Translate this page into:
Amelioration of oestradiol valerate-induced endometrial hyperplasia in female rats by methanol fraction of Mangifera indica Linn. through modulation of oestrogen receptor signalling pathway
*Corresponding author: Adeola Oluwakemi Olowofolahan, Department of Biochemistry, University of Ibadan, Ibadan, Oyo State, Nigeria. mr_adeola@yahoo.com
-
Received: ,
Accepted: ,
How to cite this article: Olowofolahan AO, Tobih SE, Olorunsogo OO. Amelioration of oestradiol valerate-induced endometrial hyperplasia in female rats by methanol fraction of Mangifera indica Linn. through modulation of oestrogen receptor signalling pathway. Indian J Physiol Pharmacol 2021;65(2):94-102.
Abstract
Objectives:
Mangifera indica is a medicinal plant that is folklorically used in the treatment of certain disorders connected with women reproductive organs, especially, uterine fibroids. This study investigated the effect of methanol fraction of M. indica (MFMI) extract on oestradiol valerate (OV)-induced endometrial hyperplasia (EH).
Materials and Methods:
The animals were randomly divided into four groups of seven rats each. These include a control group, an MFMI-alone group, a model (OV-alone) group and MFMI treatment (OV+MFMI) group. The EH was induced by intraperitoneal injection of OV. The levels of oestrogen (E2), progesterone (PG) and total cholesterol (TC) were determined using ELISA technique. The uterine histological and immunohistochemical assessments of oestrogen receptor, β-catenin and Ki-67 were carried out. Fibroblast cell count/μm2 using histomorphometry as well as gas chromatography–mass spectrometry (GCMS) analysis of MFMI was carried out.
Results:
Severe EH was induced on oestradiol valerate administration. The MFMI was able to improve the pathological features of the animal model. Furthermore, the levels of oestrogen, PG and TC were reduced by MFMI. The immune reactive expression of oestrogen receptor alpha, β-catenin and Ki-67 was downregulated by MFMI coadministration. The histomorphometric analysis of the fibroblast cell count/μm2 showed increased cell count density in the OV-treated group which was significantly ameliorated by MFMI coadministration. The GC– MS analysis revealed the presence of some pharmacologically relevant phytochemicals.
Conclusion:
This study suggests that MFMI contains phytochemicals that can ameliorate OV-induced EH in female Wistar and the possible mechanism of action involves modulation of oestrogen signalling pathway.
Keywords
Oestradiol valerate
Endometrial hyperplasia
Oestrogen
Mangifera indica
INTRODUCTION
Endometrial proliferation is a normal phenomenon during the cycle.[1] A continuous exposure of the endometrium to oestrogen causes normal endometrial proliferation to progress to endometrial cancer.[2] The progression is attributed to unopposed oestrogenic stimulation.[3,4] Series of researches have been carried out to modify oestrogen exposure pharmacologically.[5-7] Studies have shown that oestrogen synthesis could be decreased by inhibiting the aromatase enzyme that is responsible for catalysing oestrogens biosynthesis from androgens.[8-10] There are natural products with medicinal properties which have the ability to inhibit aromatase enzyme (aromatase inhibitors) without deleterious traces.[11] Plants provide an extensive reservoir of natural products which are relevant in the chemoprevention and treatment of various diseases due to their availability and little or no side effect.[12] Mangifera indica is used in ethnomedicine for the treatment of uterine fibroid, urethritis, syphilis, urethrorrhoea, vaginopathy and haemorrhages from uterus.[13,14] One of the chemical constituents includes mangiferin which is a polyphenolic antioxidant and a glucosyl xanthone.[15,16] It has been shown to have anticarcinogenic effects.[14] Our previous study has shown that methanol extract of M. indica (MEMI) induces mitochondrial-mediated apoptosis (MMA) through induction of mPT pore opening and also ameliorates monosodium glutamate (MSG)-induced rat liver and uterine damage.[17] Bioactivity-guided assay in our study also revealed that the methanol fraction of M. indica (MFMI) is the most potent with respect to MMA through induction of mPT pore opening.[18] This study, therefore, investigated the effect of MFMI on oestradiol valerate (OV)-induced endometrial hyperplasia (EH) using female Wistar rats, looking at the oestrogen receptor signalling pathway.
MATERIALS AND METHODS
Chemicals and reagents
OV, including other reagents, was purchased from Sigma-Aldrich Chemical, USA. A stock of OV (30 mg OV in 10 ml of 5% DMSO corn oil) was prepared while, 3 mg/kg OV was intraperitoneally administered. The dose of OV was chosen based on our preliminary study and literature search.[19-21]
Plant material
M. indica leaves were purchased and authenticated at the Botany Department, University of Ibadan, Nigeria, with voucher number UIH 22555.
Preparation of extract
M. indica leaves were cut, washed, shade dried for 4 weeks and pulverised to powder. It was soaked in methanol, filtered and concentrated using vacuum rotary evaporator (N-100, Eyla, Tokyo, Japan).
Preparation of the fractions
The methanol extract was further partitioned between n-hexane, dichloromethane, ethyl acetate and methanol using vacuum liquid chromatography technique, and concentrated to dryness to obtain the fractions. The MFMI was, however, used for this study.[22] The chosen dose was based on preliminary investigation and previous publications.[18,19,21]
Experimental animals
Female Wistar rats (180–200 g), kept under standard conditions, were acclimatised for 2 weeks with free access to water and chow. Their oestrous cycle were monitored[23] and the experiment was conducted in accordance with the guidelines of National Institute of Health (NIH publication 85–23, 1985) for laboratory animal care and use.
Experimental design
Rats were randomly divided into four groups of seven rats each. Group (1) received corn oil while Group (2) was administered i.p. injection of 3 mg/kg of OV. Group (3) was given 50 mg/kg MFMI while Group (4) received MFMI (50 mg/kg) followed immediately by i.p. bolus of OV. The duration of study was 12 weeks. At the end of experiment, the rats were sacrificed by cervical dislocation. Blood was collected into EDTA bottles, centrifuged at 3000 g for 20 min and the plasma obtained was used for the analysis of E2, PG and TC using detection kits.
Histopathology
Uteruses were harvested and processed for histopathology using standard laboratory procedures.[24,25]
Determination of oestradiol (E2), progesterone (PG) and total cholesterol (TC) levels
The levels of E2, PG and TC were determined using E2, PG and TC enzyme-linked immunosorbent assay kits obtained from Abcam, Shanghai, China.
