Translate this page into:
Understanding mechanoreflex and metaboreflex interactions – a great challenge
*Corresponding author: Adrian Lis, Department of Physiology, Wroclaw Medical University, Wroclaw, Poland. adrian.lis@student.umed.wroc.pl
-
Received: ,
Accepted: ,
How to cite this article: Lis A, Paleczny B, Ponikowska B. Understanding mechanoreflex and metaboreflex interactions – a great challenge. Indian J Physiol Pharmacol 2021;65(1):1-11.
Abstract
The exercise pressor reflex (EPR) plays an essential role in cardiovascular and ventilatory responses to physical activity. Despite immense meaning and increasing validation of the EPR, there is no agreement on the character of interactions between its components and other reflexes in health and disease. The data addressing this issue remain incomplete and incoherent, partially due to various challenges in testing these pathways. The mounting evidence of EPR malfunction contribution to sympathetic over-activation in heart failure and other cardiovascular diseases shows clinical importance of comprehensive understanding of these mechanisms. In this review, we briefly summarize experiments focused on the issue of interactions between mechano-, metabo, chemo-, and baroreflex during exercise. We also address potential reasons of discrepancies in the results, identify gaps in this particular scientific area and propose notional pathways for future research. This article highlights the clinical importance of the EPR deterioration in heart failure pathophysiology and discusses potential therapies focused on restoring the reflex pathways. In addition, consideration is given to the latest sophisticated experiments in this area, underlining the need of changing the paradigm in EPR interactions studying – from teleological to mechanistic approach.
Keywords
Exercise pressor reflex
Mechanoreflex
Metaboreflex
Interactions
Heart failure
INTRODUCTION
One of the most essential parts of the reflex response to physical effort in human is the exercise pressor reflex (EPR) which consists of mechanoreflex and metaboreflex.[1-3] Deactivation of EPR in healthy human results in immediate deterioration of cardiovascular and ventilatory reaction to exercise, expressed by drop in heart rate (HR) (10%), blood pressure (8%), pulmonary ventilation (10%), hemoglobin saturation (3%), and also shortening of time to exhaustion (22%) as well as increase in peripheral muscle fatigue development (67%).[4] Despite their ultimate significance in exercise physiology, there is still no agreement on precise effects and interactions between mechano- and metaboreflex. Data from human studies are conflicting, showing that EPR testing is a remarkable challenge. Comprehensive understanding of metabo and mechanoreceptors stimulation effects is of great importance not only for the physiological basis but also for clinical purposes. Since EPR dysfunction has been associated with arterial hypertension and heart failure,[1,2,5-8] precise knowledge about its interactions is vital also for developing new targeted therapies in these conditions.[1,9] In this review, we present experiments addressing the issue of interactions between mechano- and metaboreceptors, as well as between EPR and other reflexes involved in cardiovascular control during exercise. We discuss potential reasons of discrepancy in the results of the studies and attempt to synthesize available data into the coherent message. We also depict clinical importance of EPR and identify possible pathways for future research in this intriguing field.
METHODOLOGY
Scopus, PubMed, and Google Scholar databases have been searched using following keywords (inclusion criteria): Metaboreceptors, mechanoreceptors, chemoreceptors, baroreceptors, mechanoreflex, metaboreflex, chemoreflex, baroreflex, EPR, effects, and interactions. More than 300 papers have been initially considered to include into the review. In the first step, manual screening of abstracts was performed. The exclusion criteria comprised: Misguided methodology (i.e., inappropriate statistical tests), irrelevant results and conclusions, language other than English and journal not included in the “InCites Journal Citation Reports” list. Subsequently, the full texts were comprehensively read and assessed in terms of methodology (subjects, methods, and statistical tests) and the results-conclusions relationship. Papers that did not meet the quality criterion in these fields were excluded at this stage. Eventually, after final selection 76 original research articles, 13 review articles, three letters to the editor, two editorials, two symposium reports, and one exchange of views were analyzed to prepare this review.
EFFECTS OF MECHANO- AND METABOREFLEX – LESSONS FROM THE PAST
EPR is often considered as one reflex, which is misleading. It is highly important to distinct two components of this mechanism, since they exert different effects. Faultiness of the generalization approach was clearly shown in case of carotid bodies. For decades peripheral chemoreflex was considered simple pathway, it was believed that hypoxia and hypercapnia stimulate chemoreceptors in carotid bodies, which results in hyperventilation, vasoconstriction and general sympathoexcitation. Thanks to titanic scientific work and thoughtful research of many investigators with Prof. Paton on the front line – it turned out glomus cells are divided into sub-populations responsive only to hypoxia or hypercapnia exclusively and that the effect of stimulation (hyperventilation, vasoconstriction, or sympathoexcitation) depends on the kind of particular stimulus.[10,11] Therefore, when studying the EPR interactions, it is vital to first distinguish effects of individual components (mechanoand metaboreflex). One can speculate, that there are even more than two separate pathways in EPR, that is, different cells responsive for various metabolites or for different kinds of mechanical stimulation, which will be discussed in subsequent paragraphs. Although there is general agreement that both metabo- and mechanoreflex lead to vagal withdrawal and sympathoexcitation, assignment of precise cardiovascular effects to a particular component is not consistent among the studies.[1-3,12] The first reason of the discrepancy is that researchers use different methods of stimulating mechanoreceptors. In the past, this was achieved by passive cycling,[5,13-15] electrical stimulation,[6,16] or passive stretching.[17-20] Furthermore, different kind of muscles were stimulated in previous studies – thigh,[5,13-17] calf,[19,20] arm,[6] and forearm[21] muscles. Immensity of the methods proposed to evoke mechanoreceptors response shows that testing the mechanoreflex is a challenging task. Almost all of available data confirm that the main result of mechanoreceptors activation is increase in mean arterial pressure.[6,13-16,18,22,23] However, there is lack of concordance between particular studies in case of impact of mechanoreflex on other parameters, such as HR and stroke volume (SV). Fortunately, there is more consistency about cardiovascular effects of metaboreflex – the typical effect of metaboreceptors stimulation is gain in blood pressure resulting from increase in total peripheral resistance, which is generally evoked by static exercise, followed by circulatory occlusion (CO).[24,25]
THE CLINICAL IMPORTANCE OF MECHANOAND METABOREFLEX INTERACTIONS
Although mechanoreflex and metaboreflex exert individual effects on cardiovascular system, the combined outcome of simultaneous stimulation of both types’ receptors is not always a simple sum of them.[15,19,21,26] This fact is essential in clinical setting. Exercise training is well known for its beneficial health effects, particularly in cardiovascular diseases, such as hypertension, coronary artery disease, or heart failure.[27,28] On the other hand, EPR is impaired in these conditions which probably raises cardiovascular risk, decreases exercise capacity, and contributes to progression of muscle fatigue and dyspnea.[2,7,8,29-34] The deterioration of EPR is characterized partially by hyperactivity of mechanoreflex[1,2,7-9,23,31,35-44] and partially by alteration in metaboreflex activity. The character of the latter in heart failure is controversial – some experiments show abnormally high response of metaboreflex,[7,8,23,31,37-41,45,46] whereas other studies report diminished activation.[1,2,9,42-44] There are many factors that contribute to this discrepancy among the experiments, such as heterogeneous groups of patients, different exercise protocols, difficulties in isolating the metaboreflex, and different indicators of metaboreflex activation – muscle sympathetic nerve activity (MSNA), minute ventilation, blood pressure, or vascular resistance. Apart from the ambiguities, the most important aspect is to comprehend how do the metabo- and mechanoreflex act together, since in real-life conditions there is always general stimulation of EPR. However, the proportion of mechanoreflex activation to metaboreflex activation is different for various kinds of physical activity (i.e., static, dynamic, and stretching).[1,24] Therefore, understanding of interactions between particular components of EPR and mechanisms of its impairment in particular disease is vital for clinical purposes: (i) to develop specific interventions for restoring normal response to physical activity and reducing the risk of exercising and (ii) to establish if given reflex pathway should be blocked or stimulated.
