Translate this page into:
Effect of CD34+ cell dose on neutrophil and platelet engraftment kinetics in haematopoietic stem cell transplantation – A single-centre experience
*Corresponding author: Lydia Abraham, Department of Physiology, Christian Medical College, Ludhiana, Punjab, India. drlyds@yahoo.co.in
-
Received: ,
Accepted: ,
How to cite this article: Abraham L, Pramod J. Effect of CD34+ cell dose on neutrophil and platelet engraftment kinetics in haematopoietic stem cell transplantation – A single-centre experience. Indian J Physiol Pharmacol 2021;65:188-92.
Abstract
Objectives:
Haematopoietic stem cell transplantation (SCT) is curative for a number of benign and malignant hematological disorders. CD34 expression on haematopoietic progenitor cells is used to assess stem cell content in peripheral blood stem cell and bone marrow grafts. This study evaluated the relationship between numbers of CD34+ cells infused per kg and the timing of neutrophil and platelet engraftment.
Materials and Methods:
The effect of cell dose was studied in consecutive HSCT patients transplanted between November 2008 and December 2017. Neutrophil engraftment was defined as the first of 2 consecutive days with an absolute neutrophil count >0.5 × 109/L and platelet engraftment as unsupported platelet count >20 × 109/L for 7 days.
Results:
Of a total of 131 patients, 26 (19.8%) underwent an autologous SCT, while 105 (80.2%) underwent an allogeneic SCT. The median CD34 dose infused in the auto-SCT group was 5.29 × 106 CD34+cells/kg (IQR = 2.95–10.98) and 6.42 × 106 CD34+cells/kg (IQR = 4.20–9.20) in the allo-SCT group (P = 0.773). The median time to neutrophil engraftment in the auto-SCT group was 11 days (range 9.5–12) and in the allo-SCT group was 15 days (range 13–17), P ≤ 0.001. The median time to platelet engraftment in both groups was similar (12 days). When patients were divided into three groups based on CD34 dose (<5, 5–8 and >8), no difference was observed in the time to ANC or platelet engraftment. Similarly, no differences in time to engraftment were noted in each quartile of CD34 dosage in auto- and allo-SCT.
Conclusion:
Thus, it was concluded that a cell dose of approximately 5 × 106/kg provides reasonably rapid engraftment, with no advantage seen for a higher cell dose of >5.
Keywords
Haematopoietic stem cell transplantation
Stem cell transplantation
CD34
INTRODUCTION
Haematopoietic stem cell transplantation (HSCT) is curative for a number of benign and malignant hematological disorders.[1] CD34 expression on haematopoietic progenitor cells is used to assess stem cell content in peripheral blood stem cell and bone marrow grafts. It can be assumed therefore that higher CD34 expression would mean higher stem cell content and therefore better engraftment and outcomes. While it is true that an adequate number of CD34 expressing cells are important for better outcomes,[2] leading to the ‘more is better’ practice in many centres, studies have shown that a higher cell count could lead to an increase the incidence of graft-versus-host disease (GVHD).[3] Thus, there have been various studies that evaluate the optimal cell dose range for an HSCT.[4,5]
It is thus important to establish which cell dose is optimal for every HSCT centre as a reference point for treatment protocols. Through this study, we aimed to establish just that. From the inception of the transplant program in 2008 to the conclusion of this study in 2017, all transplant cases irrespective of indication or method were analyzed to determine a correlation between cell dose and engraftment outcomes.
MATERIALS AND METHODS
This was a retrospective study of the effect of cell dose which was studied in consecutive HSCT patients transplanted between November 2008 and December 2017. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee (Christian Medical College and Hospital, Ludhiana) and study protocol was approved by the same.
Informed consent was obtained from the participants before proceeding with the study. All patients were included irrespective of type of disease, whether benign or malignant. Indications for transplant included acute and chronic myeloid leukaemia, acute and chronic lymphoid leukaemia, thalassemia, aplastic anaemia, multiple myeloma, Hodgkin’s and non-Hodgkin’s lymphoma, myeloproliferative neoplasms, sickle cell anaemia and other hematological diseases.
Following HLA typing, conditioning regimens and GVHD prophylaxis, patients were posted for the procedure. Stem cells were harvested through bone marrow or peripheral blood and were analyzed by flow cytometry at our local stem cell laboratory before transplantation. Following transplant, the following end points were pre-determined. Neutrophil engraftment was defined as the first of 2 consecutive days with an absolute neutrophil count >0.5 × 109/L and platelet engraftment as unsupported platelet count >20 × 109/L for 7 days.
