Translate this page into:
An electrophysiological comparison of freshly isolated caprine articular chondrocytes versus cryopreserved chondrocytes
*Corresponding author: Dr. Upasana Kachroo, Department of Physiology, Christian Medical College, Vellore, Tamil Nadu, India. upasana_k@hotmail.com
-
Received: ,
Accepted: ,
How to cite this article: Kachroo U, Kanthakumar P. An electrophysiological comparison of freshly isolated caprine articular chondrocytes versus cryopreserved chondrocytes. Indian J Physiol Pharmacol 2020;64(3):181-7.
Abstract
Objectives:
Cryopreserved chondrocytes find numerous applications in reconstructive surgery, tissue engineering, and cell-based therapy. Cryopreserved chondrocytes may behave differently due to a change in cell biology. To assess phenotype maintenance, the electrophysiological profile of the cells can be studied. In this study, a comparison between freshly isolated and cryopreserved chondrocytes was made by recording ionic currents using patch clamp.
Materials and Methods:
Caprine articular chondrocytes were harvested and cryopreserved for 7–15 days and divided into two groups. Percentage cell viability was assessed, following which both fresh and cryopreserved cells were subjected to analysis in whole cell configuration using depolarizing voltage steps.
Results:
Outwardly, rectifying currents were recorded in both groups. Comparison of current densities at all potentials above the threshold, revealed no significant difference between fresh and cryopreserved chondrocytes.
Conclusion:
As electrophysiological properties of cryopreserved chondrocytes appear to be maintained, they may be readily utilized in place of fresh cells.
Keywords
Articular chondrocytes
Cryopreservation
Electrophysiology
Patch clamp
INTRODUCTION
Cartilage is the well-known cushion, which prevents unnecessary friction between two articular surfaces. A closer look at its architecture shows chondrocytes seen embedded in abundant extracellular matrix (ECM).[1] Since articular cartilage deals with an extraordinary amount of stress, this ECM (in part synthesized by chondrocytes) provides the necessary tensile strength to overcome that. Mechanical loading and unloading keeps chondrocytes metabolically active resulting in optimal ECM formation which, in turn, protects these cells from physical harm.[2] This fine balance may be disrupted by trauma, defect, or disease. A large amount of work in orthopedic research, autologous transplant surgeries, and tissue engineering depends on the use of preserved biological samples and the same holds true for articular cartilage and chondrocytes.[3] A highly prevalent method of preserving tissue or cells for later use is cryopreservation.[4] Cryopreservation of chondrocytes has been in vogue for many years and subsequently, cryopreservation of whole cartilage shavings has also been attempted.[3,5,6] Many experiments use cryopreserved samples (preserved cells or cells derived from preserved tissue) under the assumption that there is no phenotypic or functional variation. The previous studies have used a multitude of different techniques including, chromatography, immunocytochemical analysis, cell culture, and polymerase chain reaction (PCR), to compare cryopreserved and freshly isolated chondrocytes.[7-10] However, factors such as type and concentration of cryoprotectant used, freezing rate and thawing rate may affect the viability, and cellular proliferation and ability to produce proteoglycans after retrieval.[4,9] Literature also states that incubation of cells/tissue in additives, such as ascorbic acid, glucosamine, chondroitin sulfate or their inclusion in cryo-preservative media reduces cryoprotectant toxicity induced cellular injury thereby improving cell viability on retrieval.[11,12] Till date, there are no reports on the membrane function of cryopreserved chondrocytes, comparing them with freshly isolated cells. One of the methods to assess membrane function is to study its ion channel population and their characteristics.[13] Electrophysiological assessment of articular chondrocytes has revealed the presence of myriad channels, either associated with volume regulation, ECM production or other cellular homeostatic functions. The channelome includes, voltage dependent potassium, calcium, sodium and chloride channels, non-selective cation or transient receptor potential channels, purinergic receptors, and pumps such as Na/K-ATPase and PMCA among others.[14-16] Therefore, this study aimed to compare freshly isolated and cryopreserved chondrocytes based on their ion channel profile using patch clamp technique.
MATERIALS AND METHODS
Dissociation of chondrocytes
This study received approval by the Institutional Review Board (IRB Min No. 8441). Goat legs were obtained from a registered local abattoir (coordinates 12.93°N 79.13°E) and transported to the laboratory in cold physiological Ringer lactate solution. Legs from adult male goats (n = 4, mean age = 7.5 ± 2.5 years) were used for this study. Goat legs were then washed, de-skinned, and decontaminated with 70% ethanol. Cartilage shavings obtained from the meta-tarsophalangeal joint were incubated in Dulbecco’s Modified Eagle’s Medium-F12 (DMEM - Sigma Life Sciences) containing 0.15% Collagenase Type II (Worthington Biochemical Corporation) for 16 h at 37°C, 5% CO2 and 85% humidity.[17] At the end of incubation, contents were passed through a 40-micron filter. Cell count and percentage viability were recorded by Trypan blue dye exclusion technique.