Immunohistochemical analysis of oestrogen receptor alpha (ERα), β-catenin and Ki-67
This was carried out using commercially available kits (Elabscience Biotechnology Ltd., Peoples Republic of China) following the manufacturer’s instruction. Primary antibodies used in this study were ERα, β-catenin and Ki-67 (1:200 dilution). Immunohistochemical staining was performed following the manufacturer’s instruction. The uterine sections were deparaffinised with xylene and rehydrated in ethanol series. The endogenous peroxidase activity was blocked by incubating sections in 0.3% hydrogen peroxide in methanol for ½ h. The sections were later incubated with primary antibodies at 4°C overnight. Slides were incubated with a biotinylated anti-rabbit link secondary antibody for 30 min. The uterine sections were then incubated with 3, 3-diaminobenzidine tetrahydrochloride for 10 min and counter stained with Mayer’s hematoxylin. Immune stained slides for each group were used for the measurement of area percentage of immunoexpression of ERα, β-catenin and Ki-67. This was performed in five non-overlapping fields from four different sections of seven different rats in each group at ×400. Positive signals for ERα, β-catenin and Ki-67 were represented by a brown granular mass and were quantified using ImageJ version 2.0. The positive staining intensity was calculated as the ratio of the stained area to the total field assessed.
Gas chromatography–mass spectrometry (GC–MS) analysis of MFMI
This was carried out following standard procedures. The MFMI was subjected to GC–MS analysis and the compounds obtained were identified on the basis of their retention time, peak area and by interpretation of mass spectra.
Statistics
One-way analysis of variance was used. Comparison between the control and the treated groups was performed using the Tukey’s test (P < 0.05).
RESULTS
The effect of MFMI on E2, PG and TC levels in normal and OV-treated rats is shown in [Figure 1]. The results show that OV administration elevated E2, PG and TC compared to the control (P < 0.01). However, cotreatment with MFMI significantly ameliorated these effects by reversing the increased oestrogen, PG and TC levels when compared to the model group (P < 0.01). The uterine section (using H and E) of the control group showed normal histological architecture as depicted in [Figure 2a]. In contrast severe pathological changes, which include poor architecture, EH of the endometrium epithelia layer (blue arrow), dilated and severely degenerated endometrial gland and inflammatory cells infiltrating the endometrial stroma (black arrow), were observed in the group treated with OV as revealed in [Figure 2b]. However, coadministration with MFMI caused improvement as the photomicrograph [Figure 2c] showed moderate architecture, normal endometrial layers and normal endometrial gland and no hyperplasia was observed. The uterine section of MFMI group as shown in [Figure 2d] also showed normal histological architecture. The photomicrographs of uterine sections stained with collagen Masson’s trichrome are shown in [Figures 3a-d]. The uterine section of the control group [Figure 3a] exhibited normal deposition of collagen fibre. [Figure 3b] (OV-treated rats) showed severe deposition of collagen fibres within the endometrial layers coupled with severe hyperplasia. Nevertheless, coadministration with MFMI [Figure 3c] reversed this effect and no hyperplasia was observed in the endometrium. As revealed in [Figure 3d], the uterine section of the group treated with MFMI alone also showed normal deposition of collagen fibre. [Figure 4] shows the result for the fibroblast cell count using histomorphometry. The results showed significant increase in the number of Masson’s trichrome-stained cells in the model group when compared to the control. However, on coadministration with MFMI, there was remarkable decrease in the cell count. There was a strong immunoreactive expression of ERα in the OV-treated group when compared to the control (P < 0.01) [Figure 5]. Nonetheless, this was dramatically reduced by coadministration with MFMI compared to the OV-treated group (P < 0.01). Furthermore, β-catenin was highly expressed in the uterine section of the OV-treated rats compared to the control (P < 0.01) [Figure 6]. In contrast, this effect was drastically reduced by coadministration with MFMI when compared to the OV-treated rats (P < 0.01). [Figure 7] depicts the photomicrographs of the rat uterine sections immune stained with Ki-67. As shown from the results, there was marked expression of Ki-67-positive cells in the OV-treated rats relative to control (P < 0.001). Nevertheless, coadministration with MFMI caused significant reduction in the intensity and distribution of the Ki-67-positive cells compared to the OV-treated group (P < 0.01).

- MFMI ameliorated oestradiol valerate (OV)-induced increase in oestrogen (a), progesterone (b) and total cholesterol (c) levels. Data are presented as mean ± SEM. AP<0.01 compared with the control group; BP<0.01 compared with the OV group.
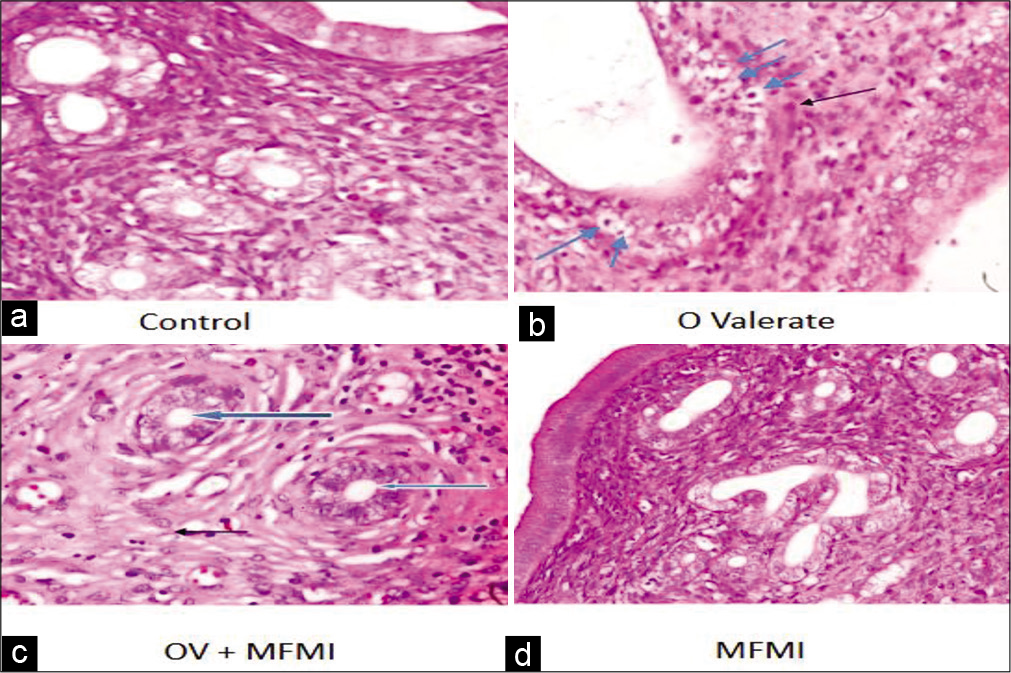
- (a-d) Photomicrographs of the rat uterine section for histological assessment stained by haematoxylin and eosin (×400). Control group and MFMI treatment alone group show normal proliferative endometrium and myometrium. However, severe endometrial hyperplasia (blue arrow) and presence of inflammatory cells infiltrating the endometrial stroma (black arrow) were observed in the oestradiol valerate treatment group. The pathology of the endometrial gland of the model rats improved on treatment with MFMI.
