Translate this page into:
Evaluation of anti-psoriatic activity of selected phytochemicals on UV-induced psoriasis in mouse tail model
*Corresponding author: Jada Naga Lakshmi, Department of Pharmacology, Chalapathi Institute of Pharmaceutical Sciences, Chalapathi, Nagar, Lam, Guntur - 522 034, Andhra Pradesh, India. 1985nagalakshmi@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Lakshmi JN, Babu AN, Nadendla RR. Evaluation of anti-psoriatic activity of selected phytochemicals on UV-induced psoriasis in mouse tail model. Indian J Physiol Pharmacol 2020:64(2):123-8.
Abstract
Objectives:
To evaluate anti-psoriatic activity of Phytochemicals on UV-Induced psoriasis in mouse tail model.
Materials and Methods:
Anti-psoriatic activity of selected phytochemicals on UV-Induced psoriasis in mouse tail model. The animals were dividing into 05 groups and each group contain 5 animals. Disease control group did not receive any treatment only exposure to UV-light, vehicle control treated with simple ointment, standard group treated with salicylic acid (1%w/w) ointment, remaining group are treated 1% and 2% selective phytochemical at two concentrations of ointment to topically on the tail skin. And the data were analysed using one way ANOVA followed by two-way ANOVA (Dunnett’s multiple comparisons test).
Results:
There was significant decrease in epidermal thickness (P < 0.05) as compared with control group. In 2% phytoconstituents has shown a significant reduction in the total epidermal thickness 8.4****±0.748, 7.6**±0.6781 and 8*±0.8366 in geraniol, glycyrrhizic acid and ellagic acid treated group, when compare to the disease induced animal, there was no lesion of Munro’s microabscess, capillary loop dilation along with elongation of rete ridges in the section of skin of rats. Psoriasis Severity Index was reduced in test treated groups as compared with that of disease control group. It was slowly reduced to 2nd week, totally (55-70%) reduction in PSI is observed at the time of third week of treatment period.
Conclusion:
The result of the study showed that the 2% of geraniol, ellagic acid, glycyrrhizicacid and hesperidin, exhibited significant activity on UV-induced psoriasis in rodents. The study implies that selected phytoconstituents are a promising research for further investigations to prove its anti-psoriatic activity.
Keywords
Psoriasis
UV-Ray
Hesperidin
Ellagic acid and geraniol
INTRODUCTION
Psoriasis is a common inflammatory condition of human skin characterised by focal to coalescing raised cutaneous plaques with consistent scaling and variable erythema. Typical histologic features of psoriasis include epidermal hyperplasia (acanthosis) with elongated rete ridges, a less discrete epidermal granular layer (hypogranulosis), parakeratosis and leucocytic infiltration of the dermis and epidermis.[1] It is a chronic inflammatory disease of the skin characterised by epidermal hyperplasia, dermal angiogenesis, infiltration of activated T cells and increased cytokine levels.[2] An increase in mitotic activity in the stratum basale, abnormal keratinisation and elongation of the dermal papillae toward the skin surface result in a thicker-than-normal stratum corneum that desquamates to produce large, silvery scales (Griffiths and Voorhees, 1996; Barker,1991; Krueger and Callis, 2003)[3] Cyclooxygenase (COX)-2 inhibiting nonsteroidal anti-inflammatory drugs, corticosteroids and immunosuppressants such as FK-506 and cyclosporine A for Th1 cells have been used clinically for psoriasis. Repeated application of corticosteroids on the dorsal skin of rats causes dramatic skin atrophy. FK- 506 and cyclosporine A exhibit side effects, such as severe nephrotoxicity and neurotoxicity (Schafer et al., 1996; Sakuma et al., 2001; Friedman et al., 2002). Clinically drugs like NSAIDS are inhibiting the COX-2 enzyme and suppress the inflammation along with suppression of immunity by corticosteroids routinely used for treatment of psoriasis.[4] Conventional therapies such as corticosteroids, Vitamin D3 analogues and calcineurin inhibitors are currently used as topical therapies in mild psoriasis.
Severe psoriasis often necessitates treatment with phototherapy or systemic agents including methotrexate, cyclosporine and acitretin.[5] Biologic therapies that act on the upregulated cytokine pathways have also been developed and approved for psoriasis treatment.[6] However, most of these regimens have well-documented arrays of adverse effects that seem to be the main factor hampering patients’ adherence to long term. Repeated application of corticosteroids on the dorsal skin of rats causes dramatic skin atrophy. Psoriatic pathological features also include excessive proliferation and impaired differentiation of epidermal keratinocytes, likely mediated by a dysregulated immune system.[7] Some people may develop skin diseases that affect the skin, including cancer, herpes and cellulites. Some wild plants and their parts are frequently used to treat these diseases.