Cryopreservation
For Sets 1, 2, and 4, aliquots of 1 million cells each were suspended in cryo-preservative medium containing 10% Dimethyl Sulfoxide (DMSO: Sigma Aldrich), 20% Fetal Bovine Serum (FBS: Gibco Life Technologies), and 70% DMEM (with the following additives: 2.5 mM/L L-glutamine [Sigma], and 62 μg/ml ascorbic acid [Sigma], 100 IU/ml penicillin-streptomycin [Gibco], and 2 μg/ml amphotericin-B [Gibco]). Due to low cell retrieval (reduced number of cartilage shavings while harvesting) in Set 3, aliquots of 0.5 million cells were prepared and suspended in cryo-preservative medium. After being gradually cooled overnight in −80°C freezer, aliquots were moved to a liquid nitrogen storage device. Cryopreservation lasted for a period of 7–15 days. Based on the duration of cryopreservation, cells were divided into: Group 1: 7–10 days, and Group 2: 11–15 days of preservation. On retrieval, rapid thawing by immersing in a 37°C water bath was performed and cells were washed twice with DMEM (with the following additives: 2.5 mM/L L-glutamine, 62 μg/ml ascorbic acid, 100 IU/ml penicillin-streptomycin, and 2 μg/ml amphotericin-B). Cell count and percentage viability were assessed again.
Patch clamp
Borosilicate glass capillary tubes (Kimax Capillaries; Fischer Scientific, USA) were utilized for fabrication of pipettes. Pipette pulling was performed using a gravity assisted, two-step Narishige-PP-830 vertical pipette puller. The resistance of pipettes thus made (for experiments) was of the order of 1.2-2.8 MΩ. Cell suspension containing at least 50,000 cells was used for each patching experiment. Cells were suspended in a bath solution with a composition of: (in mM) Glucose 10, HEPES 10, CaCl2 1, MgCl2 1, KCl 5, and NaCl 140 with a pH 7.4 using 1M NaOH, while the pipette solution had: (in mM) Glucose 10, HEPES 10, MgCl2 2, and KCl 140 with a pH 7.35 using 1M KOH. The osmolarity (in mOsm/L) for bath and pipette solutions had a range of 290–310 and 280–290, respectively. Axopatch 200B Patch-Clamp amplifier was used for acquiring data while digitization was done using Axon Instruments Digidata-1322A (Molecular Devices, CA, USA). After attaining giga-seal, sharp suction was applied to gain access into the cell and whole-cell configuration was used to make all recordings. Cell capacitance was also measured for each chondrocyte using Clampex 9.2 software’s membrane test function (Molecular Devices, CA, USA). Data filtering was done at 10 kHz (low-pass Bessel filter) while sampling rate set at 50 KHz. Depolarizing, incremental pulses (10 mV apart) which ranged from −80 mV to +70 mV were utilized to record ionic currents (VHold −80 mV). Leak subtraction was performed by measuring leak currents at hyperpolarizing potentials. Tail currents were also recorded to measure reversal potential. Comparison with calculated Nernst equilibrium potential was made to confirm the permeable ionic species. To block potassium currents, 10 mM Tetra-ethyl-ammonium chloride (TEACl) was focally perfused into the bath using OCTAFLOW II perfusion set up (ALA Scientific Instruments).
Statistical analysis
Offline analysis and pictographic representation of data was done using pClamp, MS Excel and IGOR Pro (Version 5.0.4.8, Wavemetrics Inc.,). Kruskal–Wallis Test was used for comparison of current densities across groups at significant voltages. Independent samples Mann–Whitney U-test was used to determine inter-group statistical significance while comparing capacitance. Tests were run on SPSS software-version 17.0 and P < 0.05 was considered significant.
RESULTS
Results of cryopreservation
Cell viability in freshly isolated chondrocytes from cartilage shavings obtained from all four sets was found to be 99%. From the four sets kept for cryopreservation, cells from only three sets could be recovered after thawing. Set 3 on all post-thaw days showed only cellular debris, when stained or unstained samples were checked for viable cells. Cell morphology of cryopreserved chondrocytes was seen to be similar to freshly isolated cells on microscopic examination [Figure 1]. For set 1, number of viable cells retrieved per million cells cryopreserved was 0.65 million on day 7 and reduced to 0.08 million on day 15. Percentage viability on day 7 was recorded to be 70% which reduced to 10% by day 15. For Set 2, number of viable cells retrieved per million cells cryopreserved was 0.5 million on day 7, increased to 0.7 million on day 8, and again reduced to 0.59 million on day 15. Percentage viability on day 7 was recorded to be 70% which reduced to 66% by day 15. For set 4, number of viable cells retrieved per million cells cryopreserved was 0.84 million on day 7 and reduced to 0.61 million on day 15. Percentage viability on day 7 was recorded to be 88% which reduced to 68% by day 15 [Table 1].
