Translate this page into:
Chronic treatment of 4-phenylbutyric acid ameliorates cognitive impairment after focal cerebral ischemia/ reperfusion injury in rats
*Corresponding author: Shyam Sunder Sharma, Department of Pharmacology and Toxicology, National Institute of Pharmaceutical Education and Research, Mohali, SAS Nagar, Mohali - 160 062, Punjab, India. sssharma@niper.ac.in
-
Received: ,
Accepted: ,
How to cite this article: Ramakrishna K, Srinivasan K, Sharma SS. Chronic treatment of 4-phenylbutyric acid ameliorates cognitive impairment after focal cerebral ischemia/reperfusion injury in rats. Indian J Physiol Pharmacol 2020;64(3):188-94.
Abstract
Objectives:
Stroke, apart from causing physical disabilities, it also often leads to cognitive impairment in patients. At present, there is no effective drug available for the treatment of post-stroke cognitive impairment. The present study was undertaken to evaluate the ameliorative effect of 4-Phenylbutyric acid (PBA) against cognitive and memory deficits due to focal cerebral ischemia/reperfusion (I/R) in rats.
Materials and Methods:
Focal cerebral I/R injury was achieved by the middle cerebral artery occlusion (MCAO) method. PBA (100 and 300 mg/kg, i.p.) was administered once daily for 2 weeks. The neurological score was counted to evaluate the severity of neurological motor deficits. The cognitive functions, including learning and memory, were assessed using various paradigms such as Y-maze, passive avoidance task and Morris water maze.
Results:
The chronic treatment of PBA (100 and 300 mg/kg, i.p.) dose-dependently improved the neurological motor deficits as shown by significant decrease in neurological score in MCAO-treated rats. Besides, PBA (100 and 300 mg/kg, i.p.) treatment markedly improved working memory as shown by significant increase in the relative percentage alternations compared to untreated control MCAO rats in Y-maze. PBA also significantly decreased the transfer latency in the acquisition trial and increased in probe trial in passive avoidance task suggesting an improvement in learning and memory in MCAO rats. There was also a significant improvement in spatial learning and memory, as evidenced by the reduced escape latency in acquisition trial and the increased number of entries into the platform zone, time spent in the platform quadrant and track plot in probe trial PBA-treated MCAO rats during Morris water maze task.
Conclusion:
This study, thus, demonstrates the potential of PBA in ameliorating cognitive dysfunctions in focal cerebral ischemia. Since PBA is already available for the treatment of urea cycle disorders, it may also be investigated for repurposing its use in the treatment of post-stroke cognitive impairment.
Keywords
Middle cerebral artery occlusion
4-Phenyl butyric acid
Cognitive impairment
Passive avoidance
Morris water maze
INTRODUCTION
Stroke is the dominant cause of permanent disabilities and often leading cause of death in adults.[1] Cognitive impairment is one of the major disability of strokes with decreased quality of life around the globe.[2] Cognitive impairment after the stroke occurs frequently and remains persistently for a long time. The stroke-induced cognitive impairment ranges from 20 to 80%, which widely depends on countries, races, age, sex, education, occupation, diagnostic criteria and vascular factors.[3] The hippocampal, white matter lesions and cerebral microbleeds after stroke results in cognitive impairment. The American stroke association recommended for using choline esterase inhibitors (donepezil, galantamine and rivastigmine) and NMDA antagonist (memantine) against post-stroke cognitive impairment.[3-5] However, due to diversity in the pathophysiology of stroke-induced cognitive deficits, neuroprotectants and nootropic agents failed to improve the cognitive functions.[5] Hence, there is an urge to study new molecules/approaches/targets to inhibit stroke-induced cognitive impairment. Endoplasmic reticulum (ER) has a pivotal role in the synthesis, folding and trafficking of proteins and calcium homeostasis. Any perturbation that disturbs ER homeostasis such as glucose/energy deprivation, hypoxia and oxidative stress can switch to the accumulation of unfolded proteins, which leads to ER stress.[6] The failure of unfolded protein response (UPR) evoked by ER stress to maintain ER homeostasis eventually leads to apoptotic cell death in some neurodegenerative and neurological disorders, including stroke.[7-9] A few recent studies have reported that ER stress-mediated hippocampal neuronal apoptosis is involved in cognitive impairment associated with diabetes and other chemical-induced brain injuries.[10,11] 4-Phenyl butyric acid (PBA) is a known chemical chaperone[12] possessing anti-inflammatory[13] and antioxidant activity.[14] PBA also reported to has neuroprotective potential and ameliorate cerebral ischemia,[12] spinal cord ischemia,[15] liver ischemia/reperfusion (I/R) injury[16] as well as high glucose-induced alteration in dorsal root ganglion neurons.[17] Besides, it also improves cognition and memory functions in Alzheimer’s disease mouse model.[18,19] However, no study has been reported in the literature highlighting PBA’s effects against cognitive deficits after stroke. Therefore, we planned to study whether PBA’s chronic administration would ameliorate cognitive deficits after focal ischemia/reperfusion (I/R) injury in rats.