The use of plants is as old as the humankind. Natural treatment is cheap and claimed to be safe. Flavonoids, tannins, glycosides, monoterpenes and alkaloids have been considered as effective for the treatment of various disorders in humans.[8] All five selected compounds have been widely used as active ingredients in many pharmaceutical products due to their analgesic, anti- inflammatory, anti-pain and wound healing properties.[9-12] It has been demonstrated to control multiple signalling molecules and pathways that are involved in various biological processes, such as the cell cycle, cell survival and proliferation, apoptosis, autophagy and metabolism.[13-16] Multitarget therapeutic agents are more efficacious for the treatment of complex diseases, based on these parameters, we want to evaluate test compounds for the treatment of chronic skin disorders.
MATERIALS AND METHODS
Ethical approval
All the experiments were carried out on albino mice in Chalapathi Institute of Pharmaceutical Sciences. The mice were divided into 12 groups and each group (n = 5) animals were kept in separate cages for acclimatisation at a temperature 25±2°C and relative humidity of 52–55% with 12 h light/dark cycle 1 week before and before commencement of the experiment. They were kept on standard pellet diet ad libitum and drinking water throughout the study period. The protocol of experimental study was approved by the Institutional Animal Ethics Committee (IAEC) with IAEC approval (No: 12/IAEC/CLPT/2018-19; Dt: 05/01/2019).
Test compounds
Hesperidin, Glycyrrhizicacid, ellagic acid and geraniol were purchased from Sigma-Aldrich.
Qualitative analysis of phytochemicals
-
Flavonoids (Hesperidin):
Small quantity of hesperidin was dissolved in aqueous sodium hydroxide solution. Appearance of yellow colour indicates the presence of flavonoids.
Shinoda test: Small quantity of sample was dissolved in alcohol. To this few pieces of magnesium followed by concentrated hydrochloric acid was added dropwise and heated. Appearance of magenta colour indicates the presence of flavonoids.
-
Tannins (Ellagic acid):
Ferric chloride test: Small quantity of ellagic acid was taken in test tube and it was boiled with distilled water and then filtered. To the filtrate, few drops of 5% ferric chloride solution were added. Violet colour is observed, it indicated the presence of tannins.
Ellagic acid is taken into test tube to this 1% solution of gelatine containing 10% sodium chloride. White colour precipitated indicates the presence of tannins.
Glycosides (Glycyrrhizicacid): Foam test (saponin glycosides): A 10 mg of Glycyrrhizicacid powder was taken in 50 ml measuring cylinder, added water up to 25 ml of the measuring cylinder and marked it as point ‘A.’ Shake vigorously for 5 min. Allowed it to stand for 30 min. Now marked the final volume as point ‘B’ and measured the distance from ‘A to B.’ It shows the formation of foam with increasing volume. This indicates presence of ‘Saponin glycoside.’
Aromatic oils (Geraniol): Terpenoid test (Salkowski test): A 5 ml of Geraniol was mixed with 2 ml of chloroform and 3 ml concentrated H2SO4 was carefully added to form a layer. A reddish- brown colouration of the interface was observed; it showed positive results for the presence of terpenoids.
Experimental animals and research design
Mice of both sexes were taken and all groups of animals.
Those groups treated as Group-1 (control animal), Group-2 (standard drug treated animals), Group-3 (1% hesperidin), Group-4 (2% hesperidin), Group-5 (1% ellagic acid), Group-6 (2% ellagic acid), Group-7 (1% Glycyrrhizicacid), Group-8 (2% Glycyrrhizicacid), Group-9 (1% geraniol) and Group-10 (2% geraniol).
Salicylic acid ointment was applied to mouse tail of standard group.
Remaining group animals were treated with respective test compound. This process is continued for 7 days.
Later mice’s were sacrificed; tails were collected and send to histopathological studies.
Induction of psoriasis
All groups of animals were exposed to UV light for 14 days to inducing psoriasis on tail part.
Treatment application
After the development of psoriatic lesions on tail part of mice, regularly test compounds are applied on to the skin of animal tail for daily once with the help of cotton buds.
