Translate this page into:
Behavioural and molecular effects of alcohol in the stress model of zebrafish
*Corresponding author: Rafael Antonio Vargas Vargas, Department of Basic Medical Sciences, School of Medicine, Universidad Militar Nueva Granada, Bogotá, Colombia. rvargas3200@hotmail.com
-
Received: ,
Accepted: ,
How to cite this article: Franco-Restrepo JE, Vargas RA. Behavioural and molecular effects of alcohol in the stress model of zebrafish. Indian J Physiol Pharmacol 2021;65:153-61.
Abstract
Objectives:
Stress and anxiety disorders are common health problems that have been related to an increase in the likelihood of developing addictions, which have individual and social consequences. Although socially acceptable, alcohol is a substance that can generate dependence and abuse. Alcohol misuse, its relationship with stress and its consequences have been studied; however, multiple limitations are placed on clinical research in humans. In this exploratory work, we analysed the behavioural and molecular effects of joint exposure to ethanol and an unpredictable stress protocol (USP) in adult zebrafish.
Materials and Methods:
Adult zebrafish behaviour was studied employing unpredictable stress and behavioural tests. The tests were performed in stressed and nonstressed animals with and without exposure to known concentrations of alcohol. To evaluate the behaviour, tracking techniques were used on video recordings and parameters such as distance travelled, swimming speed and place preference as well as aggression patterns with mirror proximity tests were measured. In the control and 0.75% alcohol group, the expression of candidate stress-related genes (slc6a4a, slc6a3, comta and bdnf3) was analysed by RT-qPCR.
Results:
The results showed that concentrations of 0.75% alcohol reduced the locomotor activity of the fish, which can be interpreted as an increase in the anxiolytic effect of alcohol under nonstress conditions. Expression of comta, bdnf3 and slc6a3 was reduced in the stress and stress plus 0.75% ethanol groups and expression of slc6a4a was increased in the stress plus 0.75% alcohol group.
Conclusion:
Our exploratory work contributes novel insights about the molecular and behavioural effects of the combination of unpredicted stress and alcohol misuse. The USP and ethanol exposure increase anxiety behaviour and reduce the expression of genes involved in brain homeostasis. Future study of other pharmacological compounds and additional genes will be helpful for a deeper understanding of the molecular mechanisms involved in the response to stress and alcohol use.
Keywords
Neurosciences
Mental health
Addiction
Anxiety
Animal models
INTRODUCTION
Anxiety and stress have been linked to an increased likelihood of developing addictions, such as alcohol misuse and addiction.[1] Predisposing factors for alcoholism include psychosocial stress and affective disorders, which are also risk factors for chronic diseases.[1,2] Moreover, psychosocial stress might potentiate the consumption and negative effects of alcohol and reinforce abnormal drinking behaviour.[2] Alcohol consumption has also been linked to aggressive behaviours, such as violent and sexual assault, child abuse, rape, domestic violence, aggressive acts toward the self and homicides, among others.[3] Aggressive behaviours are highly related to alcohol due to the lowered inhibition in the central nervous system by GABA.[3] Although alcohol abuse and its consequences have been studied in humans, multiple limitations are involved in such studies; thus, few clinical research studies have been performed on humans.[4] Furthermore, the complexity of the human brain limits the potential to identify basic mechanisms related to the relationship among stress, anxiety and alcohol misuse; thus, basic studies in animal models are required to investigate its possible pathophysiology.[5]
Adult zebrafish are widely used models for research in multiple areas of the neuroscience, such as learning and memory,[6,7] the sleep-wake cycle,[8] social behaviour,[9] stress, anxiety and addictions[10] and such areas have been explored using zebrafish models. Acute stress and anxiety have been studied in adult zebrafish using tests similar to those used in rodents.[11] For this purpose, several behavioural tests have been used, such as those based on novel tanks, open fields, light-dark preferences, predator avoidance and social preferences.[12] Stress has been induced in adult zebrafish models using unpredictable stress protocols (USPs) with repetitive stress, including immobilization, extreme changes in water temperature, net chase, exposure to predators, low water levels, multiple tank changes and social isolation.[13-15] Several investigations have shown responses similar to those observed in other animal models.[16] In general, zebrafish are useful for studying stress and anxiety because their environment is easy to control, they can be easily exposed to substances, their maintenance is inexpensive and their development is fast. One of the main advantages of using a zebrafish model for anxiety is the clearly visible cues of anxious behaviour in zebrafish, such as a reduction in exploratory behaviour, preference for darkness (scototaxis), deep swimming tendency (geotaxis), preference of peripheral zones (thigmotaxis), greater frequency and duration of immobile states (freezing) and increase in erratic movements.[17,18]
Furthermore, the zebrafish model has also been used to study addictions, especially the acute and chronic effects of alcohol intake, including its impacts on behaviour.[19] Acute exposure to low and moderate alcohol concentrations in adult zebrafish induces behavioural changes that include decreased anxiety, increased locomotion, increased aggressiveness and changes in place preference and shoaling behaviour.[20] Chronic exposure to ethanol produces tolerance and abstinence increases anxiety and decreases the tendency to shoaling behaviour.[21] These behavioural variations are associated with increased levels of dopamine and serotonin and decreased levels of glutamate and GABA neurotransmitters.[22] Serotonin activity has been linked with the regulation of aggressive behaviour, stress, arousal and movement, among other physiological processes in the central nervous system in several species and serotonin levels have been shown to be inversely correlated with mood (e.g. anxiety, depression and aggressiveness).[23] Furthermore, several studies have found a relationship between gene expression and behaviour.[24] In this context, some of the main candidate genes are brain-derived neurotrophic factor (bdnf), which is related to the growth, differentiation and maintenance of neurons.[25] The enzyme catechol-methyltransferase (comt) is responsible for the degradation of catecholamines, including the regulation of dopamine levels.[26] The dopamine transporter (scl6a3) and serotonin transporter (slc6a4) are responsible for dopamine and serotonin reuptake from the synaptic gap and their genetic loss or pharmacological blockage has been shown to alter neurotransmission and behaviour.[27]
In the previous years, research has been carried out in larval and adult stages of zebrafish to determine the mRNA expression of candidate genes and their relationship with exposure to substances or stress. However, because of the limited studies, additional investigations are required on the combined effects of acute environmental stress and alcohol consumption in adult zebrafish, their behavioural changes and their relationship with gene expression. In this pilot work, we explored and analysed the behavioural and molecular effects of combined exposure to an acute USP, which similar to those proposed in other animal models to induce stress[16] and ethanol exposure effects[22] in adult zebrafish.