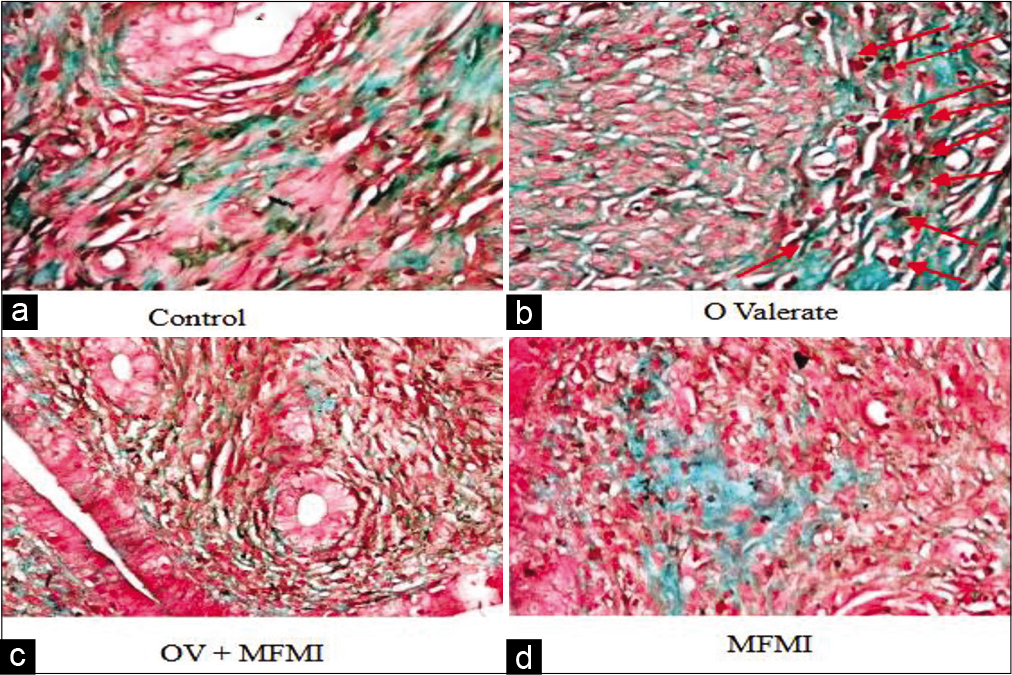
- (a-d) Photomicrographs of uterine section for collagen assessment stained by Masson’s trichrome (×400). The uterine section of control group and MFMI treatment only group shows normal deposition of collagen fibre. The uterine section of oestradiol valerate-treated rats shows severe deposition of collagen fibres within the endometrial layers as well as severe hyperplasia (red arrow). This was reversed on treatment with MFMI. The portion stained red is smooth muscle while the portion stained green is the collagen fibre.
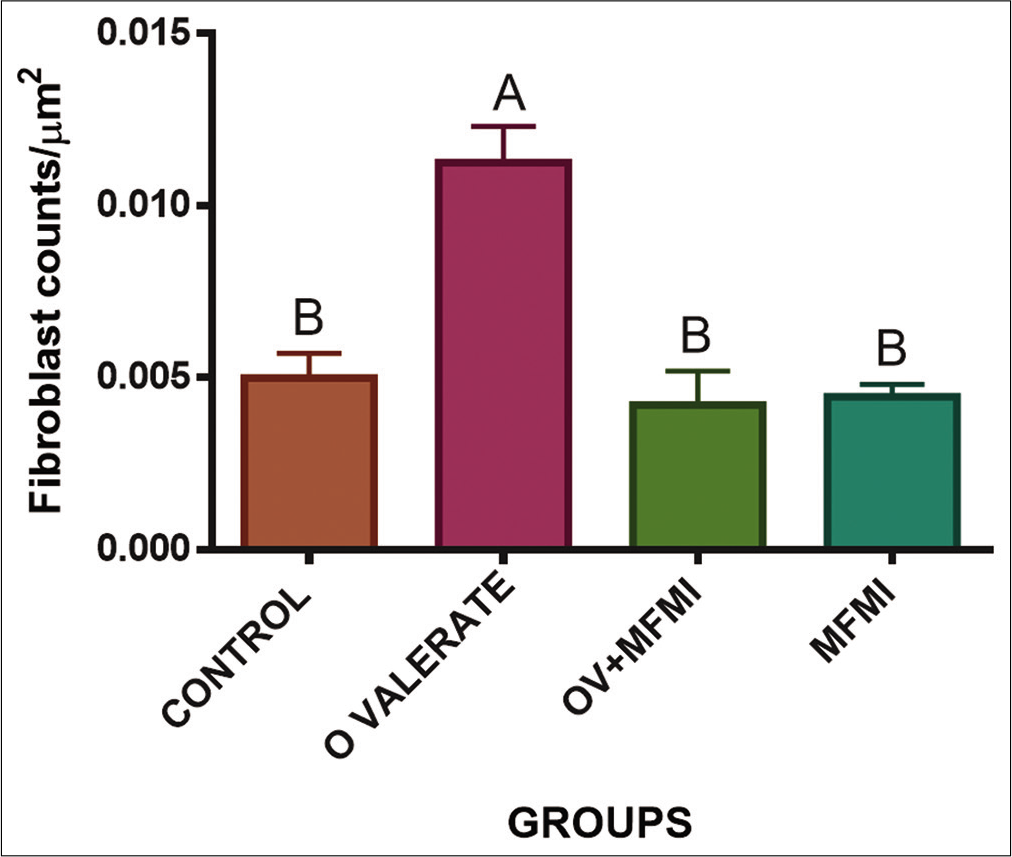
- MFMI reversed oestradiol valerate (OV)-induced fibroblast cell counts increase. Data are presented as mean ± SEM.AP<0.01 compared with the control group; BP<0.01 compared with the OV group.