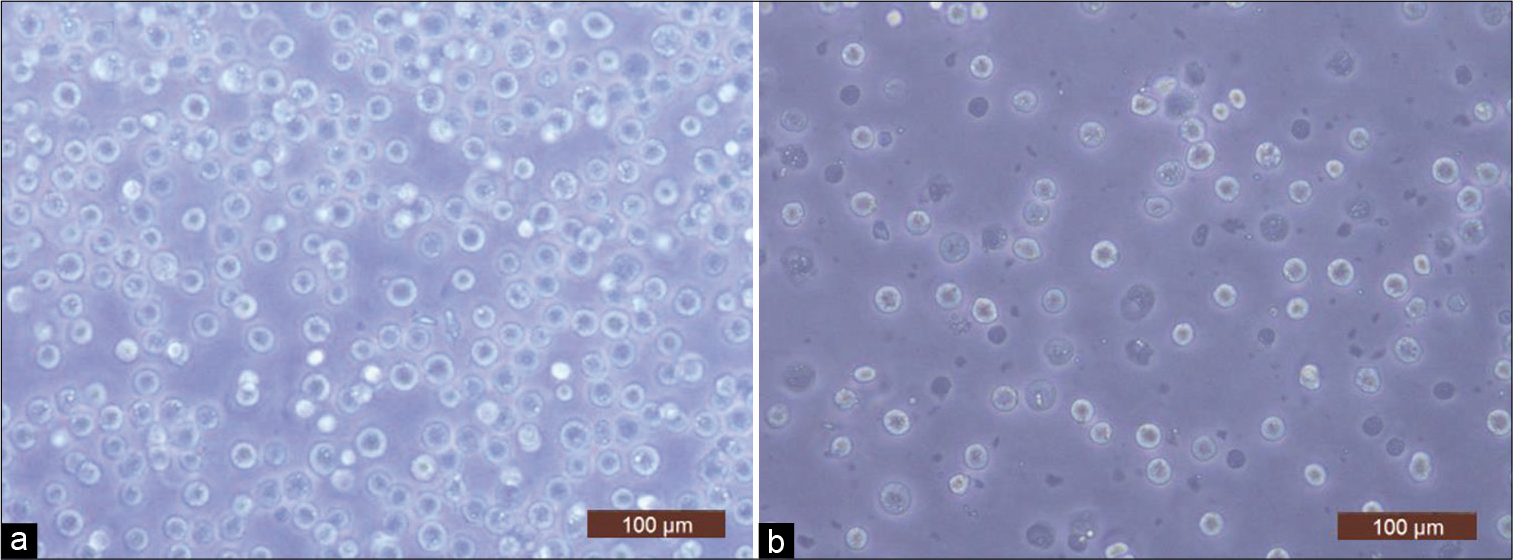
- Representative light microscope images (×20) of (a) freshly isolated chondrocytes (day 0) and (b) cryopreserved cells (day 8).
| Day of retrieval | Number of viable cells retrieved/million cells cryopreserved (in millions) | Viability (%) Trypan blue dye exclusion | ||||||
|---|---|---|---|---|---|---|---|---|
| Set 1 | Set 2 | Set 3 | Set 4 | Set 1 | Set 2 | Set 3 | Set 4 | |
| Day 7 | 0.65 | 0.50 | 0 | 0.84 | 70 | 70 | 0 | 88 |
| Day 8 | 0.59 | 0.70 | 0 | 0.82 | 74 | 72 | 0 | 86 |
| Day 9 | 0.53 | 0.76 | 0 | 0.74 | 72 | 80 | 0 | 82 |
| Day 10 | 0.35 | 0.67 | 0 | 0.75 | 58 | 70 | 0 | 84 |
| Day 11 | 0.49 | 0.65 | 0 | 0.69 | 60 | 72 | 0 | 79 |
| Day 12 | 0.17 | 0.62 | 0 | 0.68 | 39 | 68 | 0 | 74 |
| Day 13 | 0.35 | 0.61 | 0 | 0.70 | 36 | 65 | 0 | 70 |
| Day 14 | - | 0.60 | 0 | 0.68 | - | 64 | 0 | 71 |
| Day 15 | 0.08 | 0.59 | 0 | 0.61 | 10 | 66 | 0 | 68 |
Patch clamp analysis
An analysis of currents recorded in freshly isolated chondrocytes showed the presence of voltage activated, non-inactivating, and outward currents [Figure 2a]. Current magnitude varied across the cells that were patched (peak amplitudes ranging from about 2400 to 8300 pA). In addition to this, when raw tracings were used to generate Current-Voltage (I-V) curves, outward rectification was observed which was suggestive of voltage gated potassium current [Figure 2b]. Focal perfusion with 10 mM TEACl produced a notable reduction in current amplitude in all patched chondrocytes [Figure 2c], aiding in identification of the current as deriving its origin from a voltage-gated potassium channel sub-family. Reversal potential obtained from the tail currents ranged from −75 mV to −69 mV (n = 3) [Figure 2d] which was near the calculated Nernst equilibrium potential of potassium (Calculated value = −84.7 mV). This provided further evidence that potassium was the predominant ionic species that contributed to the outward current.