MATERIALS AND METHODS
Experimental animals and chemicals
Adult healthy male Sprague-Dawley rats weighing (240– 260 gm) were procured from the central animal facility. The animals were housed in a room maintained at approximately 23 ± 2°C temperature and humidity of 55 ± 5% with 12-h light/dark cycle. Rats were allowed to access feed and water ad libitum. The experimental protocol was approved by the Institutional Animal Ethics Committee (IAEC 14/48), NIPER, SAS Nagar, India. The animal care and experimentation were carried out as per guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Fisheries, Animal Husbandry and Dairying, Government of India. PBA (Sigma-Aldrich, CAS#182111) and thiopentone sodium (Neon Lab. Ltd. Mumbai, India) were procured.
Study design
The animals were randomly assigned to the following experimental groups, namely, Sham, middle cerebral artery occlusion (MCAO), MCAO + PBA 100 mg/kg and MCAO + PBA 300 mg/kg. PBA doses for this study were chosen from the previous studies.[20] The complete study design is depicted in Figure 1. After completion of experiments, all the animals were euthanised and disposed.
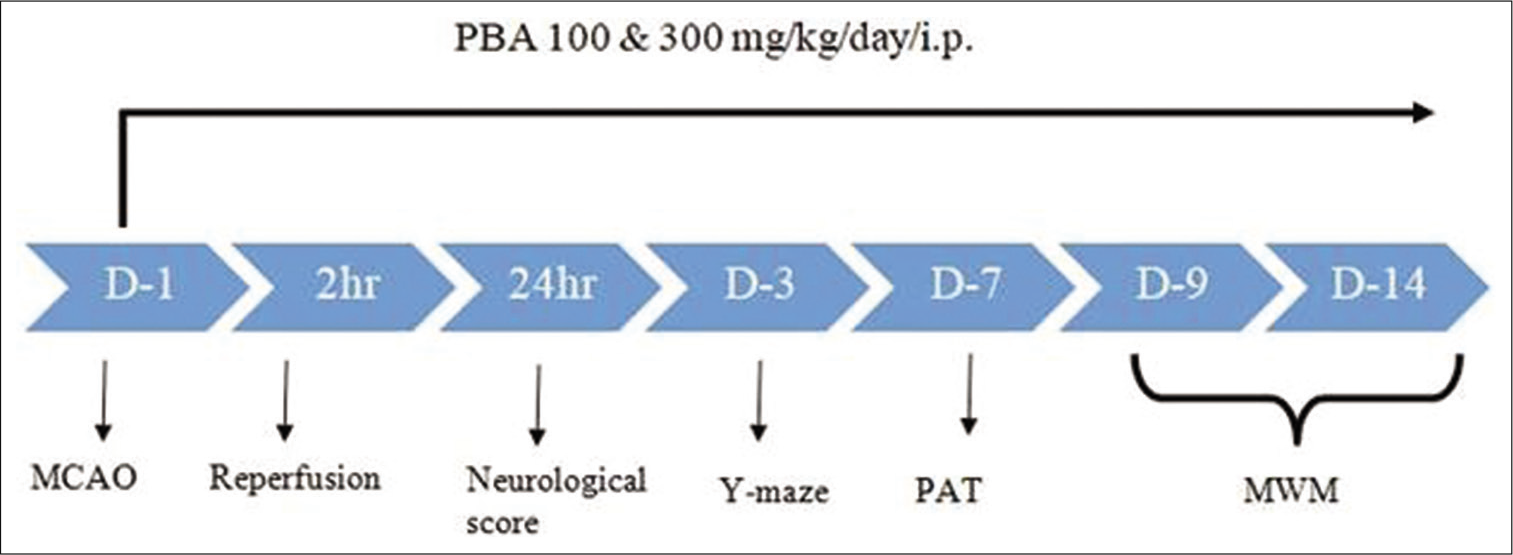
- Schematic representation of the study design. D: Day, MCAO: Middle cerebral artery occlusion, PAT: Passive avoidance test, MWT: Morris water maze test.
Induction of focal cerebral ischemia/reperfusion injury
The focal cerebral I/R injury was achieved by MCAO method. Atropine sulphate (0.5 mg/kg, i.p.) as a preanaesthetic was administered. General anaesthesia was achieved using thiopentone sodium (40 mg/kg, i.p.). Briefly, a middle neck incision was made and the common carotid artery was isolated. The internal and external carotid arteries were separated from adhering tissues and nerve; 18–23 mm of 3-0 nylon monofilament pre-coated with poly-L–lysine having a blunt tip was inserted into an external carotid artery to the right middle cerebral artery. Occlusion was confirmed because of the resistance of filament. After achieving occlusion, the monofilament was kept in place with a ligature, and after that, the incision was sutured. The monofilament was pulled out after 2 h of occlusion to resume the blood flow in the MCA and the incision was closed by suturing. The monofilament was just inserted and taken out in sham-operated animal.[21,22] Body temperature was maintained at 37°C using a rectal probe and heating pad (Harvard apparatus) throughout the surgical procedure. The proper post-operative care was taken for all groups. PBA (100 and 300 mg/kg) was administered 5 min before removing the filament in treatment groups and continued for 2 weeks.