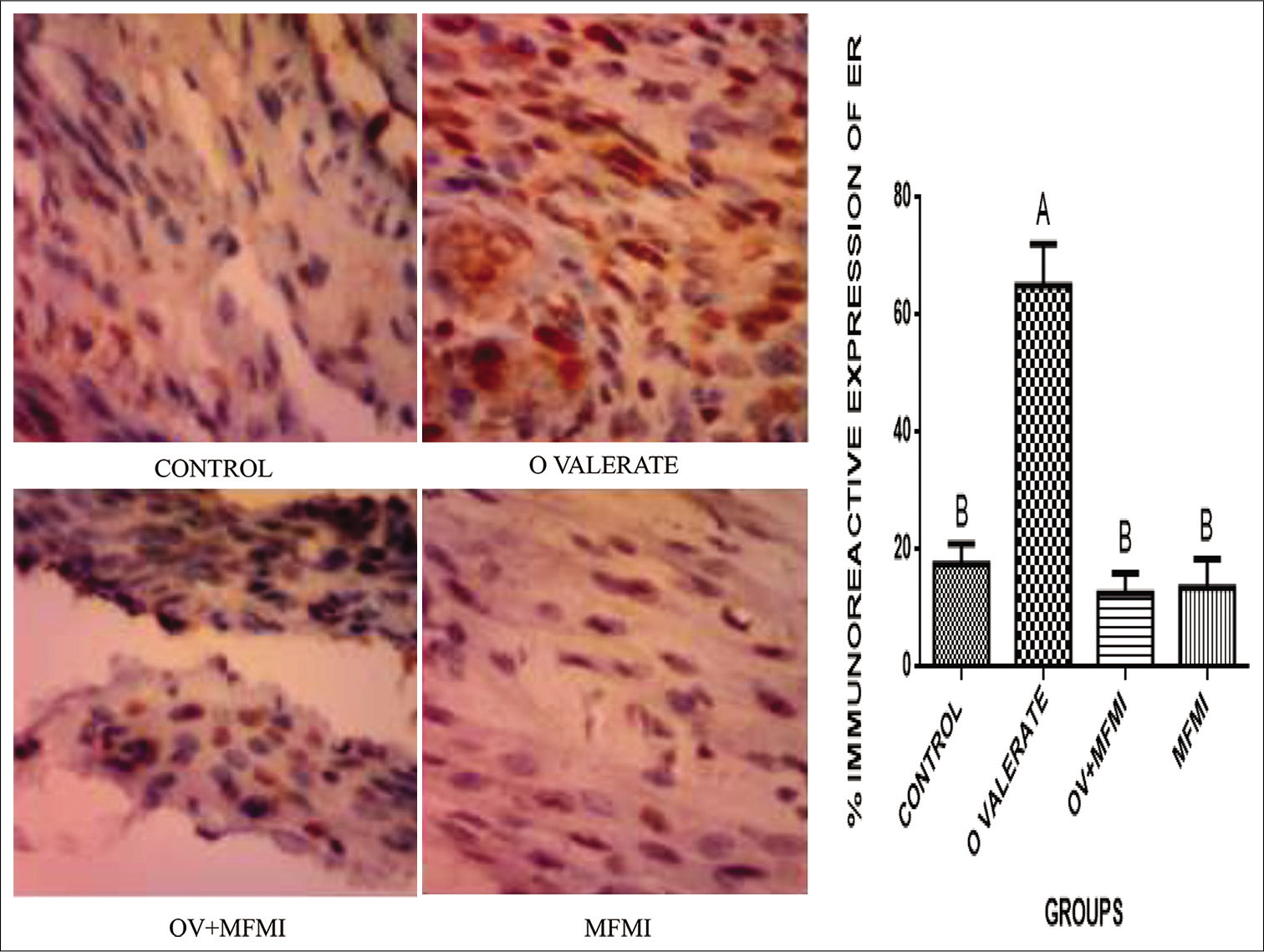
- MFMI downregulated oestradiol valerate (OV)-induced immunoreactive expression of nuclear ERα (×400). Data are presented as mean ± SEM. AP<0.0 1 compared with the control group; BP<0.01 compared with the OV group.
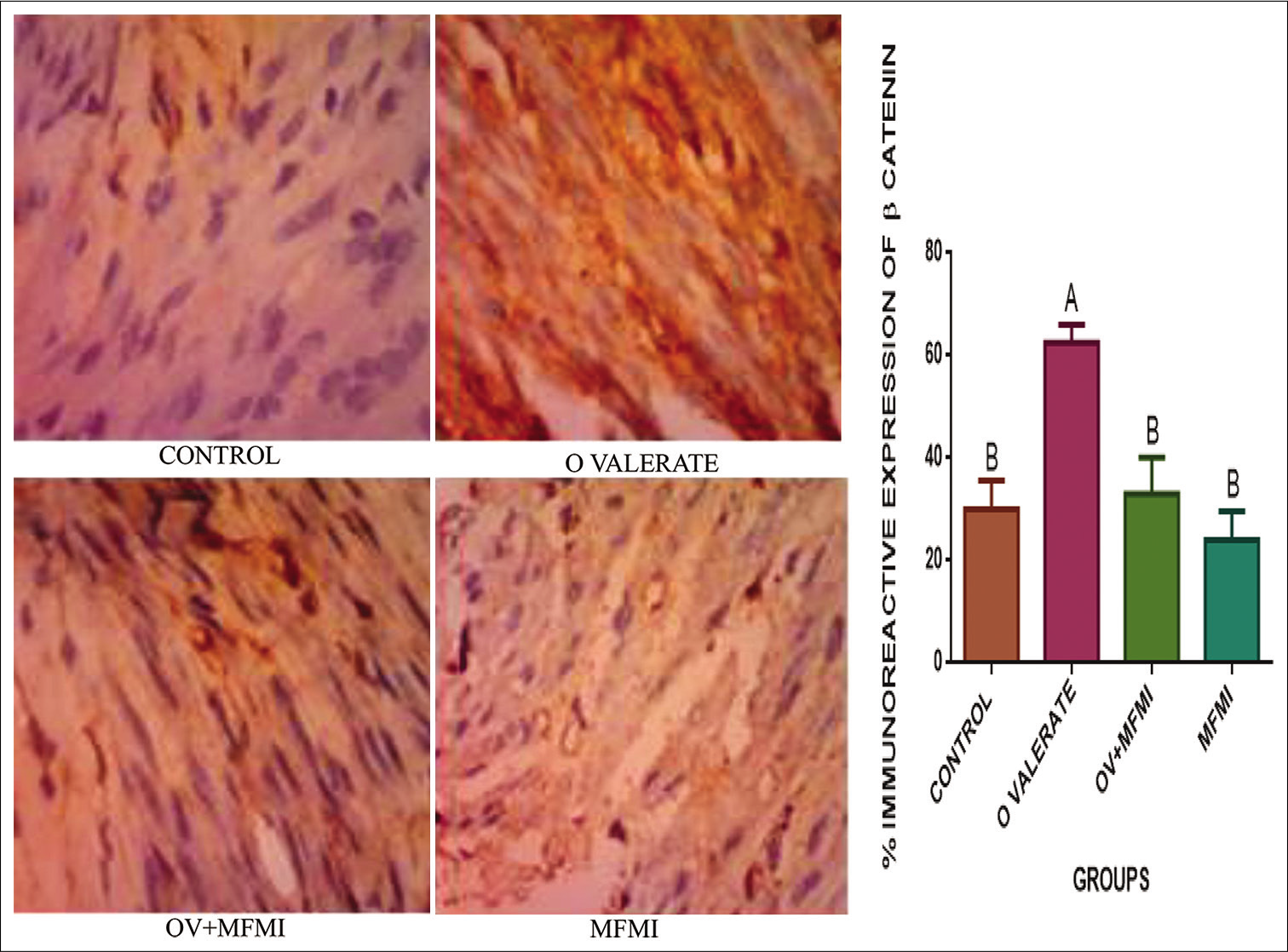
- MFMI ameliorated oestradiol valerate (OV)-induced immunoreactive expression of β-catenin (×400). Data