Translate this page into:
Oral curcumin phytosome supplementation improves anthropometric measures of adiposity and enhances endothelial function in rats on a high-fat-diet regimen
*Corresponding author: Rima Abdul Razzak, Department of Physiology, Arabian Gulf University, Manama, Bahrain. reemala@agu.edu.bh
-
Received: ,
Accepted: ,
How to cite this article: Razzak RA, Khan MN, Marwani A. Oral curcumin phytosome supplementation improves anthropometric measures of adiposity and enhances endothelial function in rats on a high-fat-diet regimen. Indian J Physiol Pharmacol. 2023;67:251-61. doi: 10.25259/IJPP_537_2022
Abstract
Objectives:
Curcumin has a protective role in endothelial function and nitric oxide (NO) production in animal models of different diseases; however, the role of curcumin on aortic reactivity in rats placed on a high-fat diet (HFD) remains unclear. This study aims to determine whether oral curcumin phytosome supplementation can reduce adiposity and enhance endothelial function.
Materials and Methods:
Rats were assigned to one of three groups: normal diet (ND), HFD for 20 weeks, and HFD supplemented with curcumin phytosome (HFD + Curcumin). Anthropometric measures were recorded weekly for the three groups, until the end of the feeding regimen. After 20 weeks of feeding on HFD, myographic investigations were conducted on thoracic aortic rings dissected from HFD and HFD + Curcumin rats. The response to high potassium chloride (KCl), incremental doses of phenylephrine (Phe) before and after L-NAME treatment, acetylcholine (ACh), or sodium nitroprusside (SNP), was evaluated. ACh-induced relaxation was also assessed in HFD + Curcumin rats, after preincubation with chromium III-mesoporhyrin.
Results:
HFD rats exhibited increased adiposity measures, some of which were negatively correlated with vasorelaxation response to ACh. HFD + Curcumin rats had reduced anthropometric measures, compared to HFD rats. Aortic rings from HFD and HFD + Curcumin rats exhibited comparable contractile responses to KCl and Phe. The difference in contractile response to Phe before and after L-NAME incubation was greater for HFD + Curcumin rats. ACh induced greater vasorelaxant responses in HFD + Curcumin rats. There was no group difference in the relaxant response to SNP. In HD + Curcumin rats, chromium III mesoporphyrin significantly reduced ACh-induced relaxations.
Conclusion:
Oral curcumin phytosome supplementation could reduce adiposity in rats placed on an HFD and may have enhanced basal and stimulated NO release from the endothelium, and heme oxygenase-1 may partly mediate this curcumin protective role. This study provides evidence that this curcumin formulation, taken as a daily supplement, may be effective in providing some protection against adiposity-associated adverse cardiovascular disorders.
Keywords
High-fat diet
Adiposity
Curcumin phytosome
Endothelial function
INTRODUCTION
In human studies, accumulating reports have demonstrated that associated with increased adiposity in overweight and obese adults, there is impairment of endothelium-dependent vasodilation,[1] a characteristic of endothelial dysfunction,[2] partly due to oxidative stress,[3] oxidative inactivation of nitric oxide (NO) and reduction of NO bioavailability.[4,5] In animal models of diet-induced obesity, emerging evidence also suggests that feeding with a high-fat diet (HFD) results in oxidative stress,[6] and HFD-induced reduction in endothelium-dependent vasodilation has been reported for different vascular beds such as the renal artery,[7] mesenteric artery[8] and skeletal muscle arterioles.[9] A relatively more recent study reported that endothelial function in rat thoracic aorta and serum NO both begin to decrease as early as 6 weeks of HFD feeding.[10]
Curcumin is a naturally occurring phenol found in Indian spice turmeric, extracted from the rhizomes of the plant Curcuma longa. There is sufficient evidence that curcumin can reduce oxidative stress in animal models, and myography studies on different vascular beds confirm the protective role of curcumin, whether supplemented by diet[11-13] or added to the control media[14-17] on endothelial function and NO production under different diseases such as diabetes mellitus, hyper-homocysteinemia, or stress conditions such as aging. To the best of our knowledge, whether dietary supplementation of curcumin can have the same beneficial effect on the aorta under HFD-induced adiposity remains unknown.