MATERIALS AND METHODS
Animals
Male wild-type short-fin zebrafish of 4–6 months of age were obtained from a local animal store in Bogotá, Colombia. The fish were kept in 7 L tanks (400 ml/fish) at a temperature of 27 ± 1°C, pH between 6.8 and 7.4 and light/dark cycle of 12:12 h and they were fed once a day in the light phase with a TetraMin (Tetra GMBH, Germany) standardized diet.[28] In addition, each tank had an aeration system and filters to guarantee adequate water conditions for the animals, which had a 2-week adaptation period before any test. A total of 84 male zebrafish were used for the experimental tests and they had average weights and sizes of 362 mg and 2.6 cm, respectively, and were distributed randomly into control and experimental groups. Only males were used due to their differences in the aggressiveness tests and biochemical markers relative to that in females.[29] All procedures were carried out based on the ethical principles of the 3 Rs and the Guide for the Care and Use of Animals in Neuroscience and in Behavioural Research.[30] All protocols were approved by the ethics committee in animal experimentation of Universidad Antonio Nariño (June 06/2017).
Environmental stress and acute ethanol exposure
To induce stress, an acute USP was performed for 3 days [Figure 1]. This protocol included a tank change on day 1 with the water temperature at 33°C for 30 min and then at 23°C for 30 min. On day 2, the fish were chased with a net for 10 min. On day 3, immobilization was carried out in 2 ml microcentrifuge tubes (open at both sides to allow oxygenated water circulation) for 60 min [adapted from 14]. The USP was carried out in groups of six fish in a different tank from the storage tank. On day 4, zebrafish were exposed to ethanol (70% ethyl alcohol, Tecnoquímicas, Bogotá, Colombia) at different concentrations (0, 0.25, 0.5, 0.75 and 1%, n = 12 each group). All dilutions were performed in a 1 L tank. Zebrafish were exposed to ethanol at 0.25 and 0.5% for 40 min, the minimum time necessary to reach concentrations of alcohol in the blood and brain similar to the concentration in the tank.[20,31] Zebrafish exposed to ethanol at 0.75 and 1% showed buoyancy and balance loss after 15–20 min and an exposure time of 40 min was fatal in the pilot tests. Therefore, the zebrafish were exposed for 20 min at these doses (0.75 and 1%). The fish were transferred to tanks with fresh water and behavioural analyses were performed after a 5-min recovery period. In addition, due to the significant differences in locomotor behaviour, a behavioural test without stress was performed with ethanol exposure at 0.75%. First, the behaviour tests were recorded without alcohol; then, the same group was exposed to 0.75% alcohol and a second recording was made (prepost test). A total of 14 groups with six fish each were generated, with seven groups for the swimming deep test (control, stress only, stress plus 0.25, 0.5, 0.75 and 1% and prepost of 0.75% without stress) and seven groups for the mirror proximity test.

- Overview of the USP, ethanol exposure and behavioural assessment. The US protocol was applied for 3 days. On day 1, water in the tanks was heated and then cooled and the zebrafish were exposed for 30 min at each temperature. On day 2, the zebrafish were chased with a net for 10 min. On day 3, the zebrafish were placed in a 2 ml microcentrifuge tube for 60 min. Finally, on day 4, the fish were exposed to ethanol (0, 0.25, 0.5, 0.75 and 1%) and mirror proximity (top) and swimming deep (bottom) behavioural tests were carried out. M: Mirror.
Behavioural tests and analysis
To evaluate the fish behaviour, computerized tracking techniques were used with the video recordings. The videos were analysed and parameters such as total distance travelled, swimming speed and place preference in the swimming deep test as well as the time spent in the region of interest (ROI) in the mirror proximity test, were measured. All behavioural tanks were custom-made in acrylic (Surtiacrylicos, Bogotá, Colombia).
After recovery in a tank with fresh water, the zebrafish were transferred to a different tank to assess their behaviour and a 5-min video was recorded with a digital camera (iPhone 6, Apple, California, USA) for further analysis. Only one fish was used in each test at the time. The swimming deep test and mirror proximity test were assessed with two different tanks; the mirror proximity test was evaluated using a rectangular tank with measurements of 18 × 9 × 10 cm (length, width and height) and it was filled up to 4 cm with fresh water. A 7 cm long mirror was introduced to assess the time spent near its close reflection (contact area of 5 cm with respect to the mirror) and video recording was performed from the top view of the tank [Figure 2A]. The time spent near the mirror could indicate aggressive behaviour.[32]

- (A) Top view of the tank used in the mirror proximity test; the dotted lines limit the defined contact area in relation to the mirror (located in the upper region of the image). Yellow solid lines show an example of tracking with the ImageJ software and the AnimalTracker plugin. (B) Side view of the trapezoidal tank used in swimming deep test. The lower third is an area of 5 cm in height and the upper third is 10 cm in height.