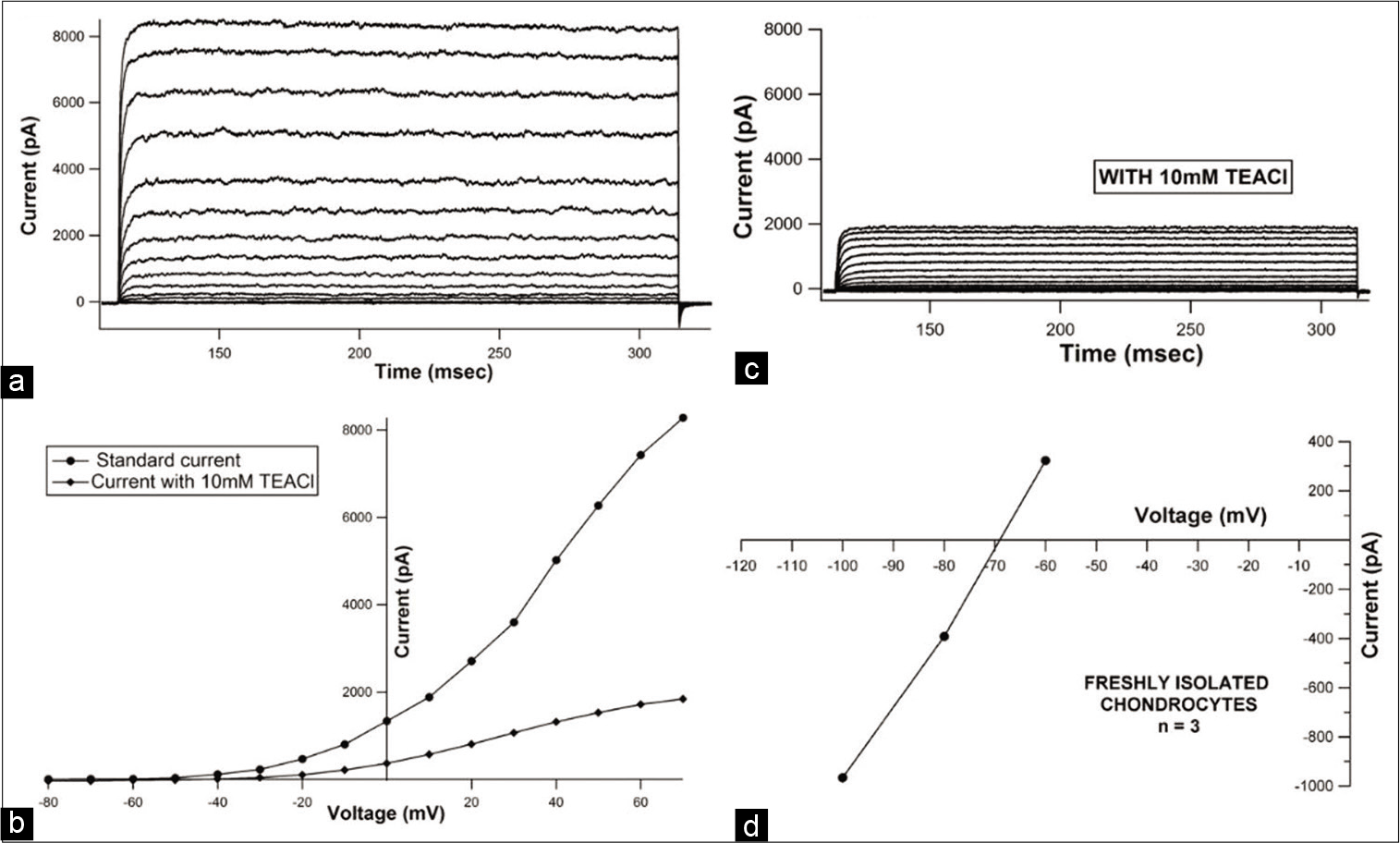
- Representative current tracings from a fresh chondrocyte (a) recorded under control conditions, and (b) after exposure to 10 mM Tetra-ethyl-ammoniumchloride (TEACl). (Protocol:VHold= −80 mV, depolarizing steps ranging from −80 mV to +70 mV with an increment of 10 mV were applied) (c) representative current-voltage (I-V) curve before and after addition of 10 mM TEACl showing drop in peak current magnitude at every voltage step seen after addition of 10 mM TEACl (d) representative leak subtracted instantaneous tail current I-V curve depicting reversal potential for a freshly isolated chondrocyte.
Inter-group comparison
When current tracings were analyzed, they revealed similar current profiles in freshly isolated chondrocytes and cryopreserved cells from both groups. Comparison of mean current densities of fresh chondrocytes (n = 6) with cells from Group 1 (n = 9) and Group 2 (n = 6) showed highest current magnitude in Group 1 (707.177 pA/pF at +40 mV) and lowest current magnitude in Group 2 (At +40 mV, 491.66pA/ pF), while that of fresh chondrocytes (At +40 mV, 706.42 pA/pF) fell in between [Figure 3].When the mean current densities were compared at specific voltages, no significant difference between these three groups was noticed. Effect of 10 mM TEACl on current magnitude of cryopreserved as well as fresh cells was also similar. It was also observed that the reversal potential for cryopreserved cells (Group 1 range: −73.5–−62 mV; n = 4 and Group 2 range: −68.5–−66.5 mV; n = 2) was in the same range as freshly isolated chondrocytes (range: −75–−69 mV; n = 3) [Figure 4]. The mean cell capacitance of freshly isolated chondrocytes and Group 1 cells was comparable with capacitances of 6.03 ± 2.8 pF (n = 6) and 6.36 ± 2.16 pF (n = 9), respectively; while there was a significant decrease in capacitance seen in Group 2, with a mean of 3.61 ± 0.83 pF (n = 6) (P value for day 0 vs. Group 2 = 0.041 and Group 1 versus Group 2 = 0.003) [Figure 5].