The observations of endothelial dysfunction in different vascular beds in animals fed with HFD provided a rationale for evaluating if dietary curcumin, administered as a lecithin formulation promotes its proper absorption and tissue distribution,[18,19] could potentiate aortic endothelial function of rats fed on HFD. As such, this study aims to assess whether there is a difference in vasoreactivity of the thoracic aorta between HFD rats and those on the same diet but supplemented daily with curcumin phytosome. Findings in this pre-clinical study, potentially supporting improved aortic endothelial function in curcumin-supplemented rats, would present evidence that this curcumin formulation, when taken as a daily supplement, may be effective in providing some protection against adiposity-associated adverse cardiovascular disorders.
MATERIALS AND METHODS
All experiments were carried out following the internationally accepted guide for the care and use of laboratory animals and were approved by the Research Ethics Committee at our university (Ethical approval No. E03-PI-11/19 September, 2020).
Experimental animals
Seventeen Sprague–Dawley male rats (7–9 weeks old, 200–230 g) were individually housed in cages at 22 ± 2°C temperature while a 12 h on/12 h off light cycle was maintained. All animals had free access to water and were on an ad libitum feeding regimen. A group of 6 rats was on normal chow/diet (ND), and a nutrient-matched semi-pure HFD was provided for 20 weeks to induce adiposity in another group of 11 rats. Both diets were purchased from Specialty Feeds Pty Ltd, Australia, in the form of 12 mm diameter pellets. The information on the ND and HFD was provided with the shipping baggage from the producing company, and it is also accessible from the company’s website (www.specialtyfeeds.com). The calculated nutritional parameters of the ND and HFD are shown in [Table 1].
| Calculated nutritional parameters | ||
|---|---|---|
| ND Specialty Feeds (SF00-100) |
HFD Specialty Feeds (SF13-092) |
|
| Protein | 19.6% | 25.5% |
| Total fat | 4.2% | 34.5% |
| Crude fibre | 4.6% | 6.0% |
| AD fibre | 7.6% | 6.0% |
| Digestible energy | 14 MJ/kg | 21.7 MJ/kg |
| % Total calculated digestible energy from lipids | 11.0% | 60.10% |
| % Total calculated digestible energy from protein | 23.0% | 20.40% |
| Total saturated fats | 0.73% | 15.18% |
ND: Normal diet, HFD: High fat diet, AD: Acid detergent, SF: Specialty Feeds
Chemicals and drugs
All chemicals were of analytical grade (Merck, Germany). Phenylephrine hydrochloride (Phe), acetylcholine (ACh), NG-nitro-L-arginine methyl ester (L-NAME), and Chromium III Mesoporphyrin Chloride were obtained from Santa Cruz Biotechnology, Inc., (California, USA). When necessary, the Krebs solution was used as a solvent for all drugs.
Curcumin phytosome as Meriva-SF (THORNE RESEARCH) was used in this study. It is a well-absorbed curcumin formula,[18,20] with each capsule containing 500 mg of curcumin phytosome complex. As per some clinical trials, the recommended daily serving of this formulation for humans is 2 capsules, thus providing 1000 mg of curcumin phytosome complex per day (1,000 mg/day, corresponding to 200 mg pure curcumin mixture/day).[21-23]
For a 70 kg person, 200 mg pure curcumin mixture/day would provide 2.86 mg curcumin mixture/kg/day. To deliver the equivalent dose to rats, the animal equivalent dose (AED) was calculated by multiplying the human dose by the rat conversion factor of 6.2.[24] The calculated AED of pure curcumin mixture in this study corresponds to 17.73 mg/kg/day, which was supplemented to rats by providing 88.65 mg curcumin phytosome complex/kg/day. For the whole duration of the HFD regimen (20 weeks), 6 of the 11 rats were daily supplemented, by once-a-day oral gavage, with the curcumin phytosome dissolved in 5 mL of 50% aqueous dimethyl sulphoxide (DMSO). The ND and HFD groups of rats received daily, an equivalent dose/volume of DMSO as well.