Evaluation of anti-psoriatic activity of test compounds
Epidermal thickness of mice tail was determined to observe the severity of psoriasis, physical observation of animal skin including erythema, itching and slivery patches on tail region. Psoriasis score index was calculated for each group to compare the efficacy of test compounds with that of standard. Psoriasis area and severity index (PASI) scores were determined by evaluating the degree of erythema, thickening and scaling on the affected dorsal skin surface and ear pinna. PASI for each was measured on a 4-point scale (0 = none; 1 = slight; 2 = moderate; 3 = marked and 4 = very marked). The severity of skin inflammation was measured by the combined scores (erythema plus scaling plus thickening) giving a range of scores of 0–12. Histological parameters are also observed by collecting the tail skin of mice.
Data analysis
The data generated were presented in frequencies, percentages and ‘Mean ± standard error mean’ and analysed with Chi-square, one-way analysis of variance (ANOVA).
RESULTS
Acute dermal toxicity
Healthy young adult animals of commonly used laboratory strains were employed. Each animal was 8–12 weeks old which was selected and all animals’ weights were within the range. The animals are acclimatised to the laboratory conditions for 5 days before the start of the study. Animals are randomly selected to use in the study and marked to provide individual identification. Approximately 24 h before the study, fur was being removed from the dorsal area of the trunk of the test animals by shaving. The test substance was applied uniformly over an area which is approximately 10% of the total body surface area. Test substances were held in contact with the skin with a help of porous gauze dressing and non- irritating tape throughout a 24 h exposure period. Animals were observed for signs of toxicity from 30 min to periodically during the first 24 h, with special attention given during the first 4 h. All animals were free from signs of toxicity.
Anti-psoriatic activity
UV-induced model for psoriasis
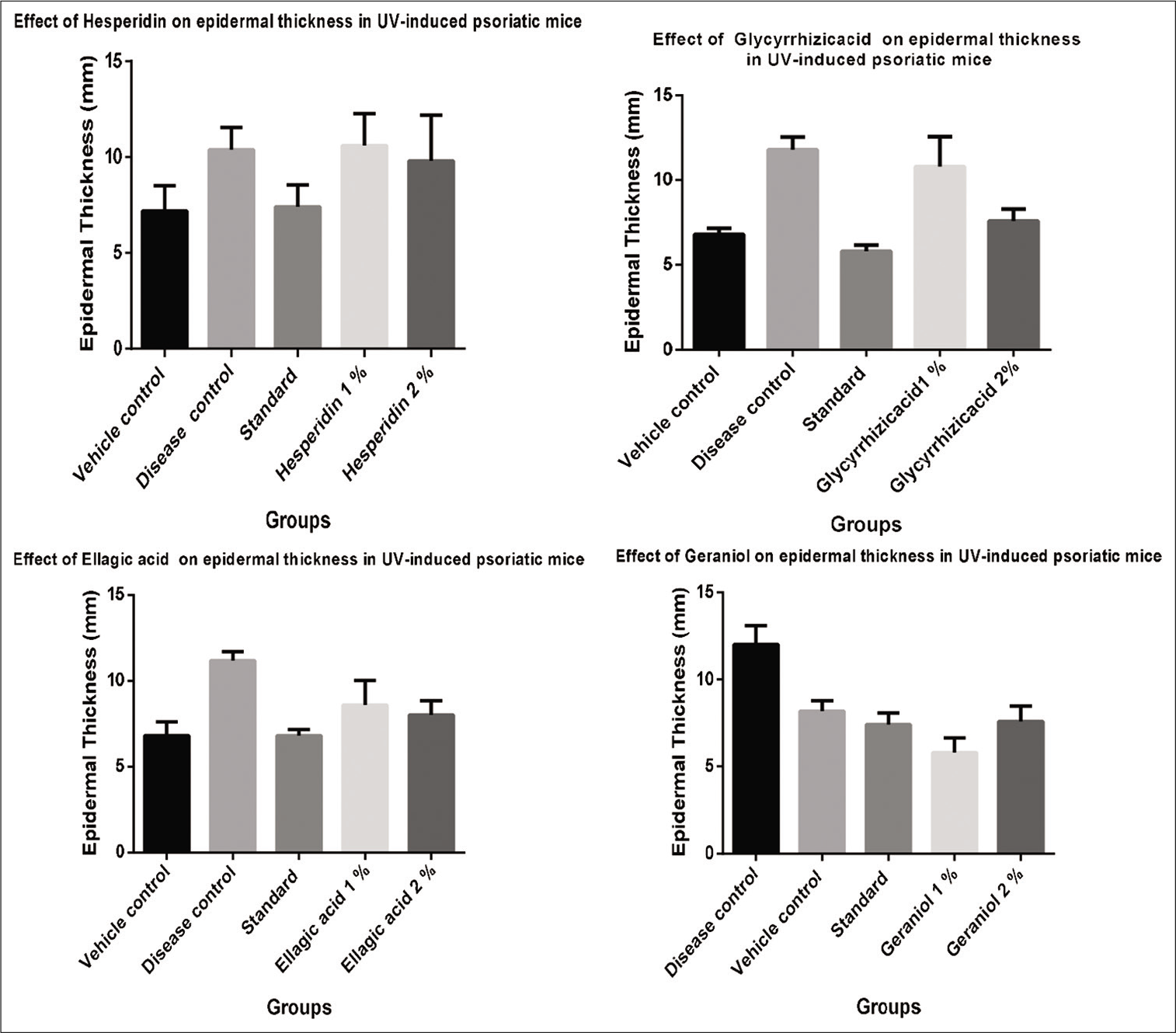