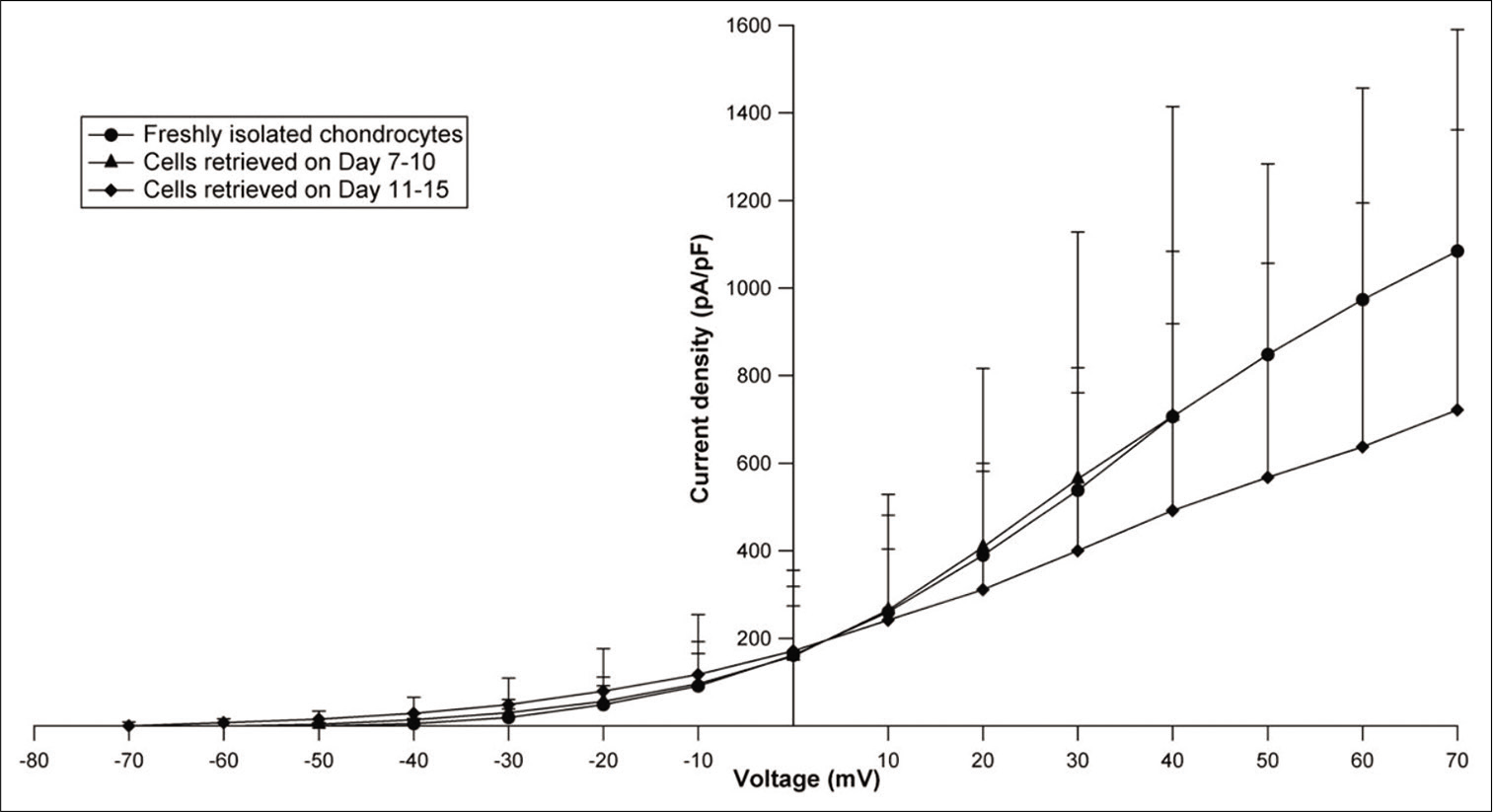
- Current-voltage curves plotted using mean current densities and membrane voltage for fresh and cryopreserved chondrocytes (Groups 1 and 2). The last 3 points for Group 1 (day 7–10) have not been plotted as the value of “n” was unequal.