Anthropometric measures of adiposity
Individual rat body weights, naso-anal length, and abdominal and thoracic circumference were recorded at the beginning of the experiment and then weekly. Measurements of the level of adiposity at the end of the feeding period include the body weight, body mass index (BMI), abdominal and thoracic circumference, and the adiposity index as defined by the Lee index, and they were expressed as % change relative to the initial value at the beginning of the feeding regimen.
The Lee index was calculated according to the formula described by Lee (1929)[25] as:
The % change in anthropometric measures at the end of the feeding regimen, relative to the initial measure was calculated as:
Preparation of rat aortas and myography study
Only HFD and HFD + Curcumin rats were included in myographic investigations. Rats were anaesthetised with pentobarbital sodium (540 mg/kg ip), the chest wall was opened, and the thoracic aorta was removed and transferred to a Petri dish (on ice) containing Krebs physiological salt solution (composition (mmol/L) — sodium chloride 118.0, sodium bicarbonate 25.0, d-glucose 11.1, potassium chloride 4.72, Calcium chloride dihydrate 2.56, Sodium phosphate monobasic monohydrate 1.13, Magnesium chloride hexahydrate 1.12, (-) ascorbic acid 0.114 and disodium ethylenediaminetetraacetic acid 0.03). Connective tissue and blood were removed and four ring segments (2 mm in length) from each aorta were cut.
The aortic ring segments were mounted in 5 mL organ bath chambers containing Krebs solution and individually gassed with 95% O2 and 5% CO2 and maintained at 37°C. Vascular reactivity was assessed in the thoracic aortic rings using a Danish Myo Technology 4-chamber myograph to measure the isometric tension generated. The wire myograph interface holds four myograph chambers/units, each containing a force transducer and a micrometer, both connected to corresponding hook pins to support the aortic ring inside the chamber. The data was rendered using a data acquisition hardware unit (PowerLab 4/3; ADInstruments, Australia) and LabChart 8 software (ADInstruments, Australia).
After a resting tension of 2 g, the vessel segments were allowed to equilibrate for 1 h. The rings were constricted with 80 mM KCl for 5 min to assess tissue viability and non-receptor-mediated contractile responses. High potassium depolarizes the smooth muscle cell membrane and opens voltage-dependent calcium (Ca2+) channels, resulting in an influx of extracellular Ca2+ and activation of contractile machinery.[26] To assess receptor-mediated contractility, aortic rings were exposed to increasing doses of Phe (10−9–10−5 M), and full concentration-response curves were obtained. The mechanism by which Phe exerts its contractile effect is by directly stimulating the α-adrenergic receptors of the G protein, acting via phospholipase C, increasing the levels of IP3 and resulting in the release of intracellular Ca2+. [27] The contraction elicited with 80 mM KCl was used as a reference value to compare the magnitude of contraction induced by the cumulative addition of phenylephrine.
To confirm endothelial integrity, we added Phe (10−6 M) to the organ bath, and after Phe (10−6 M) produced a stable and sustained contraction in the rat aorta, ACh (10−6 M) was added to the organ bath. In this experiment, more than 70% ACh -induced relaxation was considered to indicate that the endothelium was intact. Rings were then washed thoroughly, pre-constricted with Phe (10−6 M), and when the Phe-induced contraction was stabilised, increasing ACh concentrations were added to the organ bath at 2-min intervals, and cumulative concentration-response curves to ACh (10−9 – 5 × 10−5 M) were constructed. Krebs solution in the organ bath was changed 3 times after each treatment with a drug. For endothelium-independent vasorelaxant responses, aortic rings were pre-constricted with Phe (10−6 M), and sodium nitroprusside (SNP) was incrementally added to the organ bath to give concentrations of 10−10–10−7 M. Vasorelaxant responses to increased concentrations of ACh or SNP were expressed relative to Phe precontraction with 100% relaxation representing basal tension. The half-maximal effective concentration (EC50) for Phe and ACh was calculated by the statistics software by fitting the dose-response data to a sigmoidal curve.