Several studies (both in animals and humans) proved that exercise training can revert deterioration of EPR in heart failure.[7,31,42,47-50] This reversion seems to have a tremendous significance to exercise capacity in patients. According to “muscle hypothesis” of heart failure and its neurohormonal component – contribution of EPR to exercise intolerance in heart failure patients is probably greater than contribution of hemodynamic alterations, such as decline in left ventricular ejection fraction and cardiac output.[31,32,51] This hypothesis explains startling observation that despite normalization of cardiovascular parameters in heart transplant recipients, exercise tolerance remains impaired.[52,53] Training programs applied in previous experiments included 6-week of forearm exercise,[7,50] 7-week treadmill training,[47] 4-week inspiratory muscle training,[48] 6-month exercise-based cardiac rehabilitation[49] resulted mainly in reduction in blood pressure, minute ventilation, vascular resistance, and MSNA in response to acute physical activity. Data from available studies do not allow to make conclusions on the mechanisms of these advantageous effects. Unfortunately, lack of mechanistic studies is a general issue with EPR testing.[54] Lately, it was suggested that recruitment of more muscle mass in exercise programs may be beneficial in restoring normal EPR functioning in patients with heart failure.[32] However, further studies are warranted to determine the optimal training regimen.
Very little is known about the potential pharmacological agents that could modulate EPR in pathological condition. Intra-arterial gadolinium injections were used to attenuate response from mechanoreceptors in cats, mice, and rats,[54-57] although their usefulness was never confirmed in human studies. Experiments using intra-arterial injection of another agent indomethacin – showed encouraging results. This inhibitor of prostaglandin synthesis exhibited potential of reducing mechanoreflex activity in both cats[58] and humans. [59] Unfortunately, there were no further studies encompassing patients with heart failure or even healthy humans but using different route of administration. Naturally, intra-arterial injections could not be considered as a long-term treatment method in a real-life clinical setting. Chaiyakul et al.[60] displayed that stimulation of serotonin receptors (5-HT1A) within rostral ventrolateral medulla attenuates EPR response to static exercise in rats. Considering that agonists of 5-HT1A receptors are widely used in psychiatry (buspirone, tandospirone, vilazodone, and vortioxetine) it would be very interesting to assess their effects on EPR in patients with concomitant heart failure or other cardiovascular comorbidities. Very interesting contributions have been done using analgesic balm with capsaicin – an active compound of chili peppers.[61-63] First, the balm was used in decerebrated cats on the skin surface over muscles and significantly decreased blood pressure reflex responses to electrically-evoked static contractions.[62,63] Then, the approach was translated on healthy humans and confirmed that topically applied capsaicin attenuated blood pressure and MSNA during metaboreflex activation.[61] Further studies in heart failure patients is highly warranted. Lately, Li and Garry opened new opportunities in EPR interactions testing in health and variety of diseases.[54] They introduced a novel murine mode, which allows for more comprehensive investigation of mechano- and metaboreflex, due to unfettered genetic modification possibilities and exploration of the molecular mechanisms of EPR. We do hope the murine model will contribute to discovering and development of new pharmacological agents targeted at EPR.
PROTOCOLS FOR TESTING INTERACTIONS
The available data on the interactions between mechanoand metaboreflex are very limited. Most of experiments use similar method: First exercise, then CO and at last muscle stretching during CO.[17,19-21] In recent paper from our laboratory, we proposed a different, novel method for testing mechanoreflex-metaboreflex interactions.[15] In this study, participants were subjected to bilateral passive cycling, during which bilateral leg CO was added. The alternative method of mechanoreceptors stimulation and different order of interventions were performed to mimic physiological model of dynamic exercise. To activate metaboreflex all of the available studies report inflation of tourniquets to 200[15,19,20] or 250 mmHg[17,21,64] which ensures that both arterial and venous circulation are occluded.
THE EFFECTS OF SIMULTANOEUS MECHANOAND METABORECEPTORS ACTIVATION
Animal research suggests metabolites sensitize mechanoreceptors.[65-68] Unfortunately, there is some inconsistency in results of particular human studies. While metaboreceptors activation were always achieved by CO, to obtain mechanoreflex response – thigh,[15,17,64] calf,[19,20] or forearm[21] muscles were subjected to static stretching,[17,19-21] dynamic stretching,[17] passive movement performed manually by a researcher[64] or passive cycling.[15] This diversity resulted in different observations. All analyzed studies are presented comprehensively in [Table 1]. One study reported hyperadditive interaction between mechano- and metaboreflex in case of muscle sympathetic nerve activity (MSNA) and mean arterial pressure.[21] One experiment revealed hyperadditive reaction in case of heart rate and additive when it came to mean arterial pressure. [15] In one study there was hyperadditivity in case of cardiac output, heart rate and stroke volume.[19] One experiment showed simple additivity in case of diastolic blood pressure[20] and two postulated marginal interactions between these reflexes[17,66] (experiment of McDaniel et al.[66] was conducted to assess the role of hyperemia during passive movement, although results can be also addressed to the metaboreflex-mechanoreflex interactions). One can say that the discrepancies come from different kind of methods used in particular studies. However, even when the same method was applied (static calf stretching with CO) the results were different. Fisher et al.[20] showed that both metaboreflex and mechanoreflex increase diastolic blood pressure, but there is no additional gain when both reflexes act together. They also reported that not metabo- nor mechanoreflex or simultaneous stimulation had any effect on HR. Inversely, White et al.[19] displayed that static calf stretching alone did not result in any alteration, but when performed during CO – evoked increase of HR and SV. This is partially consistent with results of study from our laboratory[15] in which not passive cycling nor CO affected HR, but passive cycling during CO resulted in increase of this parameter. This can be certainly interpreted as an example of hyperadditivity between these two reflexes.