The mean thicknesses of the epidermis, in control- and drug-treated animals, were tabulated [Table 1]. There was significant decrease in epidermal thickness (P < 0.05) [Figures 1 and 2] as compared with disease control group. The section of disease control group showed regular elongation of rete ridges, capillary loop dilation with minimal grade lesion of diagnostic Munro’s microabscess and marked increase in relative epidermal thickness as compared with other groups. In 1% of (geraniol, Glycyrrhizicacid and ellagic acid) treated group, there was a minimal grade lesion of elongation of rete ridges along with capillary loop dilation in the section and absence of Munro’s microabscess. In 2% (Geraniol, Glycyrrhizicacid, ellagic acid and hesperidin) treated group, there was no lesion of Munro’s microabscess, capillary loop dilation along with elongation of rete ridges in the section of skin of rats [Figure 1]. In the standard group, there was absence of Munro’s microabscess, capillary loop dilation along with elongation of rete ridges in the section showing significant therapeutic effects when compared with test- treated groups.
| Treatment group | Hesperidin | Ellagic acid | Glycyrrhizicacid | Geraniol |
|---|---|---|---|---|
| Vehicle control (mm) | 7.2±1.3038 | 6.8±0.7999 | 6.8±0.3741 | 7.2±0.583 |
| Disease control (mm) | 10.4±1.1401 | 11.2±0.4898 | 11.8±0.7348 | 10.4±0.678 |
| Standard (mm) | 7.4**±1.1401 | 6.8**±0.3741 | 5.8*±0.3741 | 7.4**±0.509 |
| Test 1% (mm) | 9.8±2.3874 | 8.6±1.4352 | 10.8±1.7719 | 9.6 ***±0.927 |
| Test 2% (mm) | 10.4±1.8165 | 8*±0.8366 | 7.6**±0.6781 | 8.4****±0.748 |
The values are expressed in Mean±SEM, ***mean P<0.001, **P<0.05 results are significant compare to the disease control group. Analysis done by two-way ANOVA (Dunnett’s multiple comparisons test)

- Effect of phytochemicals on the pathological findings in the UV induced Psoriasis in mouse tail model. (a) Disease control, (b) Standard treated, (c) Vehicle control, (d) Disease control, (e)1% Hesperidin, (f) 2% Hesperidin, (g) 1% Ellagic acid, (h) 2% Ellagic acid, (i) 1% Glycyrrhizicacid, (j) 2% Glycyrrhizicacid, (k) 1% Geraniol, (l) 2% Geraniol.
- Effect of phytochemicals on epidermal thickness of mouse tail in UV-induced psoriatic animal.