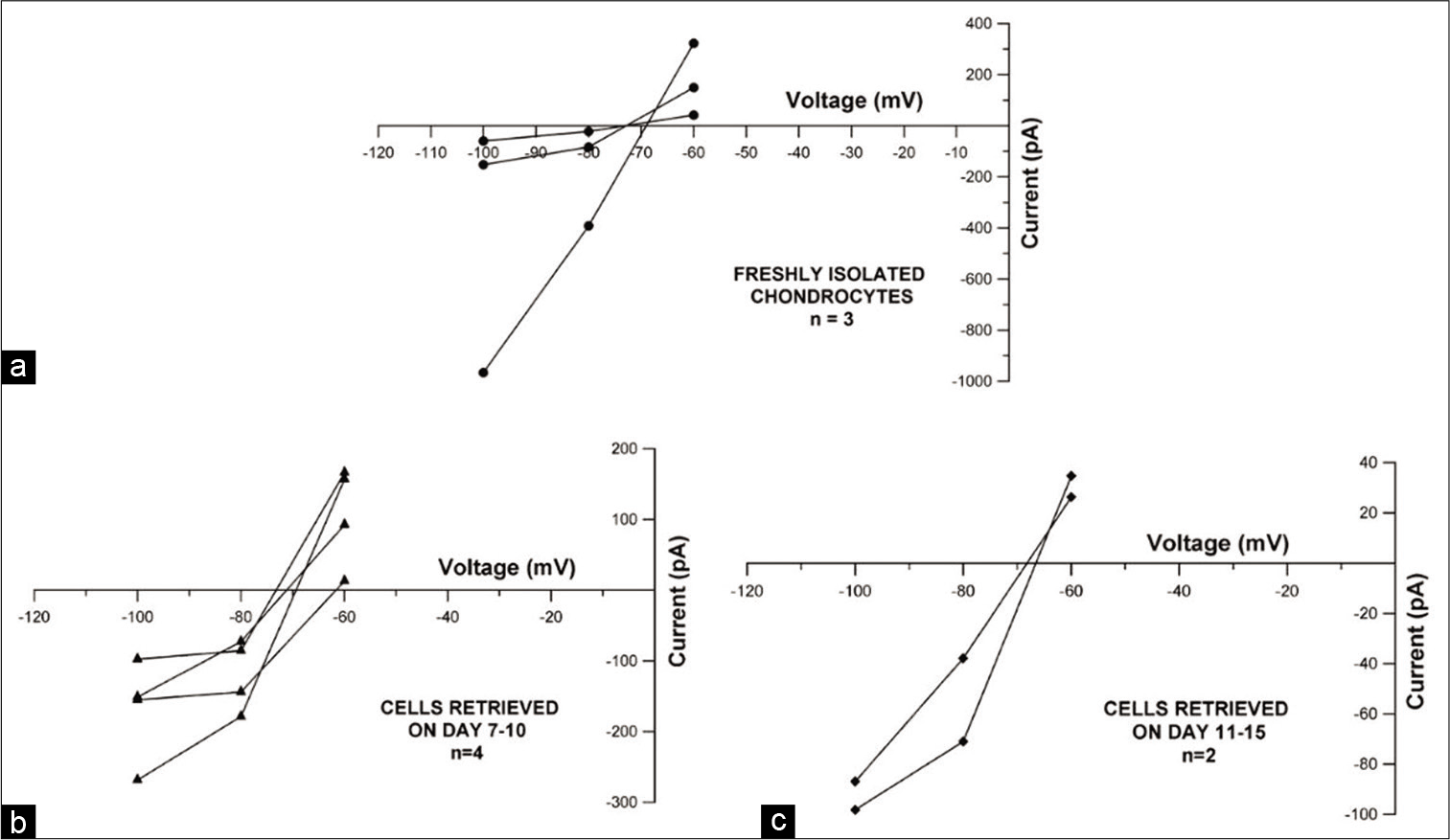
- Current-voltage plot displaying leak subtracted current versus voltage to depict reversal potential for (a) freshly isolated chondrocytes (n = 3), (b) Group 1 cryopreserved cells (n = 4) and (c) Group 2 cryopreserved cells (n = 2).
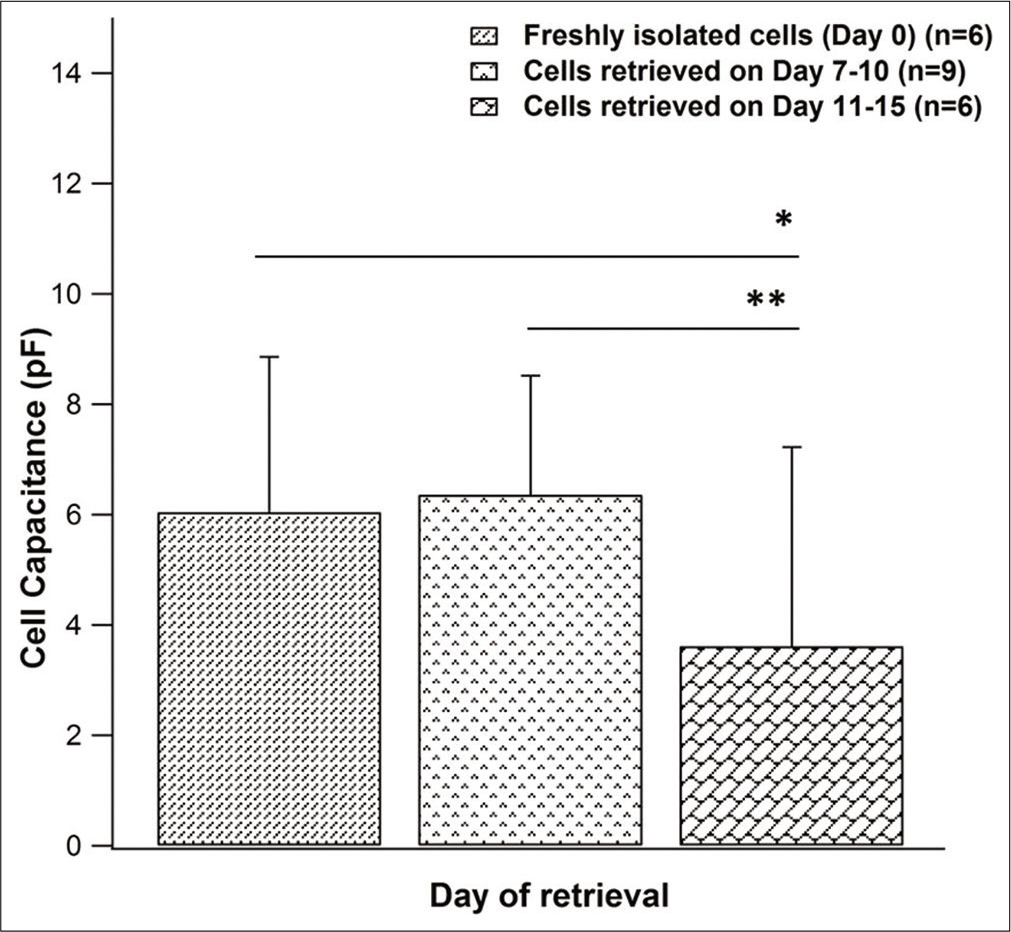
- Comparison of cell capacitance between freshly isolated chondrocytes (n = 6) and cryopreserved cells (Group 1 – n = 9, Group 2 - n = 6). Data represented as mean ± SD (*P < 0.05; **P < 0.01 using Mann–Whitney U-Test).
DISCUSSION
Subjecting freshly isolated chondrocytes to the cryopreservation protocol yielded viable cells which looked morphologically similar to fresh cells. DMSO was used as the cryoprotective agent (CPA), which is one of the most commonly used CPAs and has been employed extensively for cryopreserving chondrocytes.[18-21] The concentration of DMSO used was fixed after reviewing test sample viability and also available literature.[18,22] In our study, chondrocytes remained viable after cryopreservation with a peak viability of 88% on day 7 (Set 4) and minimum viability of 10% on day 15 (Set 1). For one of the sets, no cells could be retrieved despite using the same freezing and thawing protocol. Low cell yield before cryopreservation, as a result of questionable animal health, may have resulted in added cryoprotectant toxicity or osmotic and freezing stress, leading to cell damage and death. Some variation in viability can be accounted to the storage conditions prevalent in the cryopreservation unit since reports also suggest that cells stored in the vapor phase demonstrate better viability than those stored in the liquid phase.[23,24]
Both freshly isolated chondrocytes and cells retrieved after cryopreservation, when subjected to a depolarizing protocol showed the presence of a family of outward currents. These currents were non-inactivating and outwardly rectifying in nature, suggestive of potassium currents. The blockade of the currents with 10 mM TEACl and the reversal of the currents close to calculated Nernst potential for potassium, confirmed the permeant ion to be potassium.
Comparison of the current densities at specific voltages did not show significant difference (P value at different voltages: 0.883 at −10mV, 0.838 at 0mV, 0.781 at +10mV, 0.809 at +20mV, 0.661 at +30mV, and 0.635 at +40mV) between fresh cells and cells obtained after cryopreservation (Group 1 and Group 2). This suggests that cryopreservation did not affect the current per unit surface area of the cells. Reversal potential as well as the effect of TEACl on cryopreserved cells also gave similar results as were seen with freshly isolated chondrocytes. It can, therefore, be proposed that cryopreserved chondrocytes do not differ from their freshly isolated counterparts in terms of potassium channel function.