Neurological deficits
Five-point scale method was used to evaluate neurological deficits after 22 h of reperfusion as reported earlier.[21,22] 0 = No neurological deficit, 1 = Failure to extend paw fully, 2 = Circling, if pulled by the tail, 3 = Spontaneous circling and 4 = Did not walk spontaneously had a depressed level of consciousness.
Y-maze spontaneous activity
The Y-maze task is carried out to test the immediate spatial working memory of rodents and is based on their willingness to explore new environments. Each Y-maze arm consists of 50 cm long, 30 cm height and a 10 cm wide and was positioned at an equal angle of 120o. Each rat was placed at the centre of the maze and allowed to move for 5 min. The series of arm entries was recorded using a ceiling-mounted CCD camera and analysed using the ANY-MAZETM software. Alternation was counted when the animal entered the arm different from the two previous arms entered (e.g., ABC, BCA and CAB); if the animal went back to either of the two arms visited previously was considered as a mistake (ABA and BCB).[23] The relative alternation in the percentage of the total arm was calculated using the following formula:
Passive avoidance test
A two-compartment step-through passive avoidance apparatus (PACS-30) was used in the study (Columbus Instruments, USA). The instrument consists of two compartments (illuminated and dark) that are separated by a guillotine door. The dark chamber consists of a series of metallic rods to give an electric shock to the animal. Rats were initially placed in the lighted compartment and allowed to explore for 15 s. After 15 s, the door was open and animals were allowed to explore freely. The guillotine door was closed when the rat enters the dark compartment with all four paws and then each animal was returned to the home cage. After 1 h of habituation, acquisition trial was performed where foot shock (0.6 mA for 6 s) was delivered to the animal, and 10 s later, the rat was returned to its home page.[24] The latency for the rats to enter the dark compartment, that is transfer latency was recorded. On test day (24 h after acquisition trial), the inhibitory avoidance response was recorded by placing each rat in the illuminated compartment, as described above. The latency (time) to enter the dark chamber was recorded as a measure of retention task. A ceiling score of 300 s was given if rats did not enter the dark chamber within 5 min period.
Morris water maze test (MWT)
MWT is often used to assess spatial learning and memory. Briefly, it consists of a pool (black painted) divided into four equal quadrants, partially filled with water and a hidden platform in one of the quadrants. The animal was kept in one quadrant facing toward the wall and was given a 120-s trial to find the platform. The same was repeated for 5 consecutive days with four trials each day to each animal (intra-trial interval is 10 min). All experiments were recorded and analysed using the ANY-MAZE™ software. Escape latency was considered when an animal finds the platform. A probe trial, without platform was performed on the 6th day and animals were given a 120-s trial to find the platform for spatial memory assessment.[24,25]
Statistical analysis
Results are expressed as Mean ± S.E.M. GraphPad Prism5 was used for statistical analysis. ANOVA was used for multiple comparisons (one-way and two-way analysis). If ANOVA shows a significant difference, then post hoc analysis was performed with Newman-Keuls test (one-way ANOVA) and Bonferroni post hoc (two way ANOVA). P < 0.05 was considered statistically significant. The neurological score was expressed as median and analysed using Kruskal–Wallis one-way analysis of variance test followed by post hoc Dunn’s multiple comparison tests.
RESULTS
Effect of PBA treatment on the neurological deficits
The neurological score was significantly higher in MCAO animals as compared to sham animals (P < 0.001). The administration of PBA (100 and 300 mg/kg) significantly attenuated the neurological score (P < 0.05 and P < 0.01, respectively) in treated animals [Figure 2] when compared to untreated control MCAO animals.
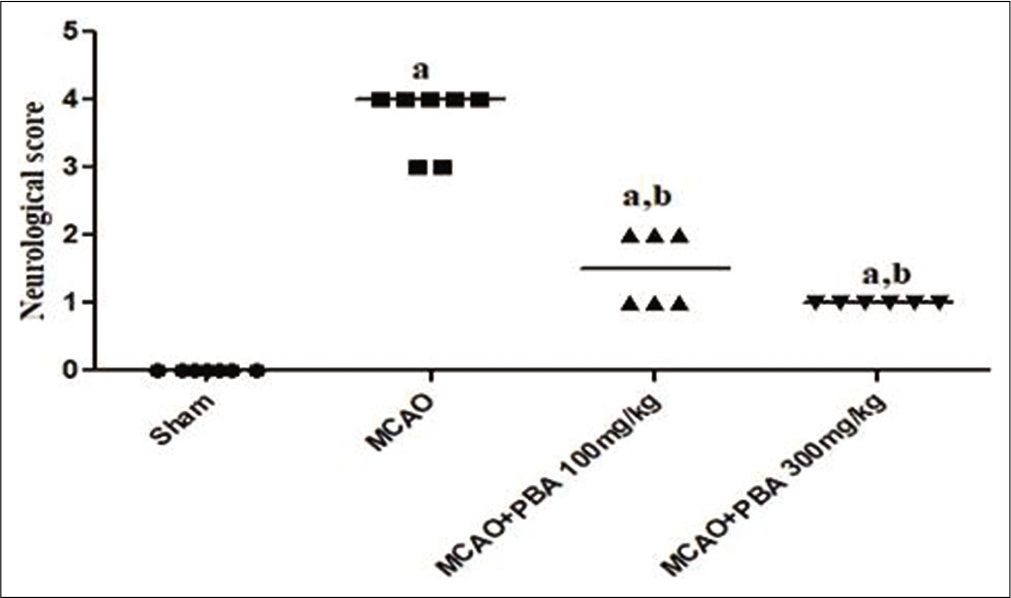
- Effect of 4-phenylbutyric acid treatment on the neurological score.