are presented as mean ± SEM. AP<0.01 compared with the control group; BP<0.01 compared with the OV group.
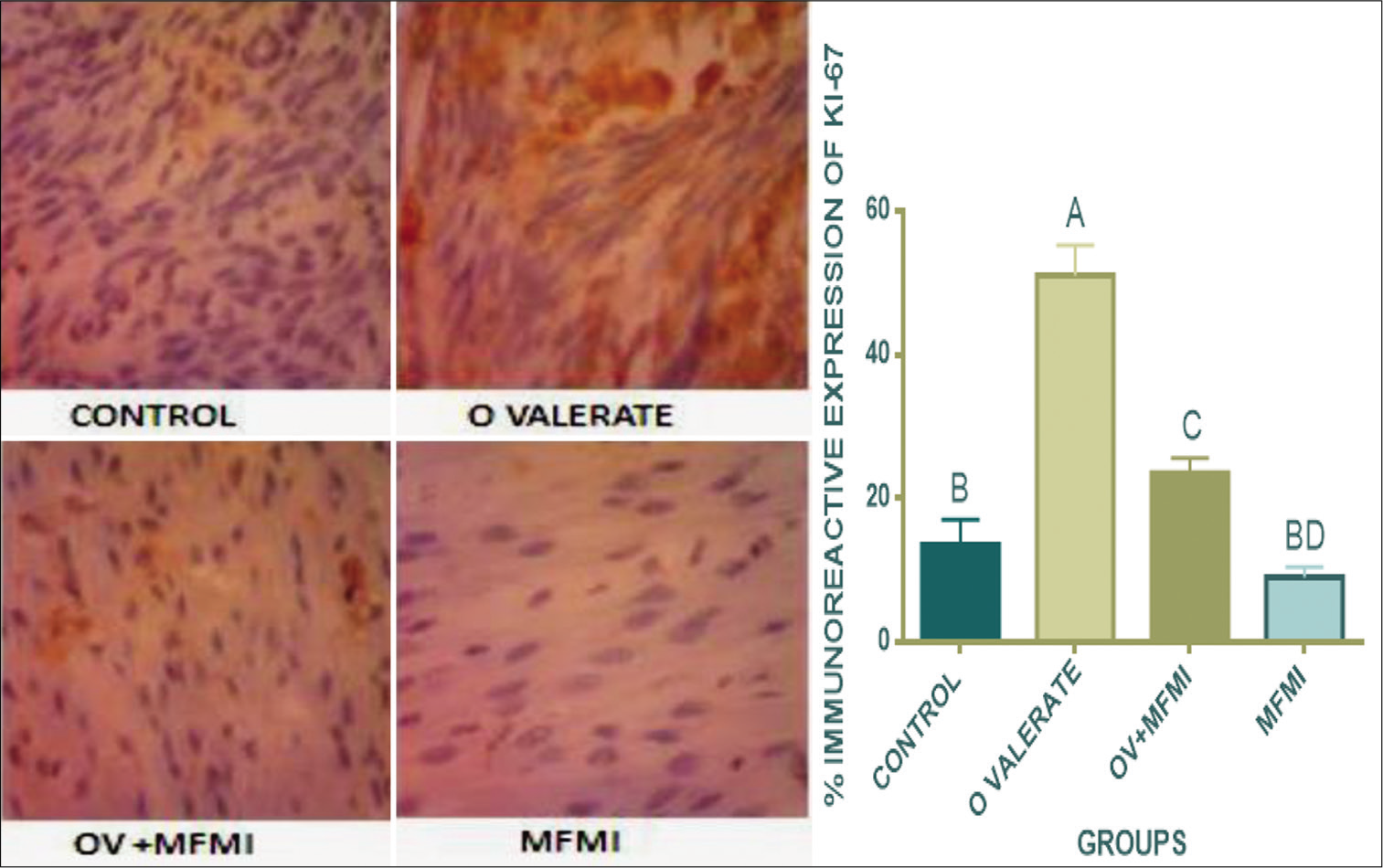
- MFMI reversed oestradiol valerate (OV)-induced immunoreactive expression of Ki-67 (×400). Data are presented as mean ± SEM.AP<0.001 compared with the control group; BP<0.001, CP<0.01 compared with the OV group; DP<0.05 compared with the OV+MFMI group.