Frequencies, proportions, means and standard deviations were calculated. Chi-square and t-test were done using SPSS version 21.
RESULTS
Out of 131 patients, 94 (72%) were male and 37 (28%) were female. [Figure 1] shows the sex distribution of patients. [Figure 2] shows the age-wise distribution of the patients where the largest group consisted of the age group of 1–9 years and the smallest group was 40–49 years.

- Sex distribution.
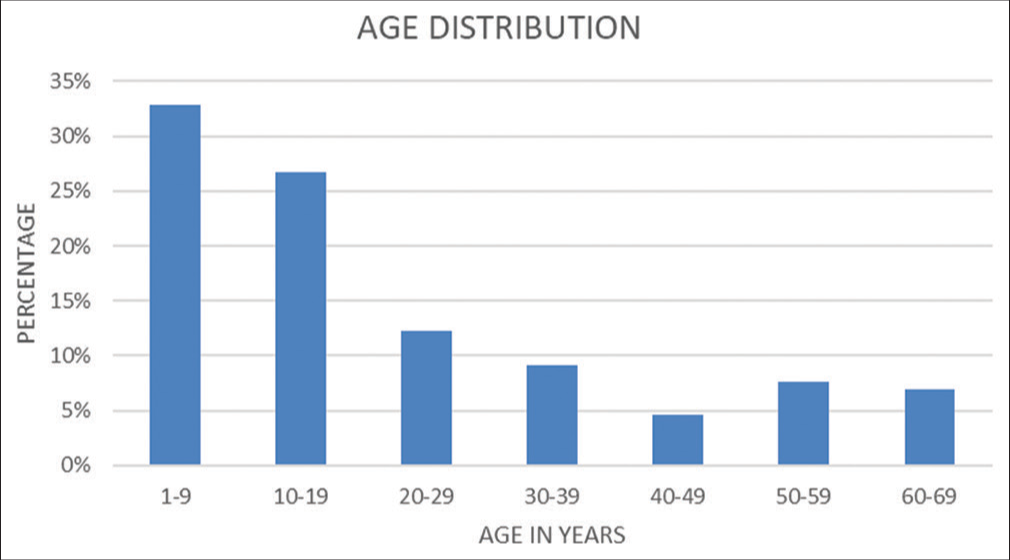
- Age distribution.
[Figure 3] shows the age and sex distribution of all the patients which shows that in all age groups, the majority were male except in the age group of 50–59 years which had equal number of males and females.
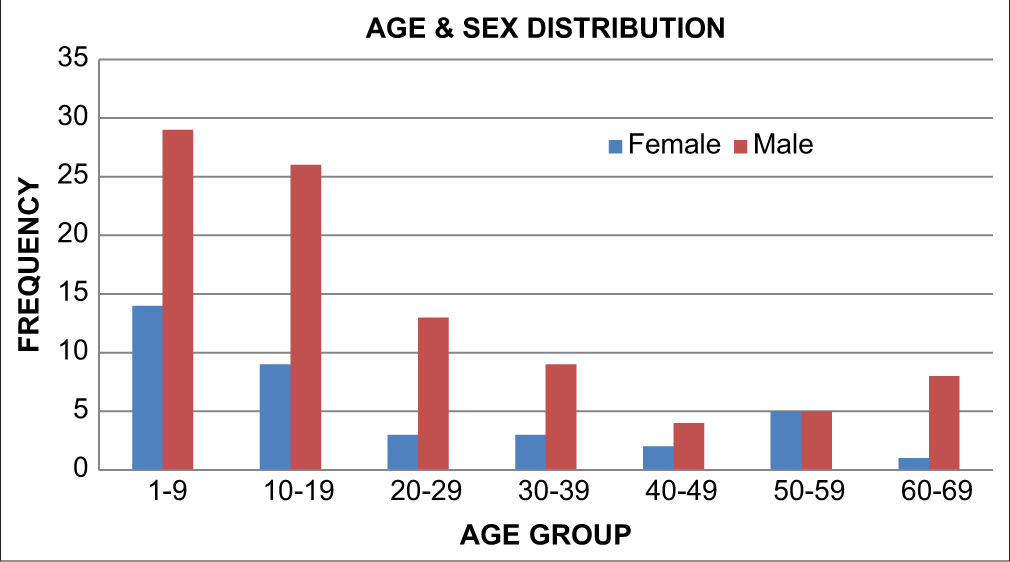
- Age and sex distribution.