| Authors Title | Subjects | Mechanoreflex stimulation method | Metaboreflex stimulation method | Mechanoreflex stimulation effect | Metaboreflex stimulation effect | Simultaneous stimulation effect |
|---|---|---|---|---|---|---|
| Venturelli et al. Central and peripheral responses (…) | 10 healthy volunteers (all males) Age: 25±1 years Height: 181±2 cm Weight: 77±2 kg | Unilateral passive knee stretching (static and dynamic) | Unilateral CO (250 mmHg) | Static stretch: (+) HR, CO (slight) (−) MAP (slight) Dynamic stretch: (+) HR, CO (−) MAP (slight) | n/a (no metaboreflex stimulation alone) | With static stretch: (+) MAP (slight) With dynamic stretch: no changes |
| White et al. The pulmonary vascular response (…) | 9 healthy volunteers (4 males) Age: 27±4 years Height: n/a Weight: n/a | Unilateral passive static calf stretching | Unilateral post-exercise CO (250 mmHg) | No changes | (+) SPAP, MAP, SBP, DBP, (maintained elevation) | (+) SPAP, MAP, SBP, DBP (maintained elevation, similar to metaboreflex stimulation alone), CO, HR, SV |
| Fisher et al. Cardiovascular responses to human calf (…) | 8 volunteers (7 males) Age: 22±1 years Height: 180±2 cm Weight: 76±3 kg | Unilateral passive static calf stretching | Unilateral post-exercise CO (200 mmHg) preceded by 3 different initial workloads | (+) DBP | (+) DBP (maintained elevation) | (+) DBP (irrespectible of initial workload) |
| Cui et al. Effects of muscle metabolites on responses (…) | 12 healthy volunteers (8 males) Age: 26±1 years Height: 178±3 cm Weight: 76±2 kg | Unilateral passive wrist stretching (static and dynamic) | Unilateral post-exercise CO (250 mmHg) | Static stretch: no changes Dynamic stretch: (+) MSNA (transient) | (+) MSNA, MAP, (maintained elevation) | With static stretch: (+) MSNA, MAP (greater than during metaboreflex stimulation alone) With dynamic stretch: Not available |
| McDaniel et al. Central and peripheral contributors (…) | 9 healthy volunteers (all males) Age: 33±9 years Height: 180±8 cm Weight: 85±17 kg | Unilateral passive movement of the knee performed manually by a researcher | Unilateral CO (250 mmHg) | (+) SV, HR, CO (transient) (−) MAP (transient) | (+) MAP (transient) | (+) MAP; SV, HR, CO (transient, slightly smaller than during mechanoreflex stimulation alone) |
| Lis et al. Passive bilateral leg cycling (…) | 34 healthy volunteers (22 males) Age: 24±4 years Height: 178±11 cm Weight: 75±15 kg | Bilateral passive cycling | Bilateral CO (200 mmHg) | (+) MAP, SBP, DBP, SV, MV (−) TPR | (+) MAP, DBP, TPR (slight) | (+) MAP, SBP, DBP, MV, HR |
The table shows particular studies and data on their authors, subjects, methods, and effects of particular interventions. Age, height, and weight are shown as mean values ± standard deviations. Analyzed parameters comprise; MAP: Mean arterial pressure, SBP: Systolic blood pressure, DBP: Diastolic blood pressure, TPR: Total peripheral resistance, SV: Stroke volume, HR: Heart rate, MV: Minute ventilation and MSNA: Muscle sympathetic nerve activity, CO: Circulatory occlusion. Parameters in bold presented hyperadditive pattern of response. n/a: Not available
It should be underlined that the character of interaction is not always positive or neutral. It might be also negative, although it does not mean that it is not beneficial. It has been perfectly shown on pulmonary arteries.[19] While stimulation of metaboreceptors with CO evoked increase in pulmonary vascular resistance, adding mechanoreflex activation with calf stretching attenuated this reaction, thus facilitating blood flow through the pulmonary vascular bed during exercise.
ISSUES WITH TESTING EPR INTERACTIONS
The main issue that we identified is the choice of method for stimulating mechanoreceptors. The majority of experiments use muscle stretching to activate mechanoreflex. This approach, based on an assumption that stretch stimulates the same Group III afferents as does contraction, is unfortunately erroneus. Hayes, Kindig and Kaufman showed in their excellent animal study, that out of 30 Group III mechanosensitive muscle afferents, 18 responded to contraction, and 14 to stretch, although only seven responded to both stimuli.[69] Furthermore, conduction velocities of the afferents differed depending on the kind of stimulus.[69] The evidence of this diversity was provided also in humans by Gladwell et al., who studied cardiovascular reactions to passive stretching during alterations in vagal tone.[70] They proposed to distinct so called “tentonoreceptors,” which would be a special subgroup of mechanoreceptors mediating increase in HR but not blood pressure, particularly in response to stretching.[70,71] Static stretching does not seem to be an optimal technique of mechanoreceptors stimulation, also for another reason – in some experiments it did not evoke any reactions when performed alone, regardless of the type of the muscle.[19,21] The situation was slightly different when dynamic stretching or dynamic passive movement was performed – this resulted in only transient alterations in cardiovascular parameters and MSNA.[21,64] Apparently, mechanoreceptors are very heterogenous in their nature and reflex effect depends on the location of the receptor (thigh, calf, arm, etc.) and kind of stimulus (stretching, contracting). Researchers need to be very careful when choosing the method in their experiment and extrapolating the results on general mechanoreflex functioning. Surely, we identify different kinds of stretching of various muscles as a main reason of the discrepancy in results of available studies. In our opinion, all of the aforementioned facts should force researchers to seek one, unified protocol for testing mechanoreflex-metaboreflex interactions. That is why we proposed passive bilateral cycling with concomitant CO.[15]
The use of CO to activate the metaboreflex is not so controversial, although some authors indicate, that the sympathoexcitation after CO might result from unloading of cardiopulmonary receptors.[72,73] That is why it is essential to occlude both arterial and venous circulation. This approach does not reduce the relative venous return and therefore enables to link the observed cardiovascular effects to metaboreceptors stimulation.[74] There is, however, another issue with CO - most of the studies use post-exercise CO, which precludes quantitative assessment of interaction (the onset of stimulation of metaboreceptors appears right after the end of mechanoreceptors activation).
The other reasons for difficulties in the assessment of mechano- and metaboreflex interactions are listed below:
Some of the Type III and IV afferents may be polymodal – susceptible both to mechanical distortion and metabolite accumulation (data from animal studies)[65,67]
Difficulty in mimicking physiological conditions that apply only to one component of the EPR (only mechanical distortion or only metabolite accumulation)
Need for exclusion of central command
Interactions with other reflexes (baroreflex and chemoreflex)[16,26,75,76]
Lack of control group in some studies (should be mechanoreflex alone, metaboreflex alone, and mechanoreflex + metaboreflex)[17,64]
Lack of complete information about changes of each parameters (e.g., only diastolic blood pressure and HR showed in results).[20]
WHAT ARE THE MECHANOREFLEXMETABOREFLEX INTERACTIONS?
Most of authors agree that there is at least additive interaction between mechano- and metaboreflex in case of blood pressure – according to some investigators even hyperadditive [Table 1]. Available data suggest that it is similar with HR. These interactions seem reasonable, since they help to provide blood supply to muscles engaged in exercise. However, some methods might not allow to display these reactions. It is worth to notice that in majority of studies the number of participants varied between 8 and 12[17,19-21,64] and only in one it was 34.[15] This raises assumptions that some interactions could not be revealed due to the small group size effect. Nevertheless, based on the available studies, we can also identify negative interaction between these two reflexes in case of the pulmonary vascular resistance and positive in case of cardiac output.