The GC–MS analysis of the MFMI revealed the presence of some phytochemicals which include n-hexadecanoic acid, hexadecanoic acid ethyl ester, dodecanoic acid 2,10-dimethylmethyl ester, 2-Methyl-Z,Z-3,13-octadecadienol, 9-Octadecenoic acid methyl ester, 11-Octadecenoic acid methyl ester, 7-Octadecenoic acid, methyl ester, Butyl 9-octadecenoate and 11-Octadecenoic acid methyl ester.[Figure 8] shows the chromatogram of MFMI while [Table 1] shows the GC–MS profile of some of the major identified compounds.
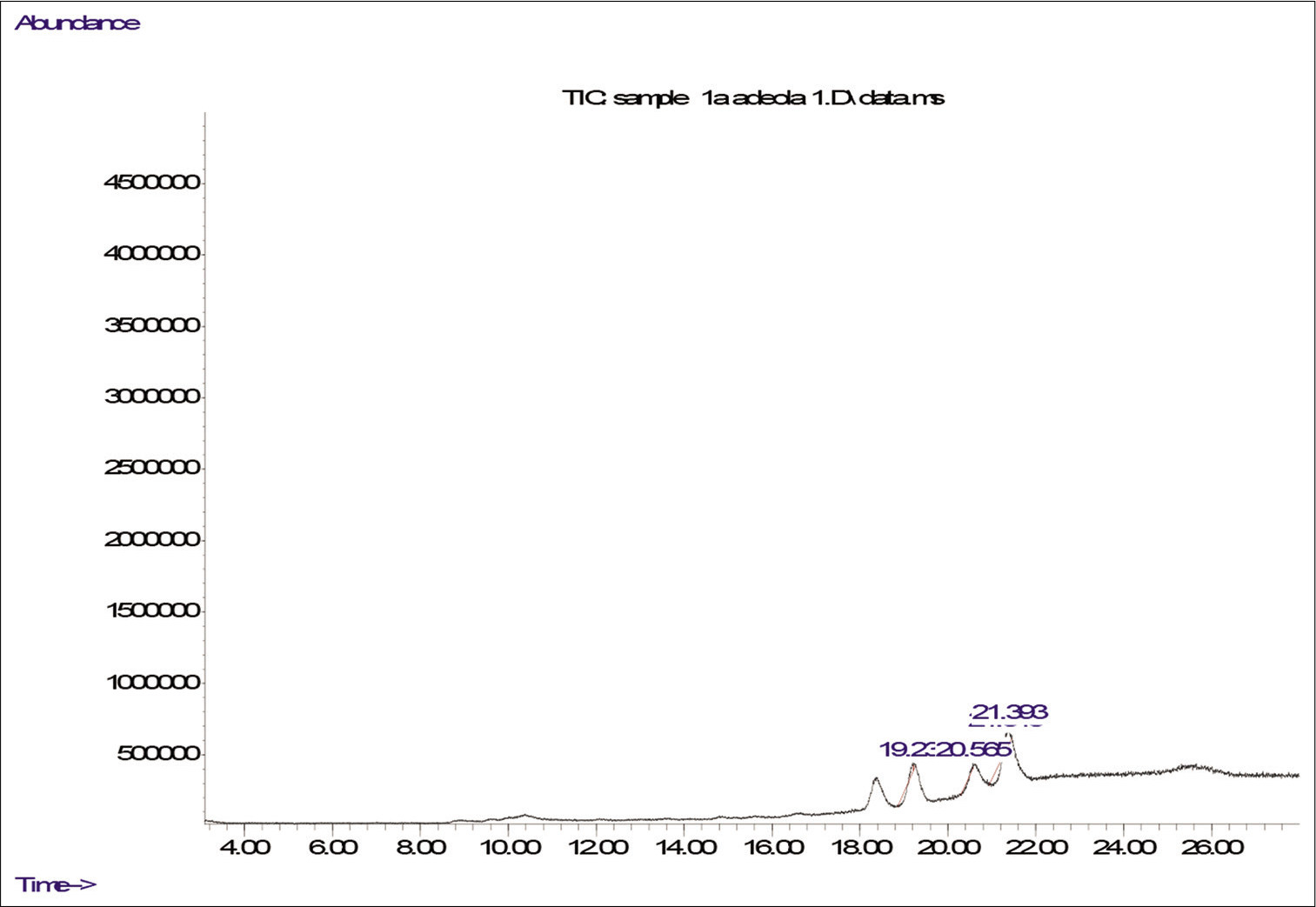
- The gas chromatography–mass spectrometry of MFMI.
| S. No. | Retention time | Compound | % peak area |
|---|---|---|---|
| 1. | 19.206 | n-Hexadecanoic acid | 6.326 |
| 2. | 19.234 | Hexadecanoic acid, ethyl ester | 18.485 |
| 3. | 20.568 | 9-Octadecenoic acid ethyl ester | 33.137 |
| 4. | 21.311 | 11-Octadecenoic acid, methyl | 5.311 |
| 5. | 21.392 | 2-Methyl-Z,Z-3,13-octadecadienol | 23.462 |
GC–MS: Gas chromatography–mass spectrometry, MFMI: Methanol fraction of Mangifera indica
DISCUSSION
The pathogenesis of EH remains unclear, but experimental evidences suggest that it could be attributed to endocrine disorder.[26] In this study, OV was used to induce EH in female rat while the effect of MFMI was investigated on the OV-treated female rats. As revealed in this study, there was a significant increase in the level of oestrogen in the group that received OV when compared to the control. This increase could be due to long-term administration of OV. According to Prasad et al.,[27] the increase in oestrogen level could lead to oxidative stress injury by causing increase in reactive oxygen species level and thereby enhance cellular proliferation. However, coadministration with MFMI caused a remarkable decrease in the level of oestrogen. This probably indicates that MFMI contains certain phytochemicals capable of reducing the elevated oestrogen level perhaps by interacting with some proteins involved in oestrogen signalling pathway. According to Shaban et al.,[28] aromatase activity was decreased by varying concentrations of M. indica peel extract. It could, therefore, be suggested that MFMI probably contains certain phytochemicals that are inhibitors of aromatase enzyme and thereby reduce the circulating level of oestrogen. Similar trend was observed with PG and TC levels which were also significantly elevated in OV-treated group. The increase in PG level could probably be due to increased levels of luteinising hormone while the increase in TC suggests that OV treatment could cause lipid metabolism disorder when administered over a long period of time in rats. However, these effects were reversed in the group that received coadministration with MFMI. From the histological study, it could be inferred that OV caused severe histopathological disorder resulting to the development of EH. However, coadministration with MFMI reversed this anomaly. No hyperplasia was observed in the group coadministered with MFMI, and also, the endometrial layers with the glands appeared normal. Although, there was still infiltration of the inflammatory cells not as severe as the OV-treated rats. The increase in fibroblast cell count/μm2 in the OV-treated group could be linked to increase in oestrogen level which can ultimately lead to the cellular multiplication.[28,29] Nevertheless, cotreatment with MFMI significantly ameliorated the OV-induced pathology. The findings from histological study and histochemical analysis suggest that MFMI is enriched with phytochemicals that possess ameliorative potential against OV-induced uterine damage and EH. This is consistent with a previous study which demonstrated the ameliorative potential of chloroform fraction of methanol extract of Drymaria cordata on MSG-induced uterine hyperplasia. [30] The immunohistochemical study showed that in the OV-treated group, there was increased immunoexpression of ERα, β-catenin and Ki-67 which was decreased on MFMI coadministration. This also suggests the presence of certain bioactive agents in MFMI that can interfere with the oestrogen signalling pathway and possibly downregulate the expression of oestrogen receptor. Dysregulated β-catenin signalling has been implicated in neoplastic transformation. [31,32] Experimental evidences have shown that there could be an interaction between steroid hormones-activated β-catenin to stimulate tumorigenesis.[33] The strong immunoreactive expression of β-catenin in the uterine section of OV-treated rats could be as a result of OV-induced oestrogenic stimulation, which subsequently led to dysregulated β-catenin signalling. However, this effect was remarkably reversed by MFMI. This is similar to the findings of Ayman et al.