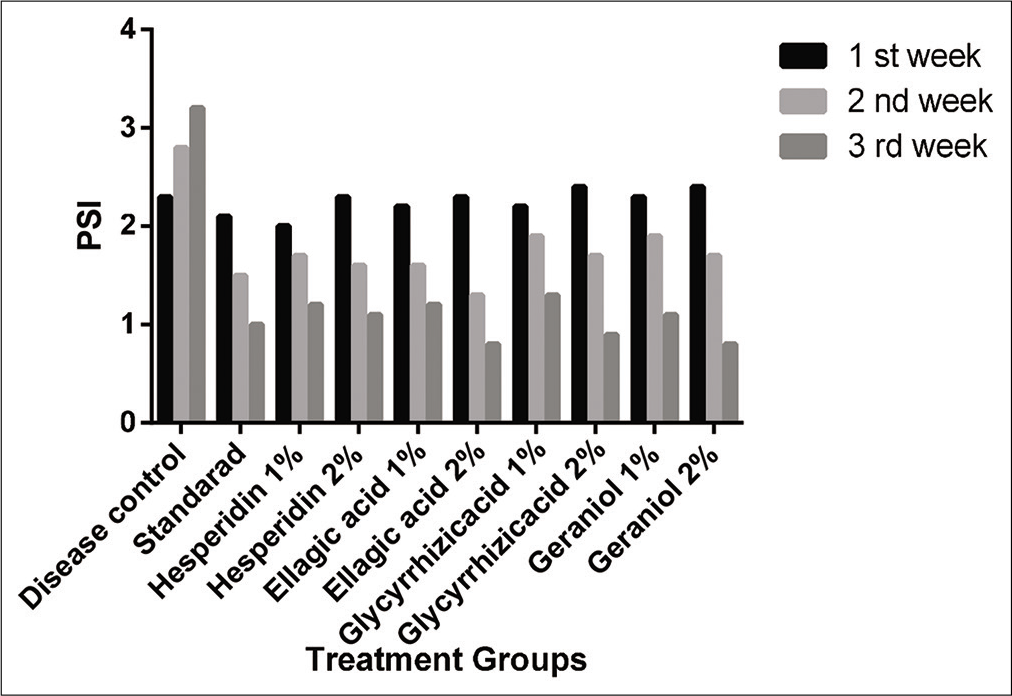
Psoriasis Severity Index was gave to all individual group animals based on their signs and presence of erythema, itching and silvery scales on the skin. Disease control group gain score of 2.0 at the 1st week, it was increased to 2.8 at the end of the experiment (3rd week), standard group gain 2.1, at the 1st week, after that application of drug on the topically to the skin, reduction in signs and symptoms of psoriasis slowly and 54% reduction is seen at 3rd week that 1.0 [Table 2 and Figure 3]. Test treated groups all were initially gained the score 2.4 to 2.8 at the 1st week, it was slowly reduced to the 2nd week, totally (55–70%) [Figure 3] reduction in PSI is observed at the time of the 3rd week of treatment period. It reveals that bio flavonoids, tannins and lipid-derived terpenoids have more effective for the treatment of chronic disorders of human skin without complications.
| Group | 1stweek | 2ndweek | 3rdweek |
|---|---|---|---|
| Disease control | 2.0 | 2.5 | 2.8 |
| Standard | 2.1 | 1.5*** | 1.0*** |
| Hesperidin 1% | 2.0 | 1.7*** | 1.2*** |
| Hesperidin 2% | 2.3 | 1.6*** | 1.1*** |
| Ellagic acid 1% | 2.4 | 1.6*** | 1.2*** |
| Ellagic acid 2% | 2.3 | 1.3*** | 0.8*** |
| Glycyrrhizicacid 1% | 2.2 | 1.9*** | 1.3*** |
| Glycyrrhizicacid 2% | 2.4 | 1.7*** | 0.9*** |
| Geraniol 1% | 2.6 | 1.9*** | 1.1*** |
| Geraniol 2% | 2.8 | 1.7*** | 0.8*** |
Note the results are obtain from one-way ANOVA, *** mean P<0.0001
- Effect of phytochemicals on Psoriasis Severity Index.
DISCUSSION
Skin is the largest exposed organ of body and easy target for allergic and immunologic reactions. Skin ailments, namely dermatitis, urticaria, angio-oedema and psoriasis, are immune-mediated chronic and inflammatory disorder. [11] Psoriasis severely affects patients with the quality of life and the treatment being expensive.[12] Medicinal plants are considered safe, as for the human health and are widely employed by the traditional healers for the treatment of various diseases including psoriasis. Medicinal plants are known to be a rich citadel of variety of chemical compounds and have attracted researcher’s attention to find new treatment for psoriasis.[17] Screening of anti-psoriatic activity of various phytoconstituents was carried out using UV-induced psoriasis in mouse tail. The irradiated mice skin treated with selected phytoconstituents at 2% has shown a significant reduction in the total epidermal thickness 8.4****±0.748, 7.6**±0.6781 and 8*±0.8366 in geraniol, Figure 2 Glycyrrhizicacid and ellagic acid treated group, when compare to the disease-induced animal. It indicating that phytochemicals have an influence to retard the hyperproliferation of the keratinocytes that occur when the skin is exposed to UV radiation. The significant retention of the stratum granulosum is probably due to its ability to enhance the keratinisation process, which is a protective strategy adopted by the skin when exposed to penetrating radiation. Further, phytoconstituents produced useful changes in the epidermis of the irradiated skin, showing its potential use in psoriasis treatment.