The swimming deep test was assessed using a trapezoidal tank, with measurements of 23 cm and 28 cm in length at the base and upper region, respectively, 7 cm in width and 15 cm in height. This shape allowed us to assess the time spent in different portions (lower third and upper two thirds) as an index of locomotor activity and anxiety. Video recording from the lateral view of the tank was carried out [Figure 2B]. We used freely available ImageJ software (NIH, Maryland, USA)[33] with the AnimalTracker plugin (developed by Gulyas et al.),[34] which allows for automatic analyses of behaviour.[35] The video recording settings were as follows: AVI format, MJPEG compressed, 30 frames per second and 840 × 420 pixel resolution. Each test was 5 min in length and all 5 min of recorded video were analysed. AnimalTracker provided data on the total distance travelled, average speed, total freezing time, time in the ROI and total trajectory images. The number of freezing episodes during the test was analysed manually.
Euthanasia and brain extraction
Once the behaviour test was completed, cryoanesthesia was performed at −4°C for 20 min and then the fish were transferred to a 1.5 ml microcentrifuge tube with 700 ml of dH2O and stored at −20°C. To prevent possible interactions in molecular tests, the use of tricaine (MS-222) was avoided. Strykowski and Schech[36] reported a higher efficacy of euthanasia at 4°C or less compared to tricaine and although this method requires a longer time of exposure, it avoids the introduction of an additional variable for molecular analyses. Whole brain extraction was performed by removing the skull of the fish and making cuts in the spinal cord, optic nerves and cranial nerves. After removing the whole brain, it was stored at −20°C until RNA extraction.
Total RNA extraction and cDNA synthesis
Once the whole brain sample was extracted, total RNA extraction was performed following the TRIzol protocol (Invitrogen, California, USA)[37] to subsequently generate first-strand cDNA using the M-MLV Reverse Transcriptase kit (Invitrogen, California, USA).[38] Small adjustments were made to the RNA extraction protocol due to the weight of the brains (<50 mg) and one brain per tube was ultimately used for RNA extraction and cDNA generation. The samples were stored at −70°C.
Quantitative real time PCR
qPCR was performed using BrightGreen 2X qPCR MasterMix (ABM Inc., Vancouver, Canada) on a CFX96 Touch Real-Time PCR Detection System (Bio-Rad, California, USA). The final volume per PCR tube was 10 μl and it included 2 μl of cDNA (10 ng), 5 μl of BrightGreen MasterMix, 0.8 μl of each primer and 1.4 μl of molecular grade water.[39] The parameters of the qPCR were 95°C for 2 min, followed by 40 cycles at 95°C for 20 s, 55°C for 20 s and 72°C for 20 s (Adapted from 36). Three samples from the three experimental groups (control, stress only and stress plus 0.75% ethanol) were used and each sample was run in triplicate. This protocol was implemented because of the cost of the molecular tests and because these groups presented significant differences. The elf1a gene (elongation factor 1 alpha 1, like 1) was used as a reference gene because it has low expression variability in zebrafish.[40,41] The mRNA expression of genes encoding slc6a4a, slc6a3, comta and bdnf3 was determined in brain samples. These orthologs were selected due to their homology with human genes. Sequences of qPCR primers were taken from the previous articles and are available. qPCR data for gene expression were analysed with the comparative CT method (2−ΔΔCT) and the fold change was calculated for candidate genes in the experimental groups and compared with that of the control group and the reference gene.[42]
Statistical analysis
Statistical analyses were carried out using SPSS statistics software V.18 (IBM, New York, USA) and JASP V.0.10.2. To evaluate the differences in behavioural parameters between groups, an ANOVA of two or more factors was performed followed by Tukey’s HSD (honestly significant difference) multiple comparison test, with a reported level of significance at P ≤ 0.05.
The prepost group was analysed using a paired t-test and a two-way ANOVA was used to determine differences in mRNA expression (fold changes) between groups.
RESULTS
In the present pilot work, we explored and analysed the behavioural and molecular effects of combined exposure to an unpredicted stress protocol and ethanol in adult zebrafish. The animals exposed to the USP underwent a different type of environmental stress each day. The experimental groups subjected to the combination of stress and different ethanol concentrations were compared with the control groups. Gene expression was analysed in animals from the three experimental groups (control, stress and stress plus 0.75%) based on a qPCR of the cDNA from brain samples.
Swimming deep tests in zebrafish exposed to both unpredicted stress and ethanol
The following parameters were evaluated in the trapezoid tank: Total distance travelled, average speed, number of freezing episodes and total duration of freezing. In addition, the exploration of specific tank areas was evaluated by measuring the dwelling time in the lower and upper third parts as well as the total distance and average speed in each zone.
Adult zebrafish exposed to both unpredicted stress and ethanol had a shorter distance travelled than the control group. The largest difference in the distance travelled was between the control group and the group exposed to 0.75% ethanol (P < 0.05, F = 10.48) [Figure 3A]. Similarly, in terms of average velocity, the differences were mainly between the control group and the group exposed to 0.75% ethanol and the USP (P < 0.05, F = 10.38) [Figure 3B]. On the other hand, the total freezing time was longer in the group exposed to 0.75% ethanol and the USP than in the other groups, especially the control group (P < 0.05, F = 9.07 ) [Figure 3C]. There were no significant differences in the number of freezing episodes between groups.