[34] who demonstrated the potency of Vitamin D3 in reducing β-catenin expression. The Ki-67 could be used to assess proliferative activity.[35] In the OV-treated group, there was strong uterine Ki-67 expression which was reversed by MFMI coadministration. This also suggests that MFMI is enriched with phytochemicals that have anti-proliferative potential. This is in agreement with Ali et al.[29] who demonstrated the anti-proliferative effect of hemin in oestrogen-induced EH through suppression of Ki-67 protein. Interestingly, the GC–MS analysis of MFMI revealed the presence of some phytochemicals that could possibly contribute toward the pharmacological effects shown in this study. The n-hexadecanoic acid has been shown to have antifibrinolytic, antifungal and antioxidant properties.[36] Its antiandrogenic, nematicide, haemolytic, hypocholesterolaemic, pesticide, 5-α reductase inhibitor and anti-inflammatory properties have been documented.[37,38] Furthermore, hexadecanoic acid, methyl ester has been reported to cause growth inhibition and apoptosis induction in human gastric cancer cells.[39] Its antioxidant, anti-inflammatory and antiproliferative potentials have been documented.[21,36,40] Its hypocholesterolaemic, nematicide, pesticide, lubricant, antiandrogenic, flavour and haemolytic 5-α reductase inhibitor properties have also been reported. [41,42] The antioxidant and anti-inflammatory properties of 9-Octadecenoic acid ethyl ester have been reported.[43] A terpenoid, 2-Methyl-Z,Z-3,13-octadecadienol identified from MFMI has several biological and pharmacological activities and it is being used as a pesticide, herbicide and pheromone. [44] Several terpenoids are biologically active and are exploited in the fight against cancer, malaria, inflammation and a variety of infectious diseases. Other identified important phytochemicals of proven biological activities are 11-Octadecenoic acid, methyl ester and 7-Octadecenoic acid, methyl ester.
CONCLUSION
Based on the findings in this study, it could be suggested that MFMI contains bioactive compound(s) which possess antiproliferative potential against oestrogen-mediated EH. Furthermore, possible mechanisms of action could be modulation of oestrogen signalling, downregulation of β-catenin signalling and suppression of Ki-67 protein. In addition, the identified phytochemicals in MFMI suggest its antioxidant and anti-inflammatory potentials.
Authors’ contributions
This work was carried out in collaboration between all authors. A. O. Olowofolahan designed the study, wrote the protocol, managed the analysis of the study, performed the statistical analysis and wrote the first draft of the manuscript. Material preparation and data collection were performed by Shalom E. Tobih and A. O. Olowofolahan while O. O. Olorunsogo approved the study and read through the manuscript. All authors read and approved the final manuscript.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- The mechanism of menstruation In: Santoro NF, Neal-Perry G, eds. Amenorrhea. Contemporary Endocrinology. Totowa: Humana Press; 2010. p. :21-34.
- [CrossRef] [Google Scholar]
- Endometrial hyperplasia and related cellular changes. In: Kurman R, ed. Blaustein's Pathology of the Female Genital Tract. New York: Springer; 1994. p. :411-37.
- [CrossRef] [Google Scholar]
- Endometrial cancer: A review and current management strategies: Part I. Gynecol Oncol. 2014;134:385-92.
- [Google Scholar]
- Mitochondrial changes in endometrial carcinoma: Possible role in tumor diagnosis and prognosis (review) Oncol Rep. 2015;33:1011-8.
- [CrossRef] [Google Scholar]
- Mitochondrial benzodiazepine receptors mediate cardioprotection of estrogen against ischemic ventricular fibrillation. Pharmacol Res. 2009;60:61-7.
- [CrossRef] [Google Scholar]
- Hormone receptor status, tumor characteristics, and prognosis: A prospective cohort of breast cancer patients. Breast Cancer Res. 2007;9:R6.
- [CrossRef] [Google Scholar]
- Understanding the pathological manifestations of aromatase excess syndrome: Lessons for clinical diagnosis. Expert Rev Endocrinol Metab. 2014;9:397-409.
- [CrossRef] [Google Scholar]
- Estrogen-related receptor gamma promotes mesenchymal-to-epithelial transition and suppresses breast tumor growth. Cancer Res. 2011;71:2518-28.
- [CrossRef] [Google Scholar]
- Isolation and Characterization of Aromatase Inhibitors from Brassaiopsis glomerulata (Araliaceae) Nat Rev Cancer. 2003;3:821.
- [CrossRef] [Google Scholar]
- Effects of diverse dietary phytoestrogens on cell growth, cell cycle and apoptosis in estrogen-receptor-positive breast cancer cells. J Nutr Biochem. 2010;21:856-64.
- [CrossRef] [Google Scholar]
- Natural products as aromatase inhibitors. Anticancer Agents Med Chem. 2008;8:646-82.