A natural polyphenols, recognised as potent antioxidants, are multifunctional molecules that can act as anti-inflammatory and antiproliferative agents through the modulation of multiple signalling pathways. This characteristic could be advantageous for the treatment of multicausal diseases caused by oxidative stress, including psoriasis. In an earlier work, Vijayalakshmi et al.[13] reported that the flavonoid quercetin showed significant reduction in epidermal thickness with respect to control in Perry’s mouse tail model. Jadranka Skuric et al.[14] reported that the flavonoids from propolis offer some protection against psoriatic complications through their roles as inhibitors of inflammation and as free radicals scavengers on animal model psoriasis, induced by the UV induced. The observed anti-psoriatic activity may be through the inhibition of keratinocyte proliferation, which may be attributed to the antioxidant properties of selected phytoconstituents, especially geranial, Glycyrrhizicacid and ellagic acid.
CONCLUSION
The result of the study showed that 2% of geranial, Glycyrrhizicacid and ellagic acid exhibited significant activity on UV-induced psoriasis in rodents. The study implies that selected phytoconstituents are a promising research for further investigations to prove its anti-psoriatic activity.
Acknowledgement
Authors acknowledge sincere thanks to the management and principal of Chalapathi Institute of Pharmaceutical Sciences, for their support to do the research work.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Reviewed paper: Preclinical models of psoriasis. Vet Pathol. 2008;45:563-75.
- [CrossRef] [PubMed] [Google Scholar]
- Psoriasis-epidemiology and clinical spectrum. Clin Exp Dermatol. 2001;26:314-20.
- [CrossRef] [PubMed] [Google Scholar]
- Topical glucocorticoids with improved risk-benefit ratio. Rationale of a new concept. Drug Saf. 1996;14:375-85.
- [CrossRef] [PubMed] [Google Scholar]
- Guidelines of care for the management of psoriasis and psoriatic arthritis. Section 4. Guidelines of care for the management and treatment of psoriasis with traditional systemic agents. J Am Acad Dermatol. 2009;61:451-85.
- [CrossRef] [PubMed] [Google Scholar]
- Guidelines of care for the management of psoriasis and psoriatic arthritis. Overview of psoriasis and guidelines of care for the treatment of psoriasis with biologics. J Am Acad Dermatol. 2008;58:826-50.
- [CrossRef] [PubMed] [Google Scholar]
- Adherence in the treatment of psoriasis: A systematic review. Dermatology. 2011;222:363-74.
- [CrossRef] [PubMed] [Google Scholar]
- The potential of plant phenolics in prevention and therapy of skin disorders. Int J Mol Sci. 2016;17:160.
- [CrossRef] [PubMed] [Google Scholar]
- Anti-psoriatic activity of Givotia rottleriformis in rats. Indian J Pharmacol. 2014;46:386-90.
- [CrossRef] [PubMed] [Google Scholar]
- Antipsoriatic activity of ethanolic extract of Woodfordia fruticosa (L.) Kurz flowers in a novel in vivo screening model. Indian J Pharmacol. 2016;48:531-6.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of the antipsoriatic activity of Aloe vera leaf extract using a mouse tail model of psoriasis. Phytother Res. 2012;26:617-9.
- [CrossRef] [PubMed] [Google Scholar]
- The impact of psoriasis on quality of life: Results of a 1998 national psoriasis foundation patient-membership survey. Arch Dermatol. 2001;137:280-4.
- [Google Scholar]
- Screening of flavonoid “quercetin” from the rhizome of Smilax china Linn. For anti-psoriatic activity. Asian Pac J Trop Biomed. 2012;2:269-75.
- [CrossRef] [Google Scholar]
- Effectivity of flavonoids on animal model psoriasis-thermographic evaluation. Period Biol. 2011;57:457-63.
- [Google Scholar]
- OECD Guidelines for Acute Toxicity of Chemicals Paris, France: Organization for Economic Cooperation and Development; 2001.
- [Google Scholar]
- Drug Discovery and Evaluation: Pharmacological Assays. (3rd ed). New York: Springer; 2008.
- [CrossRef] [Google Scholar]
- Plants and plant products with potential antipsoriatic activity-a review. Pharm Biol. 2012;50:1573-91.
- [CrossRef] [PubMed] [Google Scholar]