Cell capacitance, which is a measure of cell size,[25] was also compared across groups. It was observed that as the cells were cryopreserved for a longer duration as in the case of Group 2, there was a reduction in the mean cell capacitance.
From the results obtained from this study, it appears that the cell capacitance of freshly isolated chondrocytes and cryopreserved cells is comparable till only about 10 days of cryopreservation following which cell capacitance shows a significant decline (Freshly isolated versus Group 2 P = 0.041 and Group 1 cells versus Group 2 cells P = 0.003). To further study this effect, a larger sample size to observe the interaction of chondrocytes with the CPA used, their behavior when exposed to freezing stress, as well as a detailed electrophysiological analysis is required. Some studies have used cryopreserved cells and compared them to fresh chondrocytes to investigate cell morphology, function, potassium channel expression by means of monolayer culturing, chromatographic methods, PCR, as well as its ability to form engineered tissue when seeded in scaffolds.[10,22,26] With the results seen in our study, the presumption that cryopreserved chondrocytes have a similar functional profile as fresh chondrocytes, is supported. It may be implied that chondrocytes may be preserved in this manner to bypass the issue of inadequate sample source, reduce cost of repeated isolation, and maintain a chondrocyte bank for future use.
The observations from this study suggest that articular chondrocytes may be successfully cryopreserved over short durations with good retrieval and viability although a more stringent analysis is required for predicting cellular health. Another limitation was that cell proliferation and ability to synthesize glycosaminoglycan was not assessed. It would also be helpful if different cryoprotectants are compared as that would optimize the preservation protocol leading to better yield on retrieval. In our study, patch clamp was used to analyze the potassium currents in chondrocytes before and after cryopreservation. Since chondrocytes have been shown to have numerous other channels,[15] it would be helpful to compare whether functionality of these channels is affected by cryopreservation or not.
