KDR gene.[11] SFlt-1 protein was known as a potent natural antagonist of VEGFKDR signalling.[12] Consequently, due to the negative role of VEGFR-1 in angiogenesis, the use of EEFA with enhanced expression of VEGFR-1 may have an inhibitory effect on angiogenesis.
Furthermore, HIF-1α decreased insignificantly in HUVEC through the EEFA treatment in hypoxia [Figure 7] The previous studies have revealed that HIF-1a is an important regulatory protein to adapt tissues to hypoxia. HIF-1a was also effective in the promoting angiogenesis by activating the VEGF gene.[35,36] However, the inhibitory effects of EEFA on the expression of the VEGF gene may be linked to HIF-1a protein aggregation or HIF-1a protein synthesis and inhibiting its degradation by the ubiquitin-proteasome pathway.[37] For example, the suppressor effect of grape seed extract on HIF-1α expression was mostly related to the suppression of HIF-1α protein synthesis, but not to mRNA expression of HIF-1α.[38] Based on the previous studies, treatment with EEFA, as an anti-angiogenesis element, may play a role on the level of HIF-1a gene expression through the suppression of post-transcription processes.
Several molecular researches indicated that the anti-angiogenesis treatments have a significant effect on the progression of cancer.[29] In this research, we identified that that EEFA could possibility inhibit tumour angiogenesis by blocking the expression of angiogenesis factors in hypoxia condition. Based on the obtained results, EEFA had an anti-angiogenesis effect on HUVECs through decreasing migration and proliferation ability. Furthermore, EEFA could influence on the angiogenesis through decreasing the expression of VEGF, Akt and HIF-1a as well as increasing VEGFR-1 expression. Moreover, the bioinformatics analysis predicted that the selected genes and their interaction genes participated in VEGF, HIF-1a, ErbB and PI3K/Akt pathways. Among these pathways, VEGF,[39] HIF-1a,[40] ErbB[41] and PI3K/Akt[42] signalling pathways were found to be associated with the angiogenesis. These findings may provide the insight into the mechanisms and pathways underlying the anti-angiogenesis action of EEFA.
CONCLUSION
This is the first study to demonstrate the effect of EEFA on the expression level of VEGF/VEGFR in hypoxic-HUVECs. The obtained results provided the first proof that EEFA can effectively suppress hypoxia-induced angiogenic events including proliferation and migration through downregulation of VEGF and upregulation of VEGFR-1. Furthermore, our bioinformatics results showed that the studied gene and signalling pathways may play a significant role in the regulation of angiogenesis and cancer. These results could be a step toward improving the molecularly targeted therapies against angiogenesis in cancer by EEFA.
Declarations
Availability of data and materials
All data generated or analysed during this study and its supplementary information files are included in this published article.
Acknowledgments
The authors would like to thank the staff at the Central Research Lab of Birjand University of Medical Sciences for their help and cooperation.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
The medical research council of Birjand University of Medical Sciences.
Conflicts of interest
There are no conflicts of interest.
References
- Angiogenesis: An Integrative Approach from Science to Medicine Berlin, Germany: Springer Science and Business Media; 2008.
- [Google Scholar]
- Recent advances to accelerate re-endothelialization for vascular stents. J Tissue Eng. 2017;8:2041731417731546.
- [CrossRef] [PubMed] [Google Scholar]
- Cyanidin-3-O-glucoside modulates intracellular redox status and prevents HIF-1 stabilization in endothelial cells in vitro exposed to chronic hypoxia. Toxicol Lett. 2014;226:206-13.
- [CrossRef] [PubMed] [Google Scholar]
- A novel bHLH-PAS factor with close sequence similarity to hypoxia-inducible factor 1a regulates the VEGF expression and is potentially involved in lung and vascular development. Proc Natl Acad Sci USA. 1997;94:4273-8.
- [CrossRef] [PubMed] [Google Scholar]
- Hypoxiainducible nuclear factors bind to an enhancer element located 3'to the human erythropoietin gene. Proc Natl Acad Sci USA. 1991;88:5680-4.
- [CrossRef] [PubMed] [Google Scholar]
- Upregulation of vascular endothelial growth factor by cobalt chloride-simulated hypoxia is mediated by persistent induction of cyclooxygenase-2 in a metastatic human prostate cancer cell line. Clin Exp Metastasis. 1999;17:687-94.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular endothelial growth factor receptor-3 In: Vascular Growth Factors and Angiogenesis. Berlin, Germany: Springer; 1999. p. :85-96.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular endothelial growth factor receptor-3. Curr Top Microbiol Immunol. 1999;237:85-96.
- [CrossRef] [PubMed] [Google Scholar]
- Signal transduction by VEGF receptors in regulation of angiogenesis and lymphangiogenesis. Exp Cell Res. 2006;312:549-60.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular-specific growth factors and blood vessel formation. Nature. 2000;407:242-8.
- [CrossRef] [PubMed] [Google Scholar]
- Silibinin inhibits angiogenesis via Flt-1, but not KDR, receptor up-regulation1. J Surg Res. 2005;128:140-6.
- [CrossRef] [PubMed] [Google Scholar]
- Identification of a natural soluble form of the vascular endothelial growth factor receptor, FLT-1, and its heterodimerization with KDR. Biochem Biophys Res Commun. 1996;226:324-8.
- [CrossRef] [PubMed] [Google Scholar]
- Tumor angiogenesis-a potential target in cancer chemoprevention. Food Chem Toxicol. 2008;46:1334-45.
- [CrossRef] [PubMed] [Google Scholar]
- Angiogenesis: From plants to blood vessels. Trends Pharmacol Sci. 2006;27:297-309.