- [CrossRef] [Google Scholar]
- Medicinal plants of India with anti-diabetic potential. J Ethnopharmacol. 2002;81:81-100.
- [CrossRef] [Google Scholar]
- Pharmacological activities of mango (Mangifera indica): A review. J Pharmacogn Phytochem. 2016;5:1-7.
- [Google Scholar]
- Anticarcinogenic effects of polyphenolics from mango (Mangifera indica) varieties. J Agric Food Chem. 2010;58:4104-12.
- [CrossRef] [Google Scholar]
- Characterization and quantitation of polyphenolic compounds in bark, kernel, leaves, and peel of mango (Mangifera indica L) J Agric Food Chem. 2008;56:5599-610.
- [CrossRef] [Google Scholar]
- Mango (Mangifera indica L) peel extracts inhibit proliferation of HeLa human cervical carcinoma cell via induction of apoptosis. J Korean Soc Appl Biol Chem. 2012;55:397-405.
- [CrossRef] [Google Scholar]
- Effect of methanol extract of Mangifera indica on mitochondrial membrane permeability transition pore in normal rat liver and monosodium glutamate-induced liver and uterine damage. J Complement Altern Med Res. 2018;5:1-14.
- [CrossRef] [Google Scholar]
- 3-(Parafluorobenzoyl)-propionic acid; a metabolite of haloperidol, reversed oestradiol valerate-induced uterine hyperplasia via modulation of oestrogen receptor signalling pathways in female Wistar rats. J Basic Clin Physiol Pharmacol. 2020;31:4.
- [CrossRef] [Google Scholar]
- Anti-inflammatory and antifibrotic effects of methyl palmitate. Toxicol Appl Pharmacol. 2011;254:238-44.
- [CrossRef] [Google Scholar]
- Mechanisms underlying the protective effect of montelukast in prevention of endometrial hyperplasia in female rats. nt Immunopharmacol. 2018;62:326-33.
- [CrossRef] [Google Scholar]
- Methyl palmitate reversed estradiol benzoate-induced endometrial hyperplasia in female rats. Toxicol Mech Methods. 2021;31:43-52.
- [CrossRef] [Google Scholar]
- Induction of mitochondrial membrane permeability transition pore opening and DNA fragmentation by certain solvent fractions of Mangifera indica. Arch Basic Appl Med. 2019;7:123-9.
- [Google Scholar]
- Effect of Rumex steudelii methanolic root extract on ovarian folliculogenesis and uterine histology in female albino rats. Afr Health Sci. 2010;10:353-61.
- [Google Scholar]
- Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology (3rd ed). New York: McGraw-Hill; 1968.
- [Google Scholar]
- Some histological methods: Trichrome staining and their preliminary technique. J Tech Methods. 1929;12:75-90.
- [Google Scholar]
- Endocrine therapy toxicity: Management options. Am Soc Clin Oncol Educ Book. 2014;34:25-30.
- [CrossRef] [Google Scholar]
- Reactive oxygen species (ROS) and cancer: Role of antioxidative nutraceuticals. Cancer Lett. 2017;387:95-105.
- [CrossRef] [Google Scholar]
- Potential effect of Olea europaea leaves, Sonchus oleraceus leaves and Mangifera indica peel extracts on aromatase activity in human placental microsomes and CYP19A1 expression in MCF-7 cell line: Comparative study. Cell Mol Biol (Noisy-le-grand). 2009;62:11-9.
- [Google Scholar]
- Amelioration of estrogen-induced endometrial hyperplasia in female rats by hemin via heme-oxygenase-1 expression, suppression of iNOS, p38 MAPK and Ki67. Can J Physiol Pharmacol. 2019;97:1159-68.
- [CrossRef] [Google Scholar]
- Induction of mitochondrial membrane permeability transition pore opening and cytochrome c release by fractions of Drymaria cordata. Arch Bas App Med. 2015;3:135-44.
- [Google Scholar]
- Genetic studies of MIS signalling in sexual development. Novartis Found Symp. 2002;244:157-64. discussion 164-8, 203-6, 253-7
- [Google Scholar]
- Conditional deletion of beta-catenin in the mesenchyme of the developing mouse uterus results in a switch to adipogenesis in the myometrium. Dev Biol. 2005;288:276-83.
- [CrossRef] [Google Scholar]
- Constitutive activation of beta-catenin in uterine stroma and smooth muscle leads to the development of mesenchymal tumors in mice. Biol Reprod. 2009;81:545-52.
- [CrossRef] [Google Scholar]
- Vitamin D3 inhibits Wnt/β-catenin and mTOR signaling pathways in human uterine fibroid cells. J Clin Endocrinol Metab. 2016;101:1542-51.
- [CrossRef] [Google Scholar]
- Phytochemical and Ethnobotanical Databases; U.S. Department of Agriculture, Agricultural Research Service 1992-2016. 2016. Available form: https://www.phytochem.nal.usda.gov
- [Google Scholar]
- GC-MS analysis of phytocomponents in the methanolic extract of justicia wynaadensis (NEES) T Anders. Int J Pharma Bio Sci. 2012;3:570-6.
- [Google Scholar]
- Anti-inflammatory activity of methyl palmitate and ethyl palmitate in different experimental rat models. Toxicol Appl Pharmacol. 2012;264:84-93.
- [CrossRef] [Google Scholar]
- Isolation and characterization of methyl esters and derivatives from Euphorbia kansui (Euphorbiaceae) and their inhibitory effects on the human SGC-7901 cells. J Pharm Pharm Sci. 2005;3:528-35.
- [Google Scholar]
- Anti-inflammatory activity of methyl palmitate and ethyl palmitate in different experimental rat models. Toxicol Appl Pharmacol. 2012;264:84-93.
- [CrossRef] [Google Scholar]
- Phytochemical analyses of eight plants from two provinces of Ecuador by GCMS. Int J Herb Med. 2020;8:10-20.
- [Google Scholar]
- GC-MS analysis of bioactive components of Hugonia mystax L (Linaceae) Res J Pharm Biol Chem Sci. 2012;3:301-8.
- [Google Scholar]
- GC-MS analysis of ethanol extract of Cyperus rotundus leaves. Int J Curr Biotechnol. 2014;2:19-23.
- [Google Scholar]
- The antimicrobial properties of Chile peppers (Capsicum species) and their uses in Mayan medicine. J Ethnopharmacol. 1996;52:61-70.
- [CrossRef] [Google Scholar]