CONCLUSION
It may be stated that cryopreserved chondrocytes can be utilized in place of freshly isolated chondrocytes when there is any limitation to their use, as retrieval of viable and functional chondrocytes can advance the fields of tissue engineering, reconstructive surgery, and orthopedic research.
Acknowledgment
The authors would like to thank Dr. Visalakshi Jeyaseelan for help with statistical analysis.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
The authors would like to thank CMC Vellore Internal Fluid Research Fund for the grant to conduct the study (IRB Min No. 8441).
Conflicts of interest
There are no conflicts of interest.
References
- Reserve or resident progenitors in cartilage? Comparative analysis of chondrocytes versus chondroprogenitors and their role in cartilage repair. Cartilage. 2018;9:171-82.
- [CrossRef] [PubMed] [Google Scholar]
- The cellular physiology of articular cartilage. Exp Physiol. 1996;81:535-45.
- [CrossRef] [PubMed] [Google Scholar]
- How to manage a failed cartilage repair: A systematic literature review. Joints. 2017;5:93-106.
- [CrossRef] [PubMed] [Google Scholar]
- Cryopreservation and its clinical applications. Integr Med Res. 2017;6:12-8.
- [CrossRef] [PubMed] [Google Scholar]
- Biological freezing of human articular chondrocytes. Osteoarthritis Cartilage. 2001;9:341-50.
- [CrossRef] [PubMed] [Google Scholar]
- Cryopreservation of cartilage cell and tissue for biobanking. Cryobiology. 2011;63:292-7.
- [CrossRef] [PubMed] [Google Scholar]
- Studies on cryopreservation of articular cartilage chondrocytes. J Bone Joint Surg Am. 1984;66:253-9.
- [CrossRef] [PubMed] [Google Scholar]
- Cryopreservation effect on proliferative and chondrogenic potential of human chondrocytes isolated from superficial and deep cartilage. Open Orthop J. 2012;6:150-9.
- [CrossRef] [PubMed] [Google Scholar]
- Study of cryopreservation of articular chondrocytes using the Taguchi method. Cryobiology. 2010;60:165-76.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of chondrocyte cryopreservation on cartilage engineering. Vet J. 2008;178:244-50.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of additive compounds on glycerol-induced damage to human chondrocytes. Cryobiology. 2017;75:68-74.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of five additives to mitigate toxicity of cryoprotective agents on porcine chondrocytes. Cryobiology. 2019;88:98-105.
- [CrossRef] [PubMed] [Google Scholar]
- From Bernstein's rheotome to Neher-Sakmann's patch electrode. The action potential. Physiol Rep. 2019;7:e13861.
- [CrossRef] [PubMed] [Google Scholar]
- Potassium channels in articular chondrocytes. Channels (Austin). 2012;6:416-25.
- [CrossRef] [PubMed] [Google Scholar]
- The emerging chondrocyte channelome. Front Physiol. 2010;1:135.
- [CrossRef] [PubMed] [Google Scholar]
- The chondrocyte channelome: A narrative review. Joint Bone Spine. 2019;86:29-35.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of human articular chondrocyte and chondroprogenitor cocultures and monocultures: To assess chondrogenic potential and markers of hypertrophy. Tissue Cell. 2019;57:42-8.
- [CrossRef] [PubMed] [Google Scholar]
- Isolation, propagation, and cryopreservation of equine articular chondrocytes. Am J Vet Res. 1992;53:2364-70.
- [Google Scholar]
- Effect of cryopreservation on human articular chondrocyte viability, proliferation, and collagen expression. Cryobiology. 2001;42:2-10.
- [CrossRef] [PubMed] [Google Scholar]
- Ultrastructural changes of articular cartilage chondrocytes associated with freeze-thawing. J Orthop Res. 1993;11:1-9.
- [CrossRef] [PubMed] [Google Scholar]
- Viability, proliferation and phenotype maintenance in cryopreserved human iliac apophyseal chondrocytes. Cell Tissue Bank. 2014;15:153-63.
- [CrossRef] [PubMed] [Google Scholar]
- Two-pore domain K+ channels regulate membrane potential of isolated human articular chondrocytes. J Physiol. 2011;589:5071-89.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of liquid nitrogen vapor sensitization conditions on the quality of frozen-thawed dog spermatozoa. J Vet Med Sci. 2006;68:1055-61.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of liquid nitrogen vapor storage on the motility, viability, morphology, deoxyribonucleic acid integrity, and mitochondrial potential of frozen-thawed human spermatozoa. Fertil Steril. 2010;94:2736-41.
- [CrossRef] [PubMed] [Google Scholar]
- Analysis of depolarization-induced outward currents in goat chondrocytes using the patch clamp technique. Indian J Physiol Pharmacol. 2010;54:361-5.
- [Google Scholar]
- Cryopreservation and biophysical properties of articular cartilage chondrocytes. Cryobiology. 2005;51:330-8.
- [CrossRef] [PubMed] [Google Scholar]