- [CrossRef] [PubMed] [Google Scholar]
- Galbanic acid isolated from Ferula assa-foetida exerts in vivo anti-tumor activity in association with anti-angiogenesis and anti-proliferation. Pharm Res. 2011;28:597-609.
- [CrossRef] [PubMed] [Google Scholar]
- Herbal compound farnesiferol C exerts antiangiogenic and antitumor activity and targets multiple aspects of VEGFR1 (Flt1) or VEGFR2 (Flk1) signaling cascades. Mol Cancer Ther. 2010;9:389-99.
- [CrossRef] [PubMed] [Google Scholar]
- Traditional uses, phytochemistry and pharmacology of asafoetida (Ferula assa-foetida oleogum-resin)-a review. J Ethnopharmacol. 2011;134:1-10.
- [CrossRef] [PubMed] [Google Scholar]
- Antidiabetic and antihyperlipidemic effects of ethanolic Ferula assa-foetida oleo-gum-resin extract in streptozotocin-induced diabetic wistar rats. Biomed Pharmacother. 2019;110:197-202.
- [CrossRef] [PubMed] [Google Scholar]
- Synergistic activity for natural and synthetic inhibitors of angiogenesis induced by murine sarcoma L-1 and human kidney cancer cells. Adv Exp Med Biol. 2017;1020:91-104.
- [CrossRef] [PubMed] [Google Scholar]
- Ferulic acid prevents angiogenesis through cyclooxygenase-2 and vascular endothelial growth factor in the chick embryo chorioallantoic membrane model. Turk J Pharm Sci. 2020;17:424-31.
- [CrossRef] [PubMed] [Google Scholar]
- An overview of colorimetric assay methods used to assess survival or proliferation of mammalian cells In: Vega-Avila E, Pugsley MK, eds. Proceedings of the Western Pharmacology Society. 2011.
- [Google Scholar]
- In vitro scratch assay: A convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc. 2007;2:329-33.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of different chitosan derivatives on in vitro scratch wound assay: A comparative study. Int J Biol Macromol. 2015;76:236-41.
- [CrossRef] [PubMed] [Google Scholar]
- Tumor microenvironment conditions alter Akt and Na(+)/H(+) exchanger NHE1 expression in endothelial cells more than hypoxia alone: Implications for endothelial cell function in cancer. BMC Cancer. 2017;17:542.
- [CrossRef] [PubMed] [Google Scholar]
- Enrichr: interactive and collaborative HTML5 gene1 list enrichment analysis tool. BMC Bioinformatics. 2013;14:128.
- [CrossRef] [PubMed] [Google Scholar]
- Secalonic Acid-D represses HIF1alpha/VEGF-mediated angiogenesis by regulating the Akt/mTOR/p70S6K signaling cascade. Cancer Res. 2015;75:2886-96.
- [CrossRef] [PubMed] [Google Scholar]
- mTOR pathway and mTOR inhibitors as agents for cancer therapy. Curr Cancer Drug Targets. 2008;8:647-65.
- [CrossRef] [PubMed] [Google Scholar]
- Natural products against cancer angiogenesis. Tumour Biol. 2016;37:14513-36.
- [CrossRef] [PubMed] [Google Scholar]
- Antiangiogenic effect of Livistona chinensis seed extract in vitro and in vivo. Oncol Lett. 2017;14:7565-70.
- [CrossRef] [Google Scholar]
- Expression profile of apoptotic and proliferative proteins in hypoxic HUVEC treated with statins. Int J Oncol. 2015;46:677-84.
- [CrossRef] [PubMed] [Google Scholar]
- The role of hypoxia-induced factors in tumor progression. Oncologist. 2004;9(Suppl 5):10-7.
- [CrossRef] [PubMed] [Google Scholar]
- Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol Cell Biol. 1996;16:4604-13.
- [CrossRef] [PubMed] [Google Scholar]
- Enhancement of angiogenesis, tumor growth, and metastasis by transfection of vascular endothelial growth factor into LoVo human colon cancer cell line. Clin Cancer Res. 2000;6:622-30.
- [Google Scholar]
- The molecular basis of the hypoxia response pathway: Tumour hypoxia as a therapy target. Cancer Metastasis Rev. 1998;17:187-94.
- [CrossRef] [PubMed] [Google Scholar]
- Tumour hypoxia, chemotherapeutic resistance and hypoxia-related therapies. Cancer Treat Rev. 2003;29:297-307.
- [CrossRef] [Google Scholar]
- Targeting of HIF-alpha to the von HippelLindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science. 2001;292:468-72.
- [CrossRef] [PubMed] [Google Scholar]
- Grape seed extract inhibits VEGF expression via reducing HIF-1alpha protein expression. Carcinogenesis. 2009;30:636-44.
- [CrossRef] [PubMed] [Google Scholar]
- VEGF enhances angiogenesis and promotes blood-brain barrier leakage in the ischemic brain. J Clin Invest. 2000;106:829-38.
- [CrossRef] [PubMed] [Google Scholar]
- Inhibition of hypoxia-induced angiogenesis by trichostatin A via suppression of HIF-1a activity in human osteosarcoma. J Exp Clin Cancer Res. 2006;25:593-9.
- [Google Scholar]
- Neuregulin activation of ErbB receptors in vascular endothelium leads to angiogenesis. Am J Physiol. 1999;277:H2205-11.
- [CrossRef] [PubMed] [Google Scholar]
- PI3K/AKT/mTOR pathway in angiogenesis. Front Mol Neurosci. 2011;4:51.
- [CrossRef] [PubMed] [Google Scholar]