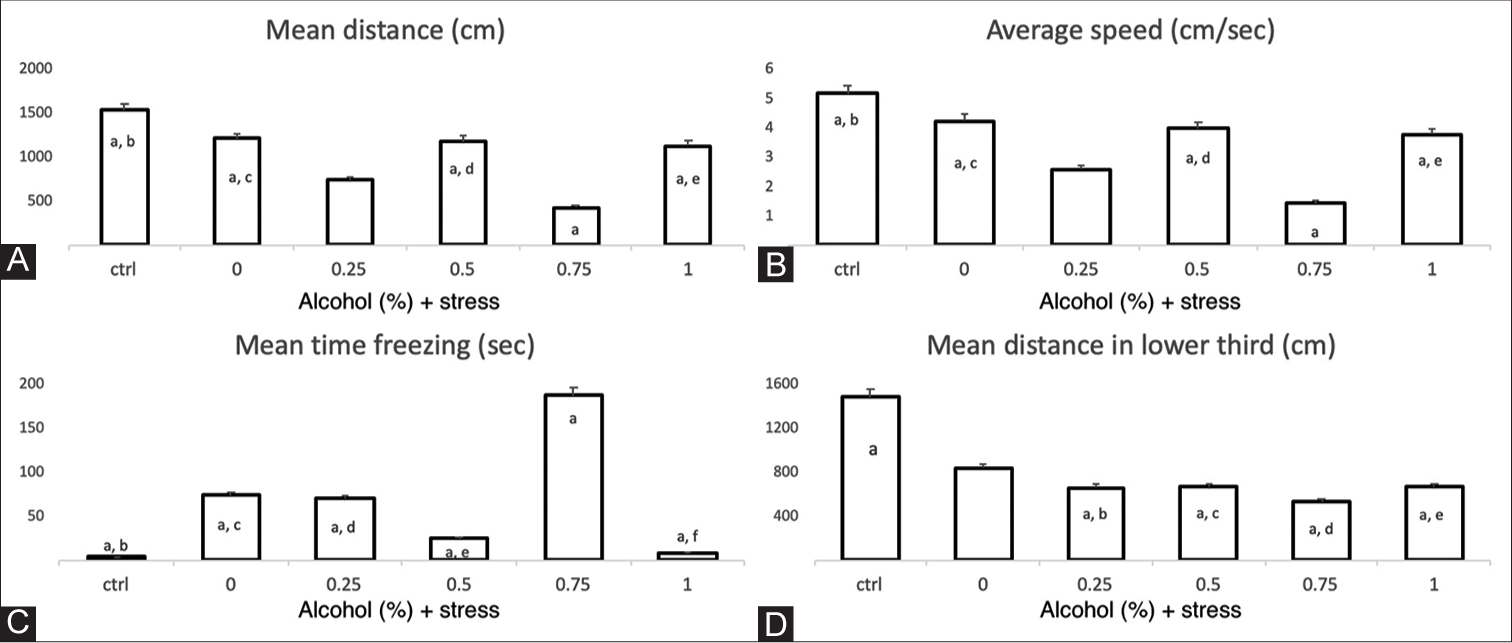
- Locomotion parameters and swimming deep test. X-axis of each graph corresponds to the control (ctrl) group and US protocol plus ethanol dose exposure groups. The individual letters correspond to the group that presents significant differences in relation to the other groups. A. Mean distance traveled. a, b: p = 5.18 x 10-8; a, c: p = 0.000096; a, d: p = 0.00021; a, e: p = 0.00064. ANOVA p = <0.001, F = 10.48. The bars indicate the means with the standard error (EE), n = 12 per group. B. Average speed. The individual letters correspond to the group that presents significant differences in relation to the other groups. a, b: p = 7.41 x 10-8; a, c: p = 0.000049; a, d: p = 0.00027; a, e: p = 0.00094. ANOVA p = <0.001, F = 10.38. The bars indicate the means with the standard error (SE), n = 12 per group. C. Freezing time. The individual letter (a) corresponds to the group that presents significant differences in relation to the other groups. a, b: p = 3.46 x 10-6; a, c: p = 0.0079; a, d: p = 0.0056; a, e: p = 0.000041; a, f: p = 6.47 x 10-6. ANOVA p = <0.001, F = 9.07. The bars indicate the means with the standard error (SE), n = 12 per group. D. Total distance in the lower third of the tank in the swimming deep test. a, b: p<0.05; a, c: p<0.05; a, d: p<0.05; a, e: p<0.05. ANOVA p = 0.014, F = 3.35. The bars indicate the means with the standard error (SE), n=6 per group.
The group exposed to 1% ethanol and the USP presented a shorter exploration time on average in the lower third of the tank of 186.1 s, while animals exposed to 0.75% ethanol and the USP showed an average exploration time of 297.6 s; however, there were no statistically significant differences between them (P = 0.11) or with the other groups. Fish exposed to stress plus ethanol (0.25, 0.5, 0.75 and 1%) travelled a smaller distance in the lower third than the control group, with statistically significant differences (P = 0.014, F = 3.35) [Figure 3D], although in the upper third portion of the tank, there were no statistically significant differences in the time of stay, total distance travelled or average speed.
Mirror proximity tests in zebrafish exposed to both unpredicted stress and ethanol
In the mirror proximity test, the total distance travelled, the average speed and the time spent in the contact area near the mirror were evaluated. The distances travelled by the groups exposed to concentrations of 0.25% and 0.75% ethanol and the USP were shorter (97 cm and 220 cm) in contrast to the control group (463 cm), with a P < 0.05 and F = 4.81 in both cases [Figure 4A]. There were no significant differences in the length of stay in the contact zone between the different groups exposed to stress and ethanol compared with the control group or differences between the groups, with P = 0.46 and F = 0.93 [Figure 4B].