Effect of PBA treatment on Y-maze spontaneous alternation test
The relative alternation percentage was significantly low in MCAO rats compared to sham-operated animals (P < 0.001). PBA (100 and 300 mg/kg) significantly increased the relative alternation in MCAO treated rats (P < 0.05 and P < 0.001, respectively) [Figure 3].
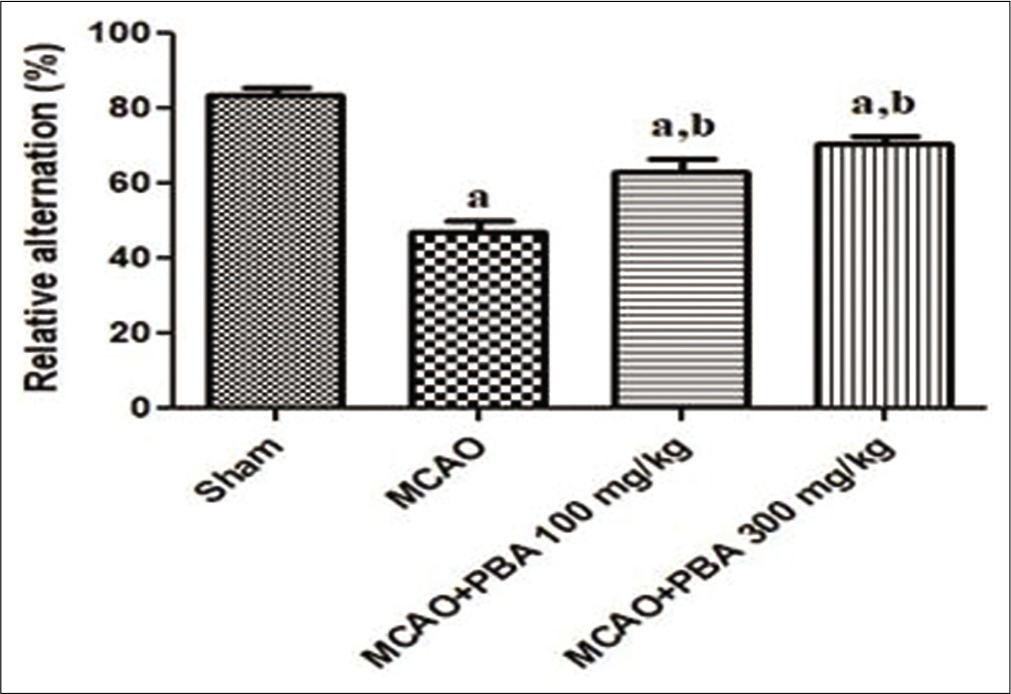
- Effect of 4-phenylbutyric acid treatment on Y-maze spontaneous alternation test.
Effect of PBA treatment on Passive avoidance test
The transfer latency was significantly increased in MCAO rats (P < 0.001) in the acquisition trial, whereas it was reduced (P < 0.001) in the retention trial of MCAO rats as compared to sham-operated animals. PBA intervention (100 and 300 mg/kg) significantly (P < 0.05) decreased the transfer latency in acquisition trial (100 mg/kg: P < 0.05 and 300 mg/kg: P < 0.01) and significantly (P < 0.001) increased the transfer latency in retention trial as compared to untreated MCAO rats [Figure 4].