INTERACTIONS OF EPR WITH OTHER REFLEXES
Mechano- and metaboreflex, as part of a greater mechanism that respond to physical activity, also do interact with other reflexes, namely, chemoreflex[26,75,77] and carotid baroreflex.[16,76] In their elegant study Silva et al.[26] exposed healthy subjects to inhalation of 12% O2 during isocapnia to stimulate the peripheral chemoreflex with and without concomitant passive knee movement (mechanoreceptors activation). They reported hyperadditive interactions between both reflexes in case of minute ventilation. Interestingly, it was achieved probably due to different pathways operating in both reflexes, since mechanoreceptors stimulation resulted in an increase of breathing frequency, whereas chemoreflex activation produced gain in tidal volume. Surely, this cooperation enables potentiation of ventilation during physical activity to provide more oxygen to working muscles. This suggests that the hyperadditive reaction can occur when particular parameter is modified by different reflexes through distinct pathways. Similarly, hyperadditive interaction between EPR and peripheral chemoreflex was reported recently in case of blood pressure and HR.[75] Authors used sophisticated method with intrathecal injection of fentanyl to block EPR afferent fibers in three different breathing protocols (ambient air, normocapnic hypoxia, and normoxic hypercapnia). They displayed also that peripheral chemoreflex and EPR act in a hypoadditive manner in case of responses in leg blood flow and leg vascular conductance. Unfortunately, the method they used did not allow to distinguish which part of EPR (mechanoreflex, metaboreflex, or both) was responsible for observed reactions. Another great contribution was made by Bruce and White,[77] who performed four different protocols of mechano- and metaboreflex activation during normoxic hypercapnia or ambient air breathing. They provided an evidence of hyperadditive interaction between central chemoreflex and isolate mechano- or metaboreflex in case of minute ventilation.[77] Likewise, the blood pressure response was hyperadditive when mechanoreflex and central chemoreflex acted together.[77] There were also other contributions focused on simultaneous activation of EPR and chemoreflex, although use of voluntary exercise without intrathecal fentanyl does not allow to exclude central command and therefore address results to EPR itself.[78-82]
Studies on interactions between EPR and carotid baroreflex also yielded interesting results. Hureau et al.[16] used lumbar intrathecal fentanyl to block EPR afferent fibers in humans during electrically evoked and voluntary knee extension. In their cleverly designed experiment they revealed that EPR does not influence carotid baroreflex sensitivity, but resets its operation point for both blood pressure and HR during exercise, thus allowing increase in both parameters when it is crucial. There is also one more enlightening fact that arises from the aforementioned study – fentanyl injection did not change resting mean arterial pressure and HR. This suggests that the input from mechano- and metaboreceptors is not tonic but appears only when exercise is performed. Therefore – in case of future therapeutic goals – simple blockade of the EPR will not be successful in hypertension at rest. Unfortunately, as authors decided to choose electrical stimulation to induce isometric muscle contraction, we cannot attribute these results particularly to mechano- or metaboreflex. This is mainly due to the fact, that electrostimulation evokes even greater metabolic perturbation than voluntary exercise[83-85] and therefore contribution of metaboreflex cannot be excluded. Nevertheless, in another study on animals it was shown that the mechanoreflex activation is responsible for resetting of the baroreflex.[86] A kind of complement to this story may be an excellent study of Kaur et al.[76] focused on interactions between metaboreflex and carotid baroreflex in canine. Authors used graded reductions in hindlimb blood flow during dynamic exercise (metaboreflex activation), followed by simultaneous bilateral carotid occlusion (baroreflex activation). The magnitude of an increase in blood pressure denoted simple additive interaction between these two reflexes, although the mechanisms for the response were different. Metaboreflex-induced increase in blood pressure occurred due to gain in cardiac output, hart rate, and ventricular contractility, whereas baroreflex evoked further rise through decrease in vascular conductance. Interestingly, this decrease occurred less in the ischemic hindlimb vasculature than in all remaining vascular beds, thus redirecting blood flow to the ischemic muscle. This unique preferential peripheral vasoconstriction protected active hindlimb muscle against the progression of ischemia. Unfortunately, this observation was never confirmed in human studies.
WHERE THE INTERACTIONS TAKE PLACE?
It is still not known on which level of nervous systems interactions of EPR take place. When it comes to interactions between peripheral chemoreflex and mechanoreflex, as Silva et al.[26] suggested, the interaction probably occurs in the central nervous system (the nucleus tractus solitarii or the retrotrapezoid nucleus), since there is no connection between these two pathways before it. For baroreflex-mechanoreflex interactions site, Gladwell et al.[70] proposed cardiac vagal neurons (parts of the nucleus ambiguus and the dorsal motor nucleus of the vagus). The issue is much more complex regarding internal interactions between metabo- and mechanoreflex – their receptors might even have the same afferent fibers.[34,87-89] For separate fibers – the first synapse is located in the spinal cord’s dorsal horn (Rexed’s laminae I, II, V, and X)[1,90,91] and this can be first potential location to interact. The primary site of EPR is probably the nucleus tractus solitarii,[1,91] which is the next probable interaction’s place. This is very likely, since in animal experiments it was found that the nucleus tractus solitarii is a complex site of various convergence patterns for baroreflex, chemoreflex, and Hering-Breuer reflex.[92,93] Other neurons that are engaged in EPR and could be presumptive interaction’s sites are nucleus ambiguous, rostral ventral medulla, lateral tegmental field, and caudal ventrolateral medulla.[1,94,95] Nevertheless, all of the possible mechanisms given above are only speculations, that still need to be tested in mechanistic studies. All interactions and their presumable neuroanatomical locations are depicted schematically on [Figure 1].