Statistical analysis
All statistical analyses were conducted with the software, OriginPro 2022b (OriginLab, Massachusetts, USA). The data passed normality as assessed by the Kolmogorov and Smirnov test. Statistical significance was calculated with Student’s t-test or Analysis of Variance with Bonferroni post hoc test as appropriate. A paired t-test was used for comparing aortic ring responses before and after drug treatment/incubation. The association between the two data sets was assessed with linear regression analysis. Values were expressed as mean ± standard error. A P < 0.05 was considered to be statistically significant.
RESULTS
Anthropometric measures of adiposity
[Figure 1] illustrates the weekly measured body weight and shows increasing values throughout the 20-week feeding regimen for the three groups of rats. The difference in body weight at the end of 20 weeks of feeding was not significant between ND and HFD rats. However, [Table 2] shows that the percent (%) increase in the anthropometric measures at the end of the feeding regimen, relative to the initial value, was greatest for HFD rats. ND and HFD rats differed significantly on % increases of most anthropometric measures. In comparison to HFD rats, HFD + Curcumin rats exhibited less % increase of measures, but in comparison to ND rats, there was no significant difference for any measure.
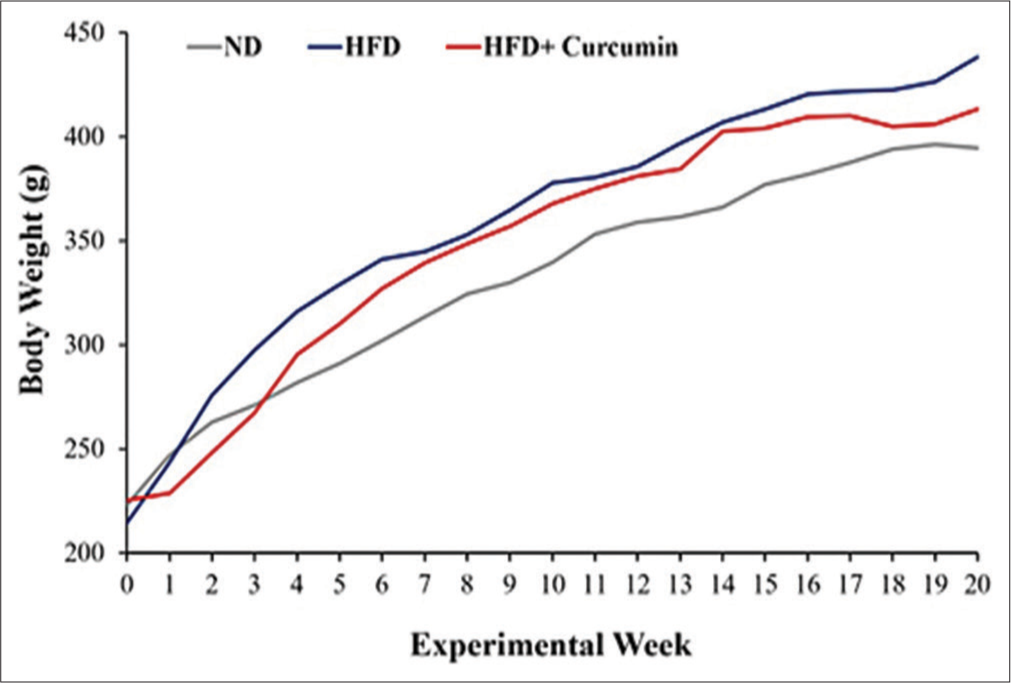
- Weekly progression of rat body weight during the 20 weeks of feeding regimen in the three groups of rats. Normal Diet (ND; n = 6), high fat diet (HFD; n = 5) or HFD + Curcumin (n = 6).