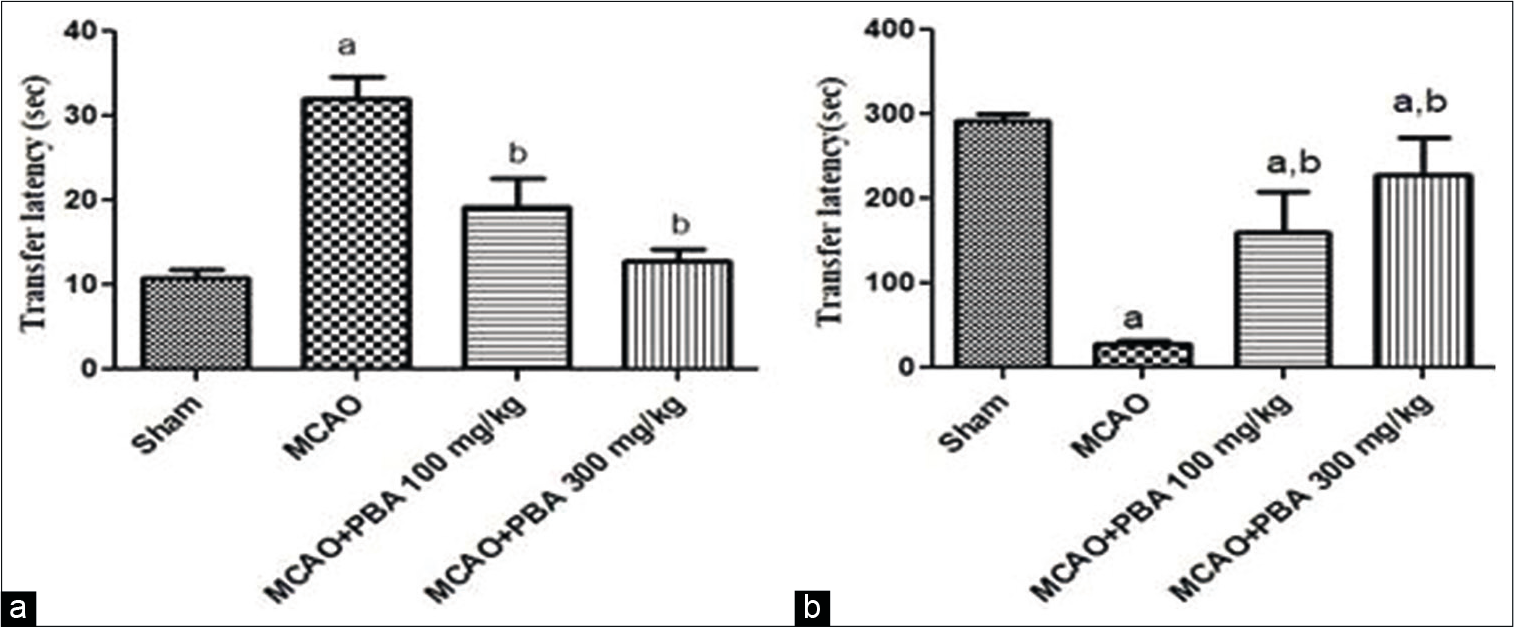
- Effect of 4-phenylbutyric acid treatment on Passive avoidance test, acquisition trial (a) and retention trial (b).
Effect of PBA treatment on Morris water maze
Effect of PBA treatment on Escape Latency (sec) in acquisition trial
The escape latency was significantly increased in MCAO rats compared to sham-operated animals (P < 0.05). A significant reduction in escape latency was observed with PBA treatment (100 mg/kg and 300 mg/kg: P < 0.01) from the day (D)-2 to D-5 when compared to MCAO stroke animals [Figure 5]. However, PBA (300 mg/kg) significantly decreased the escape latency when compared with PBA (100 mg/kg) from D4–D5 (P < 0.05).
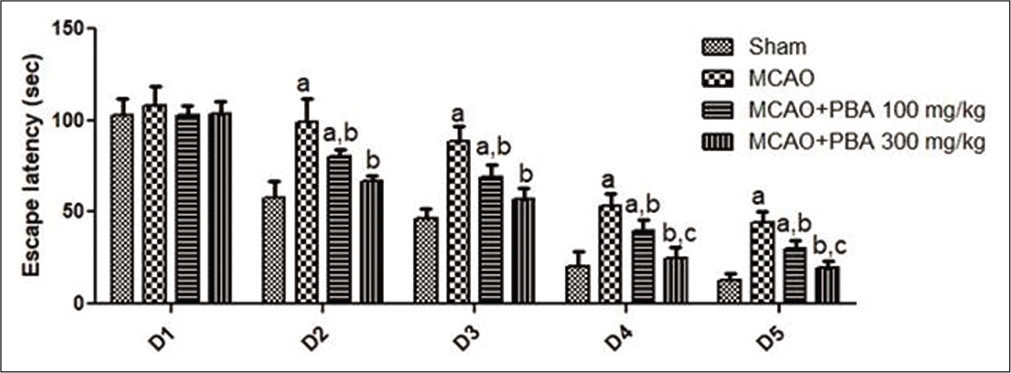
- Effect of 4-phenylbutyric acid treatment on escape latency (sec) during the acquisition trial.
Effect of PBA treatment on the number of entries into the platform zone (A) time spent in the platform quadrant (B) and Track plot (C) in the probe trial
A significant reduction in the number of entries into the platform zone was observed in MCAO rats than the sham group of animals (P < 0.001). PBA significantly and dose-dependently increased the number of entries into the platform zone in MCAO rats (PBA 100 mg/kg and 300 mg/kg: P < 0.05 and P < 0.01, respectively). PBA (300 mg/kg) significantly increased the number of entries into the platform zone compared to PBA (100 mg/kg) (P < 0.05). The time spent in the platform quadrant was significantly decreased in MCAO-challenged rats compared to sham groups (P < 0.001). PBA intervention (100 and 300 mg/kg) significantly increased the time spent in the platform quadrant during probe trial (P < 0.05 and P < 0.001) as compared to MCAO animals [Figure 6].