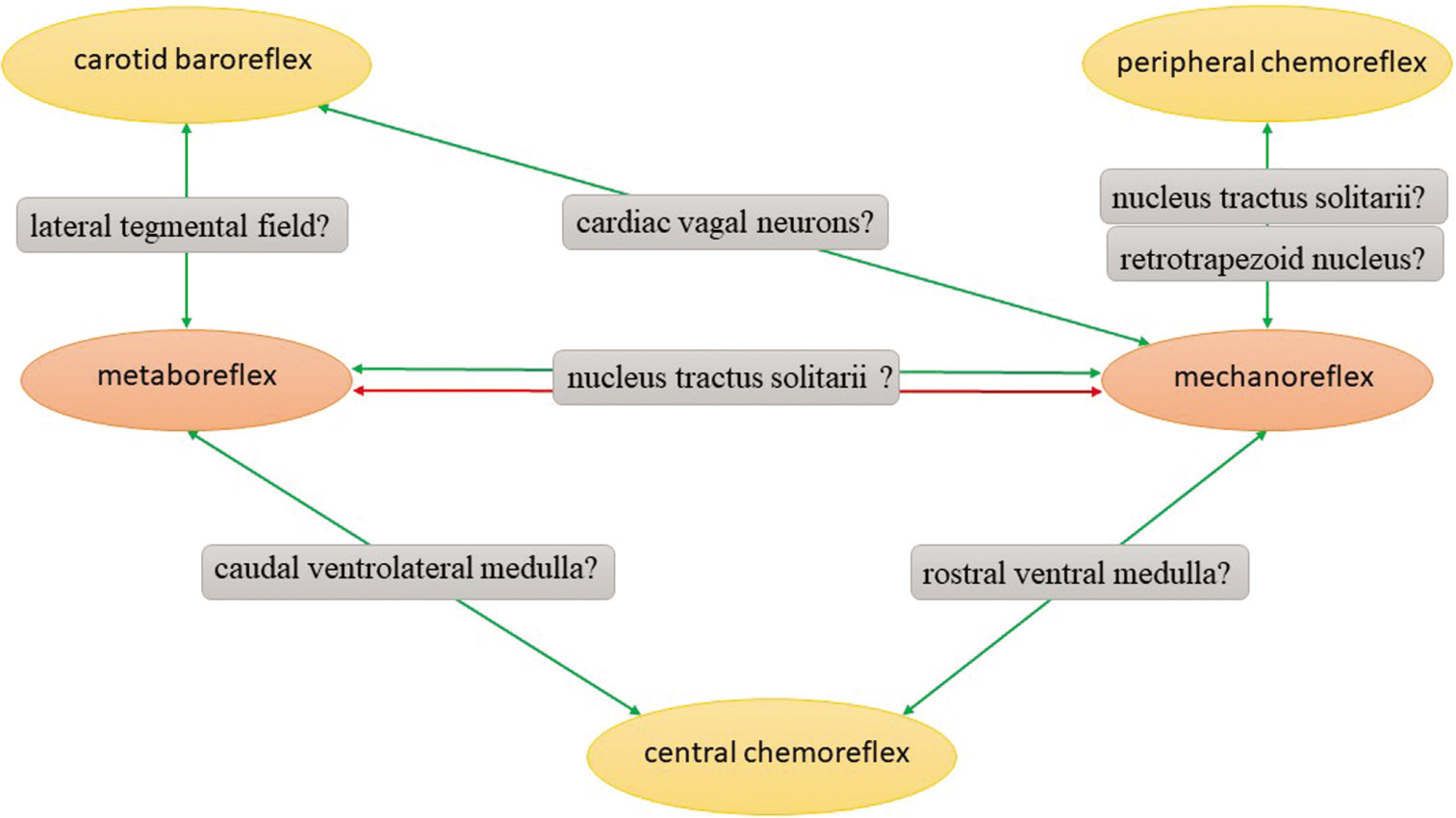
- The scheme shows interactions of exercise pressor reflex and their presumable neuroanatomical locations. Green arrows symbolize positive interactions, whereas red arrows reflect negative interactions.
PERSPECTIVES
Up to date, the issue of EPR interactions did not receive much attention from researchers. Regarding available data, we can state that the effects of mechanoreceptors activation and interactions between mechano- and metaboreceptors are complex and depend on the method of testing. That is presumably because of stimulating different types of mechanoreceptors with various methods, since mechanoreceptors seem to constitute a heterogenous group. Nevertheless, there is most likely hyperadditive interaction in case of blood pressure and HR, as well as hypoadditive interactions in case of pulmonary vascular resistance. In addition, hyperadditive interrelation in case of blood pressure, HR, and minute ventilation was identified between EPR and chemoreflex (both central and peripheral). EPR has been also proved to change baroreflex setpoint during physical activity. Apart from simply additive interaction between metaboreflex and baroreflex, the latter probably helps redirecting blood flow to the ischemic muscle during simultaneous stimulation. Given the gaps in available research, a comprehensive experiment comparing directly different methods of testing mechano- and metaboreflex simultaneously is warranted. The number of subjects should be certainly higher than usual 10, powered to accurately display particular interactions.
Surely, the big gap in the discussed area is ventilatory effects of EPR interactions. The majority of data center on cardiovascular reactions, whereas muscle afferent feedback is responsible for approximately half of the ventilatory response during moderate physical activity![34,96] This issue has also essential clinical meaning – the dyspnea resulted from abnormally augmented EPR in pathological condition awaited an exceptional term “breathless with over-excitement.”[34] We believe that progress in research focused on ventilatory effects of interactions between mechano-, metabo-, and chemoreflex will open the way to search for novel therapies, targeted on the components of EPR. We also acknowledge that there is a need for changing a paradigm in pressor reflex testing – from teleological to mechanistic approach [Figure 2]. Fortunately, last year brought studies focused on molecular alterations of EPR in heart failure patients[97] and creating animal model of mechano- and metaboreflex allowing for genetic modifications and more profound exploration of reflex deterioration mechanisms.[54] We do hope that more abundant mechanistic knowledge will allow for discover and develop pharmacological agents, possible to use in patients with defective EPR. However, even having effective pharmacotherapy, we cannot forget about the exercise training. Despite the solid evidence of salutary effects of training programs in patients with impaired EPR, this topic still needs much more exploration, focused on the mechanisms, and optimal regimens of exercise.
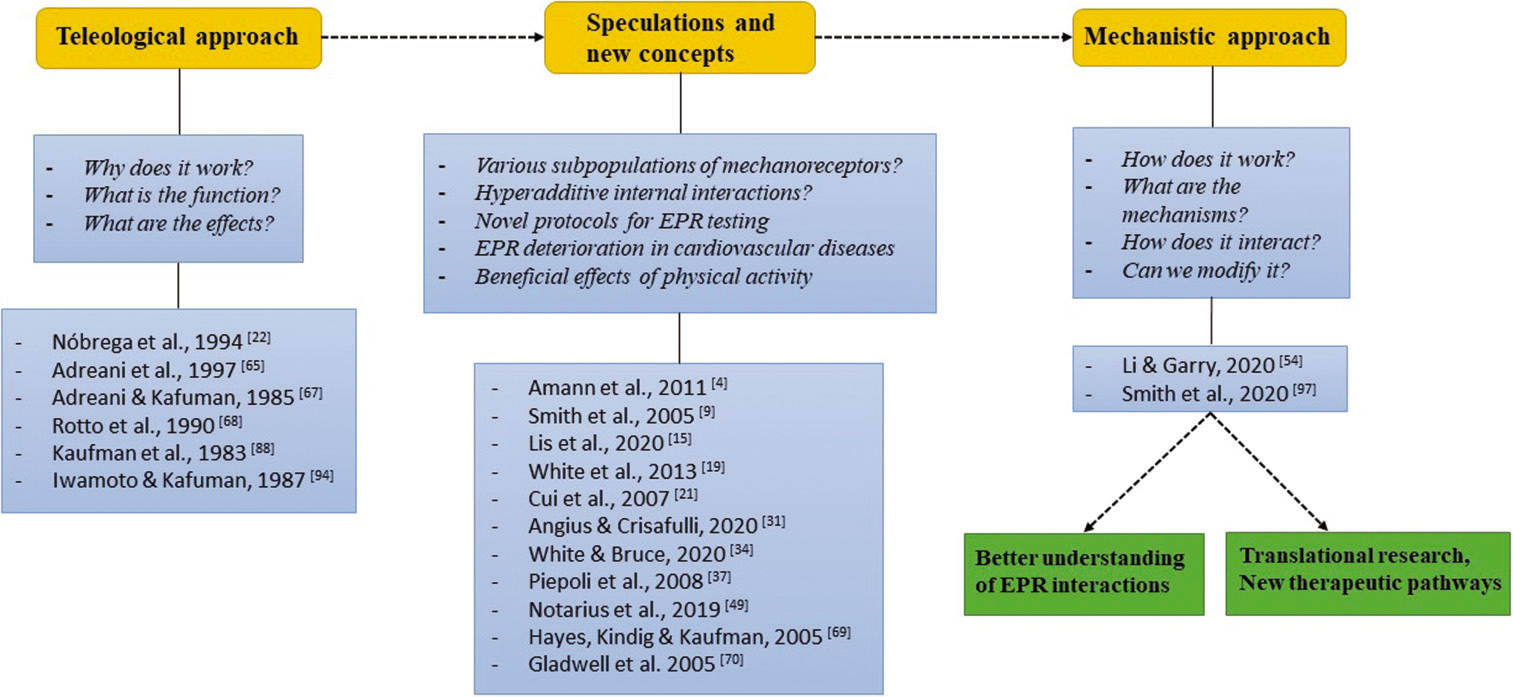
- The flow chart diagram depicts the concept of changing paradigm in exercise pressor reflex (EPR) testing. Starting with teleological approach and experiments that revealed functions and effects of EPR, going through speculations and new concepts made on the basis of teleological studies, ending on mechanistic approach, focused on precise mechanisms which enable better understanding of EPR interactions and opening new therapeutic possibilities.