Most patients received peripheral blood stem cells (92.4%) while 6.1% received bone marrow transplant and a small subset of patients (1.5%) received both [Figure 4] [BM = Bone marrow/PBSC = Peripheral blood stem cells]. In 131 patients, 26 (19.8%) underwent an autologous SCT, while 105 (80.2%) underwent an allogeneic SCT [Figure 5].
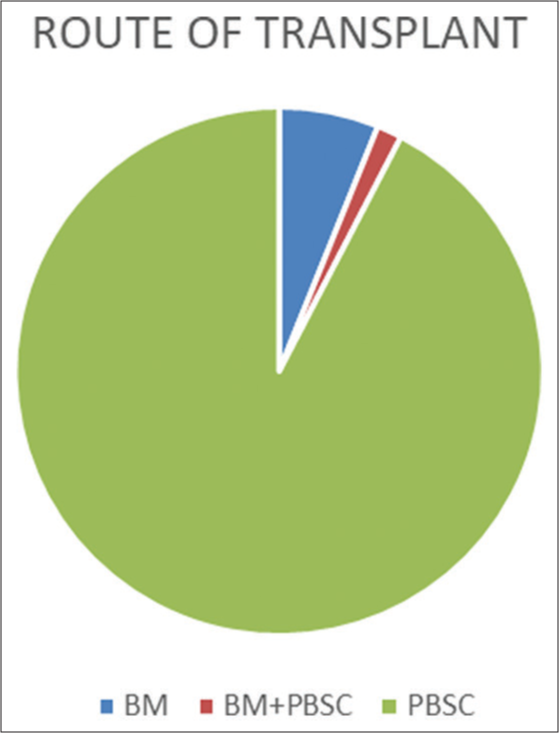
- Route of transplant
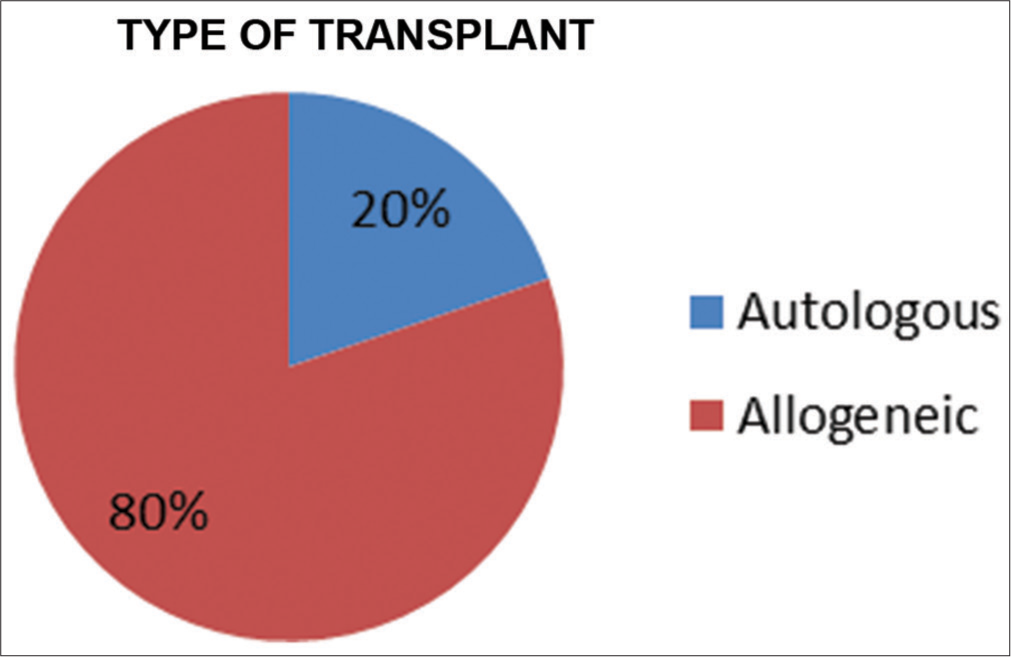
- Type of transplant
[Figure 6] shows the sex distribution among the two groups where there is a predominance of females in the autologous group but a predominance of males in the allogeneic group.