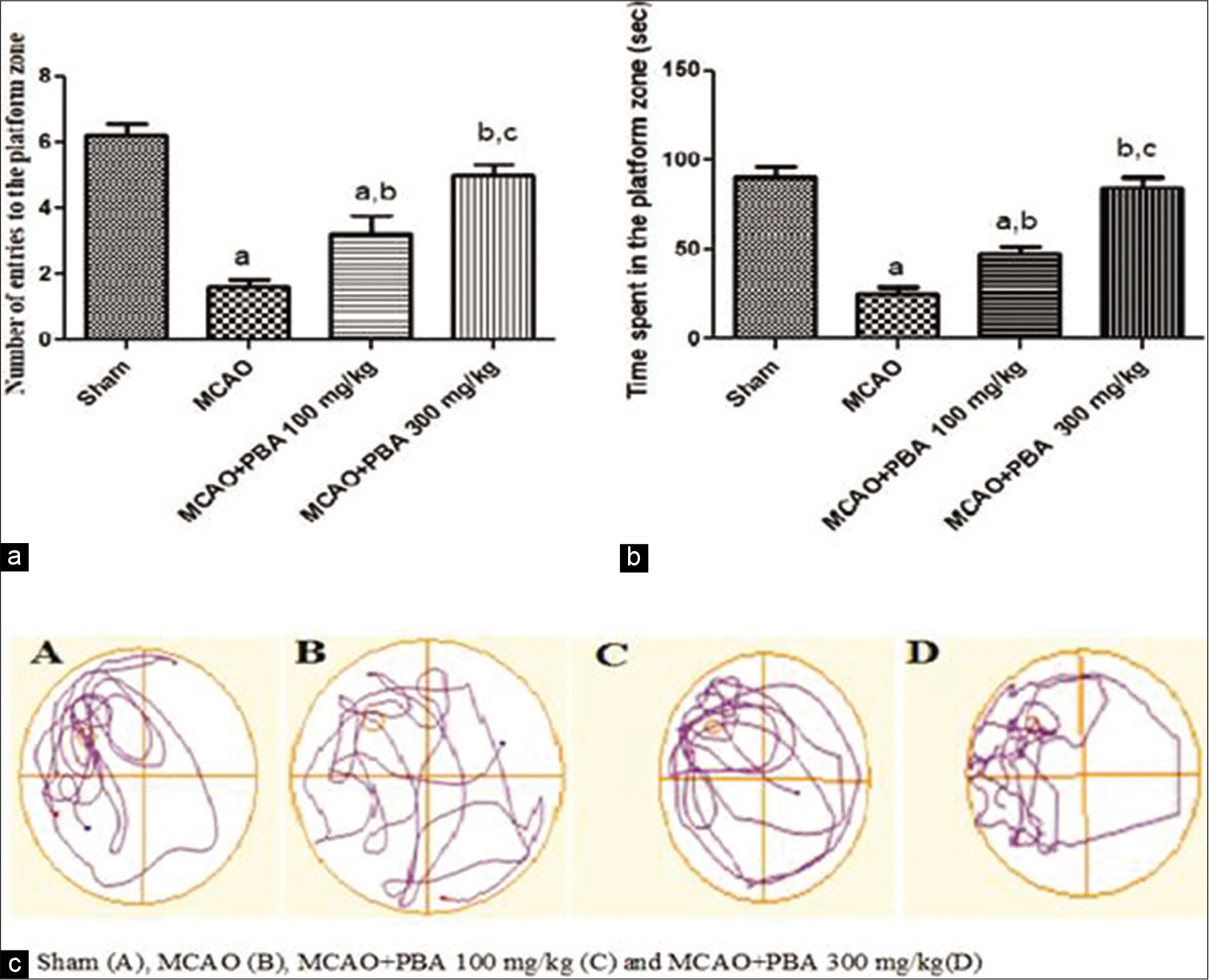
- Effect of 4-phenylbutyric acid treatment on number of entries into the platform zone (a), time spent in the platform quadrant (b) and track plot (c) during the probe trial.
DISCUSSION
This study was undertaken to study the ameliorative effects of PBA against stroke-induced cognitive impairment using a rat model of focal cerebral I/R. MCAO induces several behavioural alterations in animals which include both motor functions as well as learning and memory deficits.[26] MCAO model closely mimics the post-stroke cognitive impairment in humans.[23,27]
In this study, we used different tasks to assess the neurological motor function (neurological score) and cognitive learning and memory functions (Y-maze spontaneous alternation test, step-through passive avoidance test and Morris water maze).