Finally, studying comprehensively this intriguing, still not fully elucidated topic, one should always remember to consider not only the isolate reflexes, but interactions between them, as to “not lose sight of the forest for the trees.”
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Cardiovascular regulation by skeletal muscle reflexes in health and disease. Am J Physiol Heart Circ Physiol. 2011;301:H1191-204.
- [CrossRef] [PubMed] [Google Scholar]
- The mammalian exercise pressor reflex in health and disease. Exp Physiol. 2006;91:89-102.
- [CrossRef] [PubMed] [Google Scholar]
- Implications of group III and IV muscle afferents for high-intensity endurance exercise performance in humans. J Physiol. 2011;589:5299-309.
- [CrossRef] [PubMed] [Google Scholar]
- The mechanoreflex and hemodynamic response to passive leg movement in heart failure. Med Sci Sports Exerc. 2016;48:368-76.
- [CrossRef] [PubMed] [Google Scholar]
- Exaggerated muscle mechanoreflex control of reflex renal vasoconstriction in heart failure. J Appl Physiol 1985. 2001;90:1714-9.
- [CrossRef] [PubMed] [Google Scholar]
- Contribution of muscle afferents to the hemodynamic, autonomic, and ventilatory responses to exercise in patients with chronic heart failure: Effects of physical training. Circulation. 1996;93:940-52.
- [CrossRef] [PubMed] [Google Scholar]
- Muscle ergoreceptor overactivity reflects deterioration in clinical status and cardiorespiratory reflex control in chronic heart failure. Circulation. 2001;104:2324-30.
- [CrossRef] [PubMed] [Google Scholar]
- Mechanoreflex mediates the exaggerated exercise pressor reflex in heart failure. Circulation. 2005;112:2293-300.
- [CrossRef] [PubMed] [Google Scholar]
- Responses of glomus cells to hypoxia and acidosis are uncoupled, reciprocal and linked to ASIC3 expression: Selectivity of chemosensory transduction. J Physiol. 2013;591:919-32.
- [CrossRef] [PubMed] [Google Scholar]
- Revelations about carotid body function through its pathological role in resistant hypertension. Curr Hypertens Rep. 2013;15:273-80.
- [CrossRef] [PubMed] [Google Scholar]
- The exercise pressor reflex in animals. Exp Physiol. 2012;97:51-8.
- [CrossRef] [PubMed] [Google Scholar]
- Baroreflex control of sinus node during dynamic exercise in humans: Effect of muscle mechanoreflex. Acta Physiol (Oxf). 2008;192:351-7.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular, metabolic and plasma catecholamine responses to passive and active exercises. J Physiol Pharmacol. 2000;51:267-78.
- [Google Scholar]
- Passive bilateral leg cycling with concomitant regional circulatory occlusion for testing mechanoreflex-metaboreflex interactions in humans. Clin Auton Res. 2020;30:549-56.
- [CrossRef] [PubMed] [Google Scholar]
- Identifying the role of group III/IV muscle afferents in the carotid baroreflex control of mean arterial pressure and heart rate during exercise. J Physiol. 2018;596:1373-84.
- [CrossRef] [PubMed] [Google Scholar]
- Central and peripheral responses to static and dynamic stretch of skeletal muscle: Mechano and metaboreflex implications. J Appl Physiol 1985. 2017;122:112-20.
- [CrossRef] [PubMed] [Google Scholar]
- Augmentation of the exercise pressor reflex in prehypertension: Roles of the muscle metaboreflex and mechanoreflex. Appl Physiol Nutr Metab. 2013;38:209-15.
- [CrossRef] [PubMed] [Google Scholar]
- The pulmonary vascular response to combined activation of the muscle metaboreflex and mechanoreflex. Exp Physiol. 2013;98:758-67.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular responses to human calf muscle stretch during varying levels of muscle metaboreflex activation. Exp Physiol. 2005;90:773-81.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of muscle metabolites on responses of muscle sympathetic nerve activity to mechanoreceptor(s) stimulation in healthy humans. Am J Physiol Regul Integr Comp Physiol. 2008;294:R458-66.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular responses to active and passive cycling movements. Med Sci Sports Exerc. 1994;26:709-14.
- [CrossRef] [PubMed] [Google Scholar]
- Contribution of skeletal muscle “ergoreceptors” in the human leg to respiratory control in chronic heart failure. J Physiol. 2000;529(3):863-70.
- [CrossRef] [PubMed] [Google Scholar]
- Neural regulation of cardiovascular response to exercise: Role of central command and peripheral afferents. Biomed Res Int. 2014;2014:478965.
- [CrossRef] [PubMed] [Google Scholar]
- Chemo and ergoreflexes in health, disease and ageing. Int J Cardiol. 2005;98:369-78.
- [CrossRef] [PubMed] [Google Scholar]
- Hyperadditive ventilatory response arising from interaction between the carotid chemoreflex and the muscle mechanoreflex in healthy humans. J Appl Physiol 1985. 2018;125:215-25.
- [CrossRef] [PubMed] [Google Scholar]
- 2020 ESC Guidelines on sports cardiology and exercise in patients with cardiovascular disease. Eur Heart J. 2021;42:17-96.
- [CrossRef] [PubMed] [Google Scholar]
- The role of exercise training in heart failure. J Am Coll Cardiol. 2011;58:561-9.