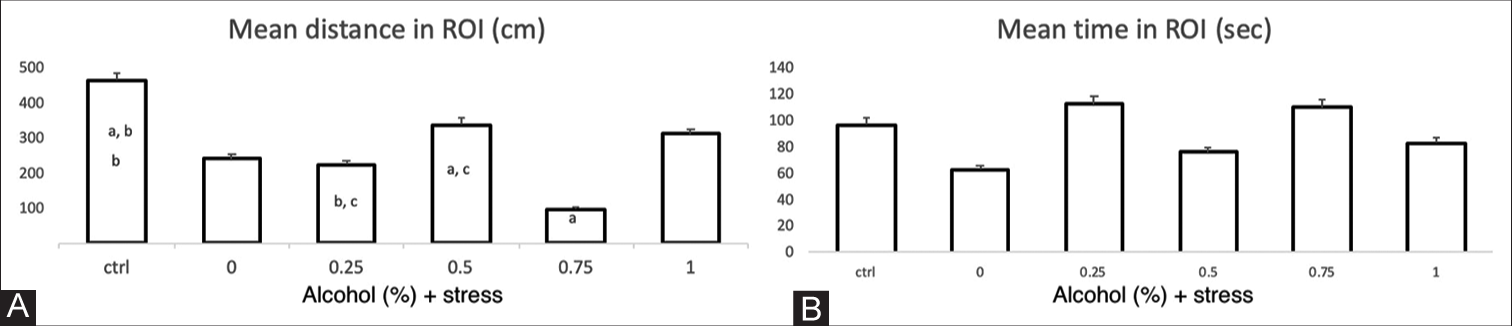
- Mirror proximity test parameters. The X-axis of each graph corresponds to the control group and the US protocol plus ethanol dose exposure groups. (A) Mean distance in the region of interest (ROI) (cm). The individual letters correspond to the group that presents statistically significant differences in relation to the other groups. a, b: P=0.00074; a, c: P=0.045; b, c: P=0.048. ANOVA P=0.002, F=4.81. The bars indicate the means with the standard error (SE), n=6 per group. (B) Mean time in the ROI. No significant differences were found, ANOVA P=0.377, F=1.097.
Overall, these data show that the USP, mainly with alcohol concentrations of 0.75%, modifies the responses in the swimming deep tests and mirror proximity test and leads to a reduction in activity.
Behavioural tests in animals exposed to ethanol without stress
A prepost alcohol test was performed for both behavioural tests (n = 6 per group) to evaluate differences relative to the animals exposed to stress. Zebrafish exposed to 0.75% alcohol (post) showed a significant decrease in locomotion compared with 0% (pre) [Figure 5A and B], having a reduction in distance travelled and in average speed (t = 2.459, P = 0.032; t = 2.63, P = 0.023, respectively). An analysis of behavioural paradigms [Figure 5C-E] did not show differences in distance and average speed for the group exposed to alcohol (mirror proximity test: t = 1.639, P = 0.162; t = 1.609, P = 0.169; swimming deep test: t = 1.914 P = 0.114; t = 1.727 P = 0.145). For the swimming deep test, the mean time spent in the lower third showed no differences (t = −0.197 P = 0.852) [Figure 5C] and the mean distance and average speed in the lower third did not show significant differences relative to the pre-test (t = 0.812 P = 0.453; t = 1.907 P = 0.115, respectively). For the mirror proximity test in the prepost test, the time spent in the ROI was not significantly increased in the alcohol exposure group (t = −1.261 P = 0.263) [Figure 5F] and the average speed (t = 2.749 P = 0.040) in the lower third was decreased compared with the pre-test. It is important to mention that the fish did not show freezing in the prepost tests relative to those under stress plus 0.75% alcohol.

- Graphs of the 0.75% prepost test without stress, locomotion parameters. A and B Mean distance and average speed of the swimming deep test and mirror proximity test, respectively, n = 12 per group. C Mean time spent in the lower third in the swimming deep test. D and E Mean distance and average speed, respectively. F Mean time spend in ROI in mirror proximity test. The x-axis indicates the behavioural tests, pre indicates 0% and post, 0.75% of alcohol. n = 6 per group. The bars indicate the means with the standard error (SE). *, P<0.05.
mRNA expression
Gene expression of the four candidate stress-related genes was analysed in three experimental groups: Control, stress and stress plus 0.75% alcohol. A significant fivefold downregulation was observed for comta and bdnf3 in the stress and stress plus 0.75% groups compared with the control group (F = 30.17, P = <0.001; F = 12.57, P = 0.007, respectively) [Figure 6A and B]. Compared with the control group, the gene expression of slc6a3 was significantly downregulated in the stress group (F = 7.406, P = 0.024) [Figure 6C]. In contrast, the expression of the slc6a4a gene was upregulated but without significant differences (F = 1.325, P = 0.334).