Neurological damage was reflected from the neurological score that was observed after 22 h of reperfusion in MCAO animals.[20] The neurological score indicates the success of MCAO just after reperfusion and evidence for the degree of severity of the injury. In the present study, MCAO animals showed higher neurological scores, whereas sham animals did not show any neurological deficits. PBA treatment significantly reduced the neurological score after 22 h of reperfusion as compared to MCAO rats. This agrees with a recent study from our lab that showed the ameliorative effects of PBA in diabetic stroke rats which was associated with reduced cerebral infarct size as well as neurological motor deficits.[20] In this study, we further explored the PBA’s effects on cognitive functions in a rodent model of ischemic stroke.
Experimental evidence suggested that the cognitive deficits observed in ischemic animals.[3] The hippocampus, basal forebrain and prefrontal cortex are involved in determining the working memory using the Y-maze task.[23] Y-maze spontaneous alternation test was carried out after 72 h of I/R injury to assess the attention as well as working memory function. In our study, MCAO rats showed a marked reduction in relative alternation (%) compared to sham animals, suggesting that MCAO animals developed cognitive impairment. PBA treatment increased the relative spontaneous alternations in MCAO animals suggesting that PBA improved the working memory in MCAO rats.
A large body of literature suggests that learning and memory involve various brain regions that include the hippocampus, temporal lobe and cholinergic nervous system.[28,29] Passive avoidance task is often used to evaluate fear learning and memory in rodent models. In the present study, MCAO animals showed increased transfer latency in acquisition trial and decreased transfer latency in retention trial suggesting significant cognitive impairment in MCAO rats as compared to sham rats. PBA treatment reduces the transfer latency in the acquisition trial and significantly increases the transfer latency in retention trial in MCAO rats. An increased transfer latency in retention trial is a measure of enhanced cognitive performance. It suggests the potential of PBA in ameliorating stroke-induced cognitive impairment.
The Morris water navigation task assessed MCAO-induced long-term spatial cognitive deficits.[30] MCAO produces significant damage to cortex and striatum leads to deficits in learning and memory which is consistently retrieved through the water maze task on MCAO in rats.[31,32] In the present study, it was identified that MCAO leads to impairment in rats’ performance in the water maze paradigm. In the current study, escape latency (acquisition trial), the number of entries into the platform zone, time spent in the platform quadrant and track plots on probe trials were considered to assess the learning and memory function. Escape latency is the time taken to reach the hidden platform. Increased escape latency in MCAO animals represents cognitive impairment, whereas PBA treatment reduces the escape latency in MCAO treated animals. The probe trial in terms of decreased number of platform entries and time spent in the platform quadrant revealed significant cognitive impairment in MCAO challenged animals compared to sham animals. PBA treatment significantly increased the number of entries and time spent in the platform quadrant during the probe trial, which indicates the retention of spatial cognition. The swimming paths on track plot during the probe trial suggest that animals find the platform and lack of platform attracts an increased number of entries and time spent in the platform zone. These findings from the MWM task suggest that PBA potentially ameliorates deficits in cognition and memory after stroke.
CONCLUSION
The current study demonstrates that PBA has potential in ameliorating the learning, memory and cognitive deficits associated with experimental ischemic stroke. Since PBA is already used clinically to treat urea cycle disorders, it may be repurposed for the use of post-stroke cognitive impairment.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
We thank National Institute of Pharmaceutical Education and Research (NIPER) and Department of Pharmaceuticals, Ministry of Chemicals and Fertilizers, Government of India for financial support.
Conflicts of interest
There are no Conflict of Interest.
References
- Rodent models of ischemic stroke: A useful tool for stroke drug development. Curr Pharm Des. 2008;14:359-70.
- [CrossRef] [PubMed] [Google Scholar]
- Chronic cerebral hypoperfusion induces post-stroke dementia following acute ischemic stroke in rats. J Neuroinflammation. 2017;14:216.
- [CrossRef] [PubMed] [Google Scholar]
- Post-stroke cognitive impairment: Epidemiology, mechanisms and management. Ann Transl Med. 2014;2:80.
- [Google Scholar]
- Prevalence of poststroke cognitive impairment. Stroke. 2013;44:138-45.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular contributions to cognitive impairment and dementia. Stroke. 2011;42:2672-713.
- [CrossRef] [PubMed] [Google Scholar]
- Cell death and endoplasmic reticulum stress: Disease relevance and therapeutic opportunities. Nat Rev Drug Discov. 2008;7:1013.
- [CrossRef] [PubMed] [Google Scholar]
- Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep. 2006;7:880-5.
- [CrossRef] [PubMed] [Google Scholar]
- Endoplasmic reticulum stress plays critical role in brain damage after cerebral ischemia/reperfusion in rats. Neurotox Res. 2010;17:189.
- [CrossRef] [PubMed] [Google Scholar]
- ER stress in Alzheimer's disease: A novel neuronal trigger for inflammation and Alzheimer's pathology. J Neuroinflammation. 2009;6:41.
- [CrossRef] [PubMed] [Google Scholar]
- Endoplasmic reticulum stress-mediated hippocampal neuron apoptosis involved in diabetic cognitive impairment. Biomed Res Int. 2013;2013:924327.