- [CrossRef] [PubMed] [Google Scholar]
- A neural link to explain the “muscle hypothesis” of exercise intolerance in chronic heart failure. Am Heart J. 1999;137:1050-6.
- [CrossRef] [Google Scholar]
- Metaboreflex-mediated hemodynamic abnormalities in individuals with coronary artery disease without overt signs or symptoms of heart failure. Am J Physiol Heart Circ Physiol. 2018;314:H452-H463.
- [CrossRef] [PubMed] [Google Scholar]
- Exercise intolerance and fatigue in chronic heart failure: Is there a role for group III/IV afferent feedback? Eur J Prev Cardiol. 2020;27:1862-72.
- [CrossRef] [PubMed] [Google Scholar]
- Exercise intolerance and skeletal muscle metaboreflex activity in chronic heart failure: Do we need to recruit more muscle in exercise training? Eur J Prev Cardiol. 2020;27:1858-61.
- [CrossRef] [PubMed] [Google Scholar]
- Enhanced ventilatory response to exercise in patients with chronic heart failure and preserved exercise tolerance: Marker of abnormal cardiorespiratory reflex control and predictor of poor prognosis. Circulation. 2001;103:967-72.
- [CrossRef] [PubMed] [Google Scholar]
- The role of muscle mechano and metaboreflexes in the control of ventilation: Breathless with (over) excitement? Exp Physiol. 2020;105:2250-3.
- [CrossRef] [PubMed] [Google Scholar]
- Enhanced metaboreflex sensitivity in hypertensive humans. Eur J Appl Physiol. 2009;105:351-6.
- [CrossRef] [PubMed] [Google Scholar]
- Muscle mechanoreflex overactivity in hypertension: A role for centrally-derived nitric oxide. Auton Neurosci. 2015;188:58-63.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular and ventilatory control during exercise in chronic heart failure: Role of muscle reflexes. Int J Cardiol. 2008;130:3-10.
- [CrossRef] [PubMed] [Google Scholar]
- Muscle metaboreflex and exercise heart rate: Insights from studies in subjects with and without heart failure. J Physiol. 2010;588:2679.
- [CrossRef] [PubMed] [Google Scholar]
- Influence of the metaboreflex on arterial blood pressure in heart failure patients. Am Heart J. 2014;167:521-8.
- [CrossRef] [PubMed] [Google Scholar]
- Relationship of skeletal muscle metaboreceptors in the upper and lower limbs with the respiratory control in patients with heart failure. Clin Sci (Lond). 2002;102:23-30.
- [CrossRef] [Google Scholar]
- The ergoreflex in patients with chronic stable heart failure. Int J Cardiol. 1999;68:157-64.
- [CrossRef] [Google Scholar]
- Molecular basis for the improvement in muscle metaboreflex and mechanoreflex control in exercise-trained humans with chronic heart failure. Am J Physiol Heart Circ Physiol. 2014;307:H1655-66.
- [CrossRef] [PubMed] [Google Scholar]
- Exaggerated renal vasoconstriction during exercise in heart failure patients. Circulation. 2000;101:784-9.
- [CrossRef] [PubMed] [Google Scholar]
- Skeletal muscle metaboreceptor exercise responses are attenuated in heart failure. Circulation. 1991;84:2034-9.
- [CrossRef] [PubMed] [Google Scholar]
- Muscle metaboreflex-induced vasoconstriction in the ischemic active muscle is exaggerated in heart failure. Am J Physiol Heart Circ Physiol. 2018;314:H11-H18.
- [CrossRef] [PubMed] [Google Scholar]
- Pathophysiology of human heart failure: Importance of skeletal muscle myopathy and reflexes. Exp Physiol. 2014;99:609-15.
- [CrossRef] [PubMed] [Google Scholar]
- Exercise training prevents the exaggerated exercise pressor reflex in rats with chronic heart failure. J Appl Physiol 1985. 2010;108:1365-75.
- [CrossRef] [PubMed] [Google Scholar]
- Inspiratory muscle training improves blood flow to resting and exercising limbs in patients with chronic heart failure. J Am Coll Cardiol. 2008;51:1663-71.
- [CrossRef] [PubMed] [Google Scholar]
- Training heart failure patients with reduced ejection fraction attenuates muscle sympathetic nerve activation during mild dynamic exercise. Am J Physiol Regul Integr Comp Physiol. 2019;317:R503-12.
- [CrossRef] [PubMed] [Google Scholar]
- Forearm endurance training attenuates sympathetic nerve response to isometric handgrip in normal humans. J Appl Physiol 1985. 1992;72:1039-43.
- [CrossRef] [PubMed] [Google Scholar]
- Symptoms and quality of life in heart failure: The muscle hypothesis. Br Heart J. 1994;72:S36-9.
- [CrossRef] [PubMed] [Google Scholar]
- Progressive improvement in hemodynamic response to muscle metaboreflex in heart transplant recipients. J Appl Physiol 1985. 2013;114:421-7.
- [CrossRef] [PubMed] [Google Scholar]
- Increased metaboreflex activity is related to exercise intolerance in heart transplant patients. Am J Physiol Heart Circ Physiol. 2007;293:H3699-706.
- [CrossRef] [PubMed] [Google Scholar]
- A murine model of the exercise pressor reflex. J Physiol. 2020;598:3155-71.
- [CrossRef] [PubMed] [Google Scholar]
- Gadolinium attenuates exercise pressor reflex in cats. Am J Physiol Heart Circ Physiol. 2001;280:H2153-61.
- [CrossRef] [PubMed] [Google Scholar]
- Gadolinium inhibits group III but not group IV muscle afferent responses to dynamic exercise. J Physiol. 2009;587:873-82.
- [CrossRef] [PubMed] [Google Scholar]
- Skeletal muscle reflex-mediated changes in sympathetic nerve activity are abnormal in spontaneously hypertensive rats. Am J Physiol Heart Circ Physiol. 2011;300:H968-77.
- [CrossRef] [PubMed] [Google Scholar]
- Prostaglandins contribute to cardiovascular reflexes evoked by static muscular contraction. Circ Res. 1986;59:645-54.
- [CrossRef] [PubMed] [Google Scholar]
- Cyclooxygenase products sensitize muscle mechanoreceptors in healthy humans. Am J Physiol Heart Circ Physiol. 2004;287:H1944-9.
- [CrossRef] [PubMed] [Google Scholar]
- Further evidence that extracellular serotonin in the rostral ventrolateral medulla modulates 5-HT(1A) receptor-mediated attenuation of exercise pressor reflex. Brain Res. 2001;900:186-94.
- [CrossRef] [Google Scholar]
- Capsaicin-based analgesic balm attenuates the skeletal muscle metaboreflex in healthy humans. J Appl Physiol 1985. 2018;125:362-8.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of topical analgesics on the pressor response evoked by muscle afferents. Med Sci Sports Exerc. 2002;34:1440-5.
- [CrossRef] [PubMed] [Google Scholar]
- Capsaicin-based analgesic balm decreases pressor responses evoked by muscle afferents. Med Sci Sports Exerc. 2004;36:444-50.