| Mean±SE | ANOVA F | Post hoc analysis (P-value) | |||||
|---|---|---|---|---|---|---|---|
| ND | HFD | HFD+Curcumin | (P-value) | ND vs. HFD | ND vs. HFD+Curcumin |
HFD vs. HFD+Curcumin |
|
| Weight | 91.57±3.04 | 109.44±4.03 | 79.86±3.67 | 16.66 (<0.001) | 0.01 | 0.10 | <0.001 |
| BMI | 22.26±3.19 | 40.79±5.48 | 18.70±2.63 | 9.39 (0.003) | 0.01 | 1.00 | 0.003 |
| Lee index | −1.04±1.09 | 4.59±2.09 | −1.42±0.92 | 5.61 (0.02) | 0.04 | 1.00 | 0.03 |
| Thoracic circ | 50.15±3.57 | 50.27±3.79 | 41.35±2.66 | 2.42 (0.13) | - | - | - |
| Abdominal circ | 38.20±3.12 | 41.41±2.54 | 29.21±2.32 | 5.42 (0.02) | 1.00 | 0.09 | 0.02 |
ND (n=6); HFD (n=5); HFD+Curcumin (n=6). ‘Circ’ refers to circumference. Statistical significance was calculated with ANOVA with Boneferroni post hoc test. P-values in bold font for statistical significance (P<0.05). ND: Normal diet, HFD: High-fat diet, BMI: Body mass index, ANOVA: Analysis of variance, SE: Standard error, Statistically significant difference in weight, BMI and Lee index parameters between ND and HFD as well as HFD and HFD + Curcumin. Also abdominal circumference was statistically smaller in HFD + Curcumin in comparison to HFD rats.
Aortic vasoconstrictor responses
The contraction elicited due to the addition of 80 mM KCl for 5 min was similar in both groups of rats, as the aortic ring tension increased from baseline level to a similar extent (Unpaired t = 0.70, P = 0.49) in both HFD (mean increase: 12.29 ± 0.82 mN, n = 17) and HFD + Curcumin rats (mean increase: 13.10 ± 0.82 mN, n = 19). For Phe-induced contractions, Unpaired t-test analysis reveals that at most Phe concentrations, except 5 × 10−8 M, the contractions as a percentage of KCl-induced contraction were comparable in aortic rings from HFD + Curcumin rats and in those from HFD rats [Figure 2]. The half-maximal EC50 values for Phe were the following: HFD: 7.56 ± 2.26 × 10−8 M; HFD + Curcumin: 1.12 ± 0.33 × 10−7 M.
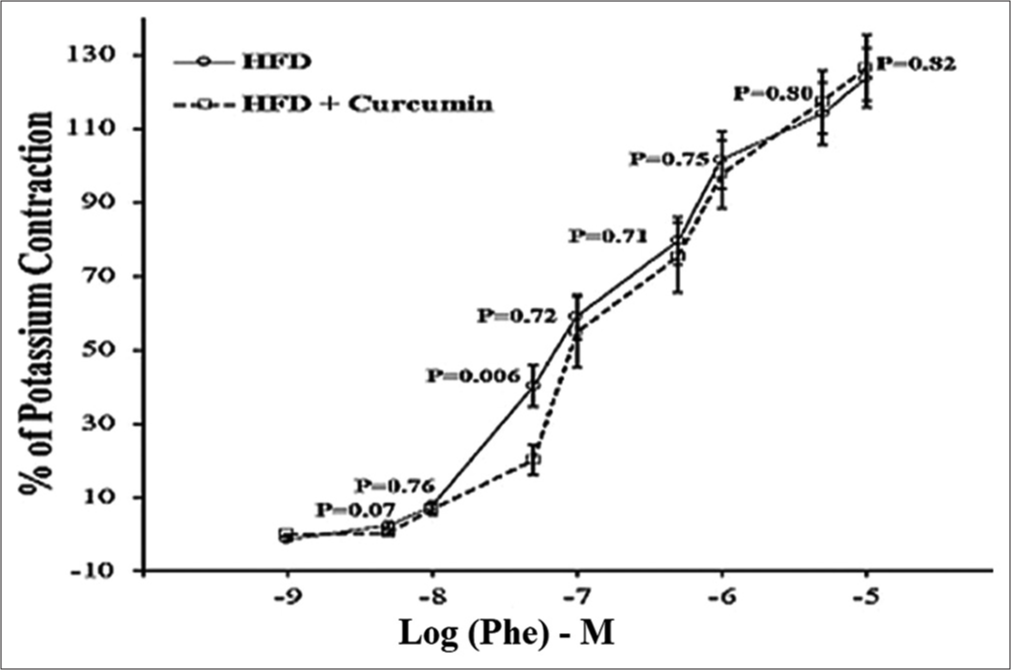