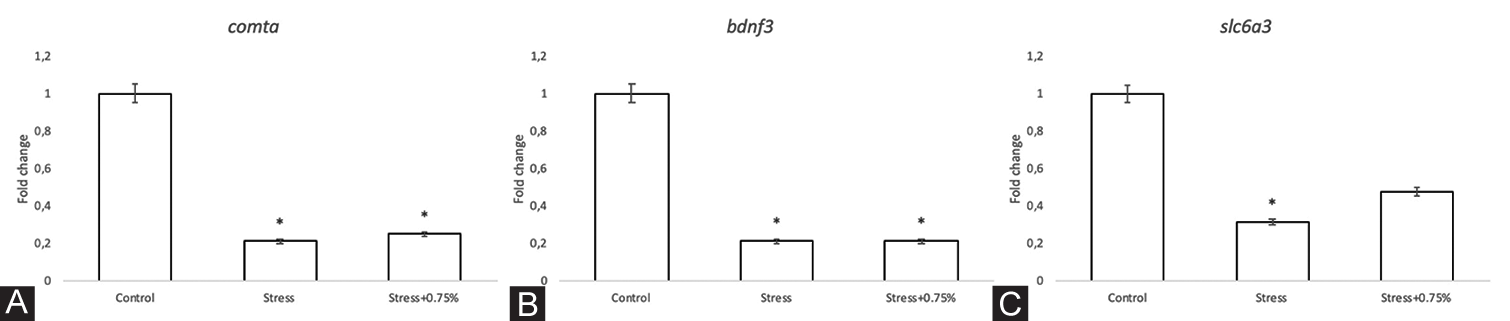
- mRNA expression. Fold changes relative to elf1a. A. comta. Groups exposed to stress and stress plus 0.75% alcohol showed a decrease of 5-fold. F = 30.17, p = <0.001 B. bdnf3. Groups exposed to stress and stress plus 0.75% alcohol showed a decrease of a 5-fold. F = 12.57, p = 0.007 C. slc6a3. Groups exposed to stress and stress plus 0.75% alcohol showed a decrease of one third to one half, respectively. F = 7.406, p = 0.024. The bars indicate the means with the standard error (SE), n = 3 per group, samples were run in triplicate. *, P<0.05.
DISCUSSION
In the present exploratory work, we analysed the behavioural and molecular effects of combined exposure to unpredicted stress and alcohol in adult zebrafish. Previously, the effects of stress and ethanol have been studied separately and mainly in zebrafish larvae. However, in the present study, the combined effect of acute stress associated with an acute dose of alcohol was unexplored.
In animal models, different protocols are used to induce stress and anxious behaviour, including physical stress (single-prolonged and restraint stress, foot shock, stress-enhanced fear learning and underwater trauma), psychological stress (predator-based psychosocial and predator scent stress) and social stress (housing and social instability and early life stress). The USP, a type of physical stress, has been proposed as a model to study anxiety, depression and mood disorders.[16,18] This model has been adapted to the zebrafish model with similar results that have indicated alterations in the physiological and biochemical responses to stress.[14,15]
In the USP implemented in this pilot work, we changed the stressing factor every day to avoid habituation, which has been suggested by other researchers.[15] Given the evidence reported, we consider that the use of an anxiogenic conditioning stimulus (manipulation with a net), in addition to the use of environmental stress factors that change between days (temperature, persecution and immobilization), are elements that can lead to anxiety and other behavioural and physiological responses that are common in many mental disorders.
The effect of alcohol on anxiety and aggressiveness in this model was evaluated using concentrations of 0.25, 0.5, 0.75 and 1% alcohol. Blood alcohol levels reached with these concentrations have been measured in the previous studies using spectrophotometry techniques and have shown that exposure to concentrations of 0.5% alcohol for 10 min led to serum concentrations of 0.065% and concentrations of 1% led to serum concentrations of 0.1%.[20] In humans, the legally permitted concentrations must be <0.08%. However, it is notable that at alcohol concentrations of 0.06%, there are already clinical manifestations of toxicity that include neurological alterations with sensorimotor impairment, decreased reflexes, dysmetria, decreased peripheral vision and alterations in reasoning.[43] To ensure adequate serum concentrations in this study, we increased the exposure time to 40 min (alcohol 0.25% and 0.5%) and 20 min (with alcohol at 0.75% and 1%) because the USP and an exposure of 40 min at these higher concentrations caused toxicity and mortality in some fish. Exposure to ethanol has previously been carried out without reported mortality; however, in the present exploratory work, exposure for 40 min was fatal, which was probably due to the associated stress factor. The causes of death are beyond the scope of this work.
In other models, it has been observed that low concentrations of alcohol are associated with a disinhibition of behaviour and a tendency toward aggression, which is partly explained by the GABAergic effect of alcohol. In contrast, at high concentrations, ethanol is a general depressant. In zebrafish, low and intermediate doses of alcohol (0.25–05%) induce a state of arousal and high aggression. Concentrations of alcohol at 1% cause motor alterations and higher doses can cause lethargy.[24]
CONCLUSION
The results presented here show a depressive effect of alcohol on exploratory behaviour and a decrease in aggressive responses in a model of US. This effect has previously been described in adult zebrafish under the effect of alcohol but without prior exposure to environmental stress.[20] In our exploratory work, the largest effect of alcohol plus stress was achieved with alcohol concentrations of 0.75%, which led to a decrease in total distance travelled and swimming speed and an increase in immobility times. This result is contrary to what happens under normal conditions, as observed in the control group and nonstressed zebrafish. A possible explanation may be related to the effect of stress on neurotransmitters, especially depletion of glutamate and increases in GABA.[21,27,31] Alcohol might enhance the inhibitory effect of GABA on stress and anxiety conditions, decreasing exploratory and aggressive behaviours and increasing freezing times. An additional element to be considered is the associated between acute exposure to alcohol and increased dopamine in a dose-dependent manner and an increase in serotonin at a dose of 1% alcohol, which is associated with a reduction in the tendency toward clumping.[27,31] Researchers have suggested that dopaminergic reinforcement circuits are involved in the feeling of well-being and protection generated by grouping. In the case of repeated stress, a decrease in 5HIAA was observed and exposure to alcohol concentrations at 1% induced an increase in dopamine without affecting 5HIAA.[41]
Although the results of gene expression presented here did not show significant differences in slc6a4a, the previous works found a direct relationship between serotonin levels and transporter expression.[44] Previously, overexpression of the serotonin transporter was described in 72-h post-fertilization larvae exposed to ethanol and reduced expression of the serotonin transporter was found in adults exposed to ethanol.[41] bdnf has been studied in animal models of stress and in postmortem brains of humans with neuropsychiatric disorders. The results presented here showed a decrease in bdnf3 in the whole brain in the stress and stress plus alcohol groups, suggesting that stress plays an important role in the regulation of neuronal maintenance and might be related to the decrease in bdnf3 expression. On the other hand, as comt regulates the levels of catecholamines, its genetic variants and its expression levels have shown a relationship with neuropsychiatric disorders in humans and behavioural changes in animal models.[45,46] The results shown here indicate that comta expression decreases, mainly in the group exposed to stress plus alcohol.