- [CrossRef] [PubMed] [Google Scholar]
- Central and peripheral contributors to skeletal muscle hyperemia: Response to passive limb movement. J Appl Physiol 1985. 2010;108:76-84.
- [CrossRef] [PubMed] [Google Scholar]
- Responses of group III and IV muscle afferents to dynamic exercise. J Appl Physiol 1985. 1997;82:1811-7.
- [CrossRef] [PubMed] [Google Scholar]
- Cyclooxygenase blockade attenuates responses of group III and IV muscle afferents to dynamic exercise in cats. Am J Physiol Heart Circ Physiol. 2006;290:H2239-46.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of arterial occlusion on responses of group III and IV afferents to dynamic exercise. J Appl Physiol 1985. 1998;84:1827-33.
- [CrossRef] [PubMed] [Google Scholar]
- Sensitization of group III muscle afferents to static contraction by arachidonic acid. J Appl Physiol 1985. 1990;68:861-7.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison between the effect of static contraction and tendon stretch on the discharge of group III and IV muscle afferents. J Appl Physiol 1985. 2005;99:1891-6.
- [CrossRef] [PubMed] [Google Scholar]
- The influence of small fibre muscle mechanoreceptors on the cardiac vagus in humans. J Physiol. 2005;567:713-21.
- [CrossRef] [PubMed] [Google Scholar]
- Heart rate at the onset of muscle contraction and during passive muscle stretch in humans: A role for mechanoreceptors. J Physiol. 2002;540:1095-102.
- [CrossRef] [PubMed] [Google Scholar]
- Left ventricular volumes and hemodynamic responses at onset of dynamic exercise with reduced venous return. J Appl Physiol 1985. 1995;79:1405-10.
- [CrossRef] [PubMed] [Google Scholar]
- Passive cycling with concomitant circulatory occlusion for testing interactions between the exercise pressor reflex afferent pathways: (re)naissance or déjà vu? Clin Auton Res. 2020;30:589-90.
- [CrossRef] [PubMed] [Google Scholar]
- Passive cycling with concomitant circulatory occlusion for testing interactions between the exercise pressor reflex afferent pathways: (re)naissance or déjà vu? -authors' response. Clin Auton Res. 2020;30:591-2.
- [CrossRef] [PubMed] [Google Scholar]
- The exercise pressor reflex and chemoreflex interaction: Cardiovascular implications for the exercising human. J Physiol. 2020;598:2311-21.
- [CrossRef] [PubMed] [Google Scholar]
- Interaction between the muscle metaboreflex and the arterial baroreflex in control of arterial pressure and skeletal muscle blood flow. Am J Physiol Heart Circ Physiol. 2016;311:H1268-76.
- [CrossRef] [PubMed] [Google Scholar]
- Muscle afferent activation causes ventilatory and cardiovascular responses during concurrent hypercapnia in humans. Exp Physiol. 2012;97:208-18.
- [CrossRef] [PubMed] [Google Scholar]
- Hypoxia potentiates exercise-induced sympathetic neural activation in humans. J Appl Physiol 1985. 1991;71:1032-40.
- [CrossRef] [PubMed] [Google Scholar]
- Differential effects of metaboreceptor and chemoreceptor activation on sympathetic and cardiac baroreflex control following exercise in hypoxia in human. J Physiol. 2007;585:165-74.
- [CrossRef] [PubMed] [Google Scholar]
- A respiratory response to the activation of the muscle metaboreflex during concurrent hypercapnia in man. Exp Physiol. 2010;95:194-201.
- [CrossRef] [PubMed] [Google Scholar]
- Activation of the carotid chemoreflex secondary to muscle metaboreflex stimulation in men. Am J Physiol Regul Integr Comp Physiol. 2014;306:R693-700.
- [CrossRef] [PubMed] [Google Scholar]
- Cardiovascular responses to forearm muscle metaboreflex activation during hypercapnia in humans. Am J Physiol Regul Integr Comp Physiol. 2015;309:R43-50.
- [CrossRef] [PubMed] [Google Scholar]
- A comparison of voluntary and electrically induced contractions by interleaved 1H-and 31P-NMRS in humans. J Appl Physiol 1985. 2003;94:1012-24.
- [CrossRef] [PubMed] [Google Scholar]
- Electrical stimulation as a modality to improve performance of the neuromuscular system. Exerc Sport Sci Rev. 2007;35:180-5.
- [CrossRef] [PubMed] [Google Scholar]
- Differential changes in muscle oxygenation between voluntary and stimulated isometric fatigue of human dorsiflexors. J Appl Physiol 1985. 2006;100:890-5.
- [CrossRef] [PubMed] [Google Scholar]
- Muscle mechanoreflex induces the pressor response by resetting the arterial baroreflex neural arc. Am J Physiol Heart Circ Physiol. 2004;286:H1382-8.
- [CrossRef] [PubMed] [Google Scholar]
- Reflex cardiovascular responses evoked by selective activation of skeletal muscle ergoreceptors. J Appl Physiol 1985. 2001;90:308-16.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of static muscular contraction on impulse activity of groups III and IV afferents in cats. J Appl Physiol Respir Environ Exerc Physiol. 1983;55:105-12.
- [CrossRef] [PubMed] [Google Scholar]
- Responses in muscle afferent fibres of slow conduction velocity to contractions and ischaemia in the cat. J Physiol. 1983;342:383-97.
- [CrossRef] [PubMed] [Google Scholar]
- Role of NO in modulating neuronal activity in superficial dorsal horn of spinal cord during exercise pressor reflex. Am J Physiol Heart Circ Physiol. 2002;283:H1012-8.
- [CrossRef] [PubMed] [Google Scholar]
- Central projections from ergoreceptors (C fibers) in muscle involved in cardiopulmonary responses to static exercise. Circ Res. 1981;48:I48-62.
- [Google Scholar]
- Convergence properties of solitary tract neurones driven synaptically by cardiac vagal afferents in the mouse. J Physiol. 1998;508(1):237-52.
- [CrossRef] [PubMed] [Google Scholar]
- Pattern of cardiorespiratory afferent convergence to solitary tract neurons driven by pulmonary vagal C-fiber stimulation in the mouse. J Neurophysiol. 1998;79:2365-73.
- [CrossRef] [PubMed] [Google Scholar]
- Caudal ventrolateral medullary cells responsive to muscular contraction. J Appl Physiol 1985. 1987;62:149-57.
- [CrossRef] [PubMed] [Google Scholar]
- c-Fos expression in the midbrain periaqueductal gray during static muscle contraction. Am J Physiol Heart Circ Physiol. 2000;279:H2986-93.
- [CrossRef] [PubMed] [Google Scholar]
- Group III and IV muscle afferents contribute to ventilatory and cardiovascular response to rhythmic exercise in humans. J Appl Physiol 1985. 2010;109:966-76.
- [CrossRef] [PubMed] [Google Scholar]
- Metabo-and mechanoreceptor expression in human heart failure: Relationships with the locomotor muscle afferent influence on exercise responses. Exp Physiol. 2020;105:809-18.
- [CrossRef] [PubMed] [Google Scholar]