- Contractile responses to increasing concentrations of phenylephrine (Phe) expressed as means ± standard error (SE) in % of the initial reference contraction to potassium chloride (KCl) (80 mM) in 17 aortic rings from 5 high-fat diet (HFD) rats and in 19 aortic rings from 6 HFD + Curcumin rats. P-value obtained with unpaired t-test. M: Molar.
Phe-induced contractions with L-NAME
Several aortic rings from both groups of rats were randomly selected for pre-incubation with L-NAME (10−4 M) for 30 min, to investigate its effect on either Phe-induced or ACh-induced responses. When comparing baseline tension before and after 30 min of L-NAME treatment, L-NAME significantly increased the baseline tone of the aortic rings. The L-NAME-induced increase in tension relative to K+ contraction in rings from HFD rats (78.83 ± 16.03 %; n = 10) was greater by 15%, compared to HFD + Curcumin rats (63.31 ± 14.94 %; n = 9), but the group difference was not statistically significant (t = 0.70, P = 0.49, df = 17).
Among the aortic rings exposed to L-NAME, 7 aortic rings from 4 HFD and aortic rings from 4 HFD + Curcumin rats, with a maximum of 2 rings from each rat, were selected for investigation on the effect of L-NAME on Phe-induced contractions. The aortic rings were exposed to incremental doses of Phe (10−9–10−5 M), before and after pre-treatment with L-NAME for 30 min. [Figure 3] represents the vasoconstrictor responses to increasing concentrations of Phe before and after treatment with L-NAME in (a) HFD and (b) HFD + Curcumin rats. It demonstrates the greater augmentation of Phe-induced contractions by L-NAME in aortic rings from HFD + Curcumin rats in comparison to those from HFD rats.
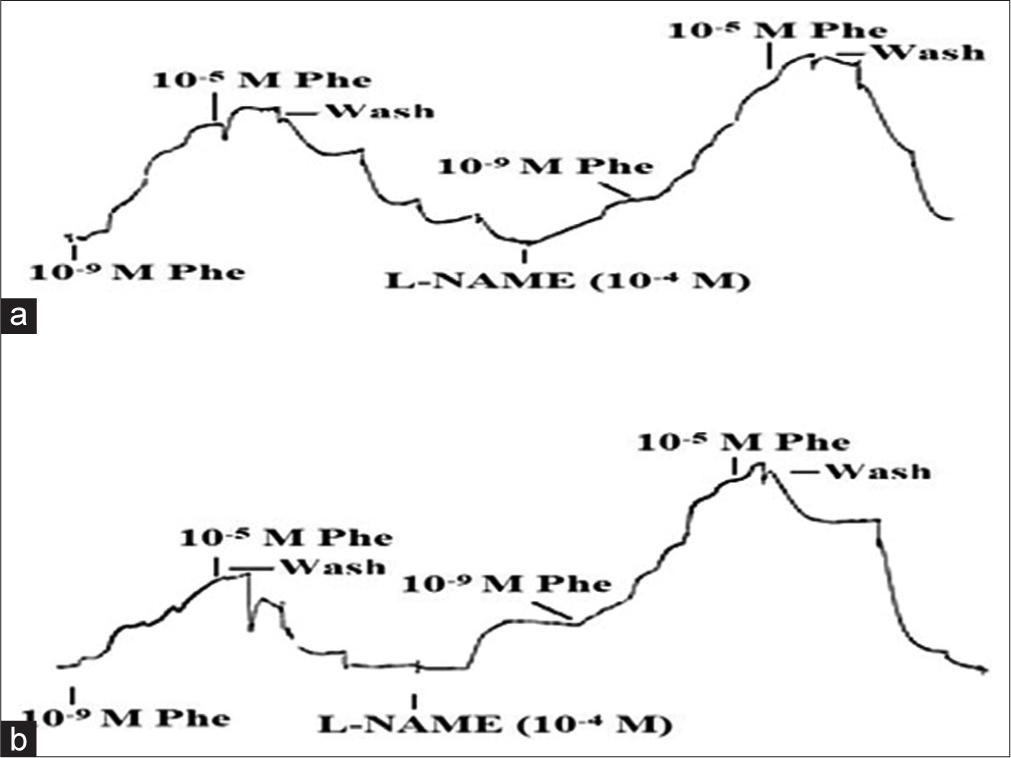
- Trace of aortic ring vasoconstrictor response to increasing concentrations of phenylephrine (Phe) before and after treatment with NG-nitro-L-arginine methyl ester (L-NAME) in (a) High-fat diet (HFD) and (b) HFD + Curcumin rats.