Future molecular analyses would allow for the identification of the effects of US and alcohol on additional genes involved in multiple neurotransmission and neuroplasticity mechanisms.[47] It is expected that the information obtained from this study will be useful to advance the knowledge about the factors related to alcohol misuse around the world and its relationship with aggression, anxiety and stress-related disorders, stress lifestyles and increased alcohol consumption.[48,49] In addition, this experimental model could be helpful for identifying new psychoactive serotoninergic or catecholaminergic substances and exploring their potential use in the treatment of anxiety disorders, pathological aggression and substance use disorders.[50]
Funding and acknowledgment
The authors thank Diego A. Forero MD, PhD. for his guidance and advice with the genetic tests and analysis of the gene expression results.
Compliance with ethical standards
The research presented in the manuscript has been conducted in accordance with ethical standards.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Stress and drug abuse In: Techniques in the Behavioral and Neural Sciences. Vol 15. Netherlands: Elsevier; 2005. p. :333-56.
- [CrossRef] [Google Scholar]
- Anxiety, anxiety sensitivity, and perceived stress as predictors of recent drinking, alcohol craving, and social stress response in heavy drinkers. Alcohol Clin Exp Res. 2017;41:836-45.
- [CrossRef] [PubMed] [Google Scholar]
- Alcohol and violence: Neuropeptidergic modulation of monoamine systems. Ann N Y Acad Sci. 2015;1349:96-118.
- [CrossRef] [PubMed] [Google Scholar]
- The proximal effects of acute alcohol consumption on male-to-female aggression: A meta-analytic review of the experimental literature. Trauma Violence Abuse. 2016;17:520-31.
- [CrossRef] [PubMed] [Google Scholar]
- Post-traumatic stress disorder. N Engl J Med. 2017;376:2459-69.
- [CrossRef] [PubMed] [Google Scholar]
- The zebrafish brain in research and teaching: A simple in vivo and in vitro model for the study of spontaneous neural activity. Adv Physiol Educ. 2011;35:188-96.
- [CrossRef] [PubMed] [Google Scholar]
- Zebrafish and relational memory: Could a simple fish be useful for the analysis of biological mechanisms of complex vertebrate learning? Behav Proc. 2017;141:242-50.
- [CrossRef] [PubMed] [Google Scholar]
- The ontogeny of sleep-wake cycles in zebrafish: A comparison to humans. Front Neural Cir. 2013;7:178.
- [CrossRef] [PubMed] [Google Scholar]
- Social behavior: A neural circuit for social behavior in zebrafish. Curr Biol. 2018;28:R828-30.
- [CrossRef] [PubMed] [Google Scholar]
- Zebrafish models of anxiety-like behaviors In: The Rights and Wrongs of Zebrafish: Behavioral Phenotyping of Zebrafish. Cham: Springer; 2017. p. :45-72.
- [CrossRef] [Google Scholar]
- Translating rodent behavioral repertoire to zebrafish (Danio rerio): Relevance for stress research. Behav Brain Res. 2010;214:332-42.
- [CrossRef] [PubMed] [Google Scholar]
- The developing utility of zebrafish models of neurological and neuropsychiatric disorders: A critical review. Exp Neurol. 2018;299:157-71.
- [CrossRef] [PubMed] [Google Scholar]
- Neuroendocrine regulation of the stress response in adult zebrafish, Danio rerio. Prog Neuropsychopharmacol Biol Psychiatry. 2015;60:121-31.
- [CrossRef] [PubMed] [Google Scholar]
- Unpredictable chronic stress model in zebrafish (Danio rerio): Behavioral and physiological responses. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35:561-7.
- [CrossRef] [PubMed] [Google Scholar]
- Modeling consequences of prolonged strong unpredictable stress in zebrafish: Complex effects on behavior and physiology. Prog Neuropsychopharmacol Biol Psychiatry. 2018;81:384-94.
- [CrossRef] [PubMed] [Google Scholar]
- Animal models for posttraumatic stress disorder: An overview of what is used in research. World J Psychiatry. 2015;5:387-96.
- [CrossRef] [PubMed] [Google Scholar]
- Zebrafish models to study drug abuse-related phenotypes. Rev Neurosci. 2011;22:95-105.
- [CrossRef] [PubMed] [Google Scholar]
- Zebrafish models for translational neuroscience research: From tank to bedside. Trends Neurosci. 2014;37:264-78.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of acute and chronic ethanol exposure on the behavior of adult zebrafish (Danio rerio) Pharmacol Biochem Behav. 2006;85:752-61.
- [CrossRef] [PubMed] [Google Scholar]
- Does acute alcohol exposure modulate aggressive behaviors in the zebrafish (Danio rerio), or is the bark worse than the bite? Int J Comp Psychol. 2010;23:32-9.
- [Google Scholar]
- Modeling withdrawal syndrome in zebrafish. Behav Brain Res. 2010;208:371-6.
- [CrossRef] [PubMed] [Google Scholar]
- Chronic and acute alcohol administration induced neurochemical changes in the brain: Comparison of distinct zebrafish populations. Am Acids. 2014;46:921-30.