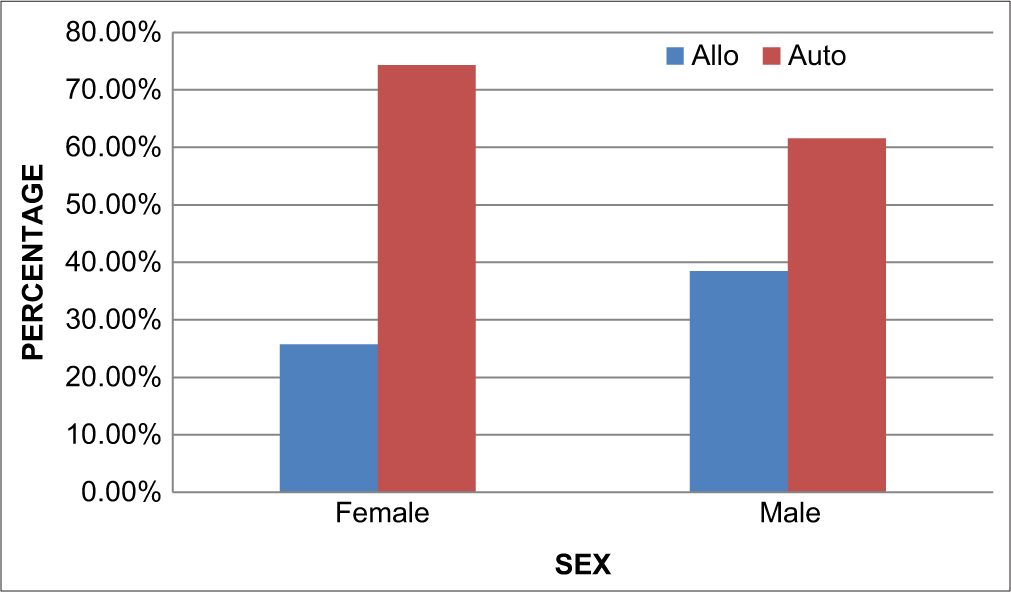
- Sex vs type of transplant
[Figure 7] shows the distribution of age groups between the two groups where the patients receiving allogeneic transplants were mainly in the younger age group of 1–9 years whereas the patients receiving autologous transplants were in the older age group of 50–59 years.
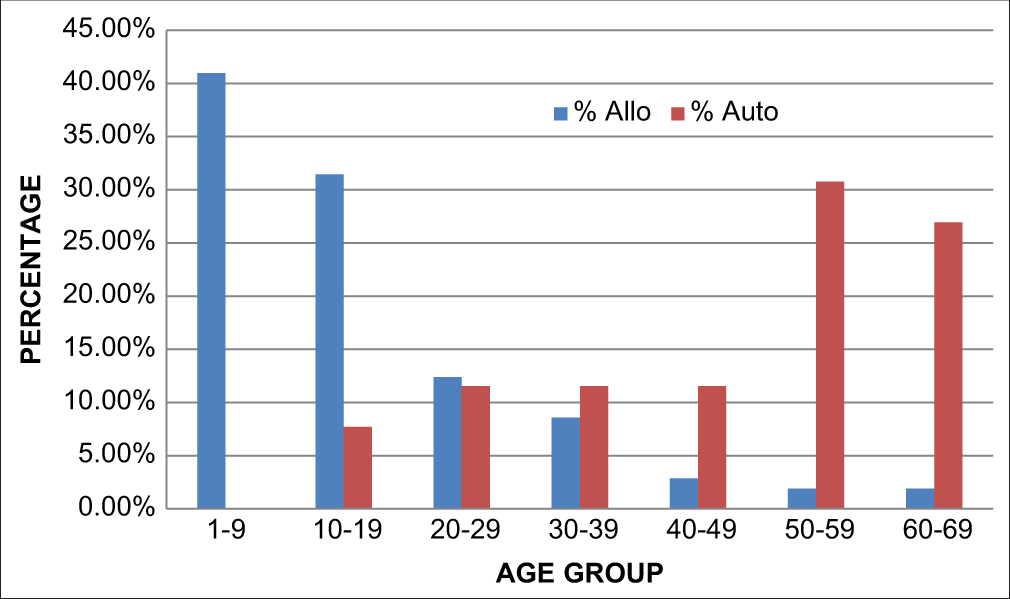
- Age vs type of transplant
Median CD34 dose infused in the auto-SCT group was 5.29 × 106 CD34+cells/kg (IQR = 2.95–10.98) and 6.42 × 106 CD34+cells/kg (IQR = 4.20–9.20) in the allo-SCT group (P = 0.773) [Figure 8].

- Cell dose vs type of transplant
The median time to neutrophil engraftment in the auto-SCT group was 11 days (range 9.5–12) and in the allo-SCT group was 15 days (range 13–17), P ≤ 0.001 [Figure 9]. The median time to platelet engraftment in both groups was similar (12 days).