- [CrossRef] [PubMed] [Google Scholar]
- Endoplasmic reticulum stress mediates distinct impacts of sevoflurane on different subfields of immature hippocampus. J Neurochem. 2017;142:272-85.
- [CrossRef] [PubMed] [Google Scholar]
- Sodium 4-phenylbutyrate protects against cerebral ischemic injury. Mol Pharmacol. 2004;66:899-908.
- [CrossRef] [PubMed] [Google Scholar]
- 4-Phenylbutyric acid attenuates pancreatic beta-cell injury in rats with experimental severe acute pancreatitis. Int J Endocrinol. 2016;2016:1-11.
- [CrossRef] [PubMed] [Google Scholar]
- Sodium phenylbutyrate controls neuroinflammatory and antioxidant activities and protects dopaminergic neurons in mouse models of Parkinson's disease. PLoS One. 2012;7:e38113.
- [CrossRef] [PubMed] [Google Scholar]
- Sodium 4-phenylbutyrate protects against spinal cord ischemia by inhibition of endoplasmic reticulum stress. J Vasc Surg. 2010;52:1580-6.
- [CrossRef] [PubMed] [Google Scholar]
- Endoplasmic reticulum stress inhibition protects steatotic and non-steatotic livers in partial hepatectomy under ischemia-reperfusion. Cell Death Dis. 2010;1:e52.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of 4-phenyl butyric acid on high glucose-induced alterations in dorsal root ganglion neurons. Neurosci Lett. 2016;635:83-9.
- [CrossRef] [PubMed] [Google Scholar]
- Phenylbutyric acid protects against spatial memory deficits in a model of repeated electroconvulsive therapy. Curr Neurovasc Res. 2014;11:156-67.
- [CrossRef] [PubMed] [Google Scholar]
- Phenylbutyrate ameliorates cognitive deficit and reduces tau pathology in an Alzheimer's disease mouse model. Neuropsychopharmacology. 2009;34:1721.
- [CrossRef] [PubMed] [Google Scholar]
- Sodium phenylbutyrate ameliorates focal cerebral ischemic/reperfusion injury associated with comorbid Type 2 diabetes by reducing endoplasmic reticulum stress and DNA fragmentation. Behav Brain Res. 2011;225:110-6.
- [CrossRef] [PubMed] [Google Scholar]
- Neuroprotective effects of 6-hydroxy-2, 5, 7, 8-tetramethylchroman-2-carboxylic acid (trolox), an antioxidant in middle cerebral artery occlusion induced focal cerebral ischemia in rats. Neurol Res. 2007;29:304-9.
- [CrossRef] [PubMed] [Google Scholar]
- Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke. 1989;20:84-91.
- [CrossRef] [PubMed] [Google Scholar]
- Neurological and behavioral outcomes of focal cerebral ischemia in rats. Stroke. 1992;23:267-72.
- [CrossRef] [PubMed] [Google Scholar]
- Amelioration of diabetes-induced cognitive deficits by GSK-3β inhibition is attributed to modulation of neurotransmitters and neuroinflammation. Mol Neurobiol. 2014;50:390-405.
- [CrossRef] [PubMed] [Google Scholar]
- Applications of the Morris water maze in the study of learning and memory. Brain Res Rev. 2001;36:60-90.
- [CrossRef] [Google Scholar]
- Enriched environment improves post-stroke cognitive impairment and inhibits neuroinflammation and oxidative stress by activating Nrf2-ARE pathway. Int J Neurosci 2020:1-10.
- [CrossRef] [Google Scholar]
- Long-term post-stroke changes include myelin loss, specific deficits in sensory and motor behaviors and complex cognitive impairment detected using active place avoidance. PLoS One. 2013;8:e57503.
- [CrossRef] [PubMed] [Google Scholar]
- Epigallocatechin-3-gallate reduces neuronal apoptosis in rats after middle cerebral artery occlusion injury via pi3k/akt/enos signaling pathway. Biomed Res Int. 2018;2018:6473580.
- [CrossRef] [PubMed] [Google Scholar]
- Epigenetic mechanisms of neurodegenerative diseases and acute brain injury. Neurochem Int. 2020;133:104642.
- [CrossRef] [PubMed] [Google Scholar]
- Neuroprotective effect of dimethyl fumarate on cognitive impairment induced by ischemic stroke. Ann Transl Med. 2020;8:375.
- [CrossRef] [PubMed] [Google Scholar]
- Hemopexin alleviates cognitive dysfunction after focal cerebral ischemiareperfusion injury in rats. BMC Anesthesiol. 2019;19:13.
- [CrossRef] [PubMed] [Google Scholar]
- Hydroxysafflor yellow a (HSYA) improves learning and memory in cerebral ischemia reperfusion-injured rats via recovering synaptic plasticity in the hippocampus. Front Cell Neurosci. 2018;12:371.
- [CrossRef] [PubMed] [Google Scholar]