- [CrossRef] [PubMed] [Google Scholar]
- Stress induces rapid changes in serotonergic activity: Restraint and exertion. Behav Brain Res. 2000;111:83-92.
- [CrossRef] [Google Scholar]
- Drinks like a fish: Zebra fish (Danio rerio) as a behavior genetic model to study alcohol effects. Pharmacol Biochem Behav. 2000;67:773-82.
- [CrossRef] [Google Scholar]
- Brain-derived neurotrophic factor (BDNF) in stress and affective disorders. Mol Psychiatry. 2002;7:519-9.
- [CrossRef] [PubMed] [Google Scholar]
- Identification, characterization, and ontogenic study of a catechol O-methyltransferase from zebrafish. Aquat Toxicol. 2011;102:18-23.
- [CrossRef] [PubMed] [Google Scholar]
- A review of monoaminergic neuropsychopharmacology in zebrafish. Zebrafish. 2010;7:359-78.
- [CrossRef] [PubMed] [Google Scholar]
- Regular care and maintenance of a zebrafish. Laboratory: An introduction. J Vis Exp. 2012;69:4196.
- [CrossRef] [PubMed] [Google Scholar]
- Gender differences in aggression and cortisol levels in zebrafish subjected to unpredictable chronic stress. Physiol Behav. 2016;171:50-4.
- [CrossRef] [PubMed] [Google Scholar]
- Guidelines for the Care and Use of Mammals in Neuroscience and Behavioral Research United States: National Academies Press; 2003.
- [Google Scholar]
- Acute and chronic alcohol dose: Population differences in behavior and neurochemistry of zebrafish. Genes Brain Behav. 2009;8:586-99.
- [CrossRef] [PubMed] [Google Scholar]
- A comparison of methodologies to test aggression in zebrafish. Zebrafish. 2015;12:144-51.
- [CrossRef] [PubMed] [Google Scholar]
- The imageJ ecosystem: An open platform for biomedical image analysis. Mol Reprod Dev. 2015;82:518-29.
- [CrossRef] [PubMed] [Google Scholar]
- AnimalTracker: An ImageJ-based tracking API to create a customized behaviour analyser program. Neuroinformatics. 2016;14:479-81.
- [CrossRef] [PubMed] [Google Scholar]
- A review of freely available, open-source software for the automated analysis of the behavior of adult zebrafish. Zebrafish. 2019;16:1662.
- [CrossRef] [PubMed] [Google Scholar]
- Effectiveness of recommended euthanasia methods in larval zebrafish (Danio rerio) J Am Assoc Lab Anim Sci. 2015;54:81-4.
- [Google Scholar]
- TRIzol™ Reagent Protocol, In Vitro Gen User Guide Waltham, MA: Thermo Fisher Scientific; 2016.
- [Google Scholar]
- SuperScript® III First-Strand Synthesis System for RT-PCR, In Vitro Gen Waltham, MA: Thermo Fisher Scientific; 2003.
- [Google Scholar]
- Tibolone attenuates inflammatory response by palmitic acid and preserves mitochondrial membrane potential in astrocytic cells through estrogen receptor beta. Mol Cell Endocrinol. 2019;486:65-78.
- [CrossRef] [PubMed] [Google Scholar]
- Characterization of housekeeping genes in zebrafish: Male-female differences and effects of tissue type, developmental stage and chemical treatment. BMC Mol Biol. 2008;9:102.
- [CrossRef] [PubMed] [Google Scholar]
- The utility of zebrafish to study the mechanisms by which ethanol affects social behavior and anxiety during early brain development. Prog Neuropsychopharmacol Biol Psychiatry. 2014;55:94-100.
- [CrossRef] [PubMed] [Google Scholar]
- Analyzing real-time PCR data by the comparative CT method. Nat Protoc. 2008;3:1101-8.
- [CrossRef] [PubMed] [Google Scholar]
- Official blame for drivers with very low blood alcohol content: There is no safe combination of drinking and driving. Inj Prev. 2015;21:e28-35.
- [CrossRef] [PubMed] [Google Scholar]
- Upregulating serotonin transporter expression and downregulating monoamine oxidase-A and indoleamine 2, 3-dioxygenase expression involved in the antidepressant effect of sodium valproate in a rat model. Neuroreport. 2014;25:1338-43.
- [CrossRef] [PubMed] [Google Scholar]
- The association of catechol-O-methyltransferase and interleukin 6 gene polymorphisms with posttraumatic stress disorder. Psychiatr Danub. 2019;31:241-8.
- [CrossRef] [PubMed] [Google Scholar]
- Estradiol replacement enhances fear memory formation, impairs extinction and reduces COMT expression levels in the hippocampus of ovariectomized female mice. Neurobiol Learn Memory. 2015;118:167-77.
- [CrossRef] [PubMed] [Google Scholar]
- Meta-analysis of six genes (BDNF, DRD1, DRD3, DRD4, GRIN2B and MAOA) involved in neuroplasticity and the risk for alcohol dependence. Drug Alcohol Depend. 2015;149:259-63.
- [CrossRef] [PubMed] [Google Scholar]
- Substance use and suicide risk in a sample of young Colombian adults: An exploration of psychosocial factors. Am J Addict. 2017;26:388-94.
- [CrossRef] [PubMed] [Google Scholar]
- The global burden of mental, neurological and substance use disorders: An analysis from the global burden of disease study 2010. PLoS One. 2015;10:e0116820.
- [CrossRef] [PubMed] [Google Scholar]
- Mood stabilizing drugs regulate transcription of immune, neuronal and metabolic pathway genes in Drosophila. Psychopharmacology. 2016;233:1751-62.
- [CrossRef] [PubMed] [Google Scholar]