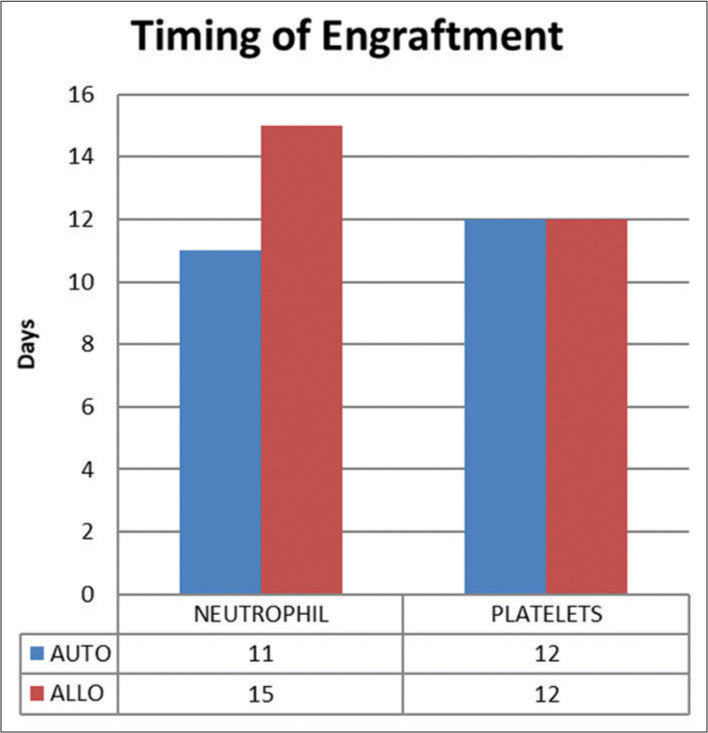
- Timing of engraftment
When patients were divided into three groups based on CD34 dose (<5, 5–8 and >8 × 106 CD34+cells/kg), no difference was observed in the time to ANC or platelet engraftment [Figure 10]. Similarly, no differences in time to engraftment were noted in each quartile of CD34 dosage in auto- and allo-SCT.
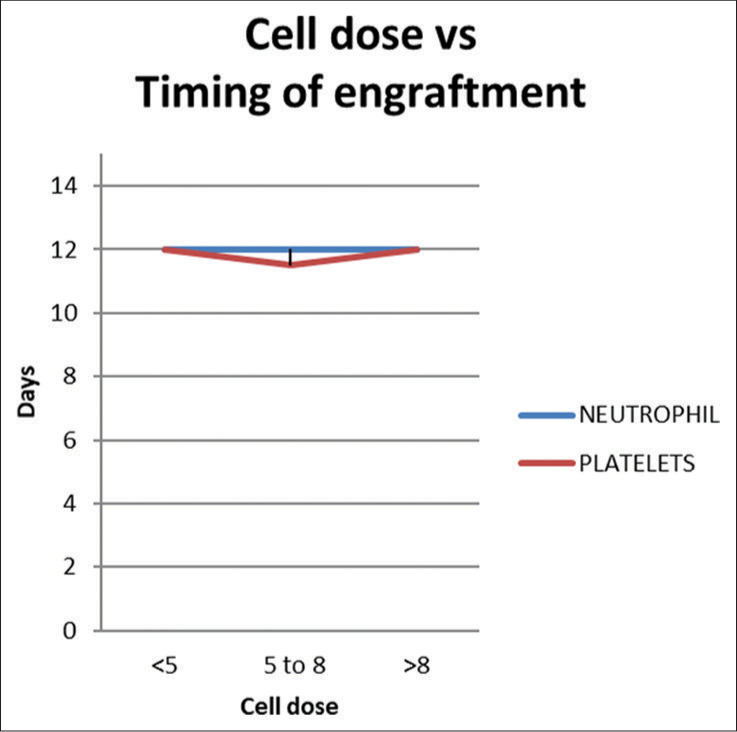
- Cell dose vs timing of engraftment
DISCUSSION
Stem cells are defined as undifferentiated cells capable of indefinite self-renewal and generation of a functional progeny of highly specialised cells. Stem cells have different properties and functions depending on their location. HSCs are characterised by the ability to self-renew and differentiate into all mature blood lineages.[6] The differentiation and proliferation of haematopoietic cells are regulated by various cellular mechanisms such as cytokines. The most primitive HSCs express the cell surface antigen CD34 and receptors for other growth factors.[7]
Different studies define different cell doses for stable engraftment. A nucleated cell dose of 5 × 106/kg is generally considered adequate, although cell doses of 3 × 106/kg can be used.[8] A study done in 2015 found that a higher CD34+ cell dose was associated with a faster neutrophil engraftment in bone marrow patients and faster platelet engraftment in peripheral blood stem cell recipients.[9] A poor or inadequate harvest would require a repeat procedure and further hospitalisation of the donor. A single-centre analysis done in Oslo which studied 189 PBSC recipients from sibling donors concluded that a CD34 cell dose of 6–7 × 106/kg showed better survival and lesser mortality while a dose of <5 × 106/kg had increased relapse but lower GVHD and higher than 6.5 × 106/kg had lower acute GVHD.[10] Another study done in 2015 done in 205 patients from a single institution concluded that there is no ideal cell dose at transplant within a range of 104–105.[11] Thus, it becomes very important to define the minimum cell dose required to give the best engraftment.
CONCLUSION
Our study showed that a cell dose of approximately 5 × 106/kg provides reasonably rapid engraftment, with no advantage seen for a higher cell dose of >5. However, engraftment is not purely a function of stem cell content in the graft and can be influenced by other factors including the underlying disease, pre-HSCT therapy, conditioning regimen, use of cytokines post-HSCT, graft quality and post-HSCT complications/events (e.g., GVHD, medications and infections). This study was limited in this wherein we included all HSCT cases irrespective of all other factors. Thus, further studies are required which explore these variables and define cell doses as per the case.
Data availability
The data that support the findings of this study are available on request from the corresponding author LA. The data are not publicly available due to institutional restrictions.
Acknowledgement
I thank Dr. Joseph John, Dr. Chepsy Philip, Dr. Suvir Singh, Mr. Sohan Singh and others of the Department of Haematology, Christian Medical College and Hospital, Ludhiana, for graciously opening their department to us and their ever-present expertise and assistance throughout all aspects of our study.
Declaration of patient consent
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Allogeneic and autologous transplantation for haematological diseases, solid tumours and immune disorders: Current practice in Europe 2009. Bone Marrow Transplant. 2010;45:219-34.
- [CrossRef] [PubMed] [Google Scholar]
- Marrow transplantation for treatment of aplastic anemia: An analysis of factors associated with graft rejection. N Engl J Med. 1977;296:61-6.
- [CrossRef] [PubMed] [Google Scholar]
- Higher mortality after allogeneic peripheral-blood transplantation compared with bone marrow in children and adolescents: The histocompatibility and alternate stem cell source working committee of the international bone marrow transplant registry. J Clin Oncol. 2004;22:4872-80.
- [CrossRef] [PubMed] [Google Scholar]
- Donor, recipient, and transplant characteristics as risk factors after unrelated donor PBSC transplantation: Beneficial effects of higher CD34+ cell dose. Blood. 2009;114:2606-16.
- [CrossRef] [PubMed] [Google Scholar]
- The number of infused CD34+ cells does not influence the incidence of GVHD or the outcome of allogeneic PBSC transplantation, using reduced-intensity conditioning and antithymocyte globulin. Bone Marrow Transplant. 2010;45:1189-96.
- [CrossRef] [PubMed] [Google Scholar]
- Hematopoietic stem cells: The paradigmatic tissue-specific stem cell. Am J Pathol. 2006;169:338-46.
- [CrossRef] [PubMed] [Google Scholar]
- A ligand-receptor signaling threshold model of stem cell differentiation control: A biologically conserved mechanism applicable to hematopoiesis. Blood. 2000;96:1215-22.
- [CrossRef] [PubMed] [Google Scholar]
- CD34+ cell dose predicts relapse and survival after T-cell-depleted HLA-identical haematopoietic stem cell transplantation (HSCT) for haematological malignancies. Br J Haematol. 2000;108:408-14.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of total nucleated and CD34+ cell dose on outcome after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2015;21:889-93.
- [CrossRef] [PubMed] [Google Scholar]
- The CD34+ cell dose matters in hematopoietic stem cell transplantation with peripheral blood stem cells from sibling donors. Clin Hematol Int. 2020;2:74-81.
- [CrossRef] [PubMed] [Google Scholar]
- T cell depleted allogeneic stem cell transplants for hematological malignancies: A 20 year experience shows no relationship between choice of transplanted T cell dose or delayed t cell add-back on major outcomes. Blood. 2013;126:1-2.
- [CrossRef] [Google Scholar]