For each aortic ring and each Phe concentration, we calculated the difference in Phe-induced contraction as % of K+- induced contraction, between the L-NAME-incubated and non-incubated conditions. For all Phe concentrations, the difference in % contraction was greater in aortic rings from HFD + Curcumin rats than those from HFD rats, however, statistical significance was not found except for Phe concentration of 5 × 10−8 M [Table 3].
| Log (Phe) – M | (Phe+L-NAME) – (Phe) (%) | t (P-value) | |
|---|---|---|---|
| HFD | HFD+Curcumin | ||
| −9 | 10.10±2.60 | 27.12±12.66 | 1.32 (0.21) |
| −8.3 | 31.97±12.31 | 35.62±10.55 | 0.22 (0.83) |
| −8 | 37.24±9.90 | 40.68±10.31 | 0.23 (0.82) |
| −7.3 | 24.47±7.82 | 61.90±9.44 | 2.87 (0.02) |
| −7 | 32.09±7.69 | 61.78±13.47 | 1.88 (0.09) |
| −6.3 | 56.91±9.02 | 97.48±33.0 | 1.19 (0.26) |
| −6 | 55.35±12.05 | 97.91±32.10 | 1.24 (0.24) |
| −5.3 | 66.16±12.08 | 96.74±39.54 | 0.70 (0.50) |
| −5 | 62.60±18.30 | 89.83±35.30 | 0.67 (0.52) |
Each point is the mean+standard error of ‘n’ aortic rings.
HFD: n=7; HFD+Curcumin: n=7. P-value with Unpaired t-test. P values in bold font for statistical significance (P<0.05). HFD: High-fat diet, L-NAME: NG-nitro-L-arginine methyl ester, Log (Phe) – M: Log (Phenylephrine)-Molar, Phe: Phenylephrine
Aortic vasorelaxation
To detect whether greater adiposity in HFD rats, at the end of 20 weeks of feeding regimen, was associated with worse aortic endothelial function, we assessed the vasorelaxant response to ACh (10−7–10−5 M) after constriction of the aortic rings by Phe and then evaluated the correlation between the ACh-induced relaxation with the anthropometric measures of adiposity. We found evidence of a negative correlation between the vasorelaxation response at some concentrations of ACh and certain anthropometric measures [Figure 4]. The greater the % weight gain or the % increase in BMI for the rat, the less vasorelaxation to ACh in several aortic segments obtained from that rat.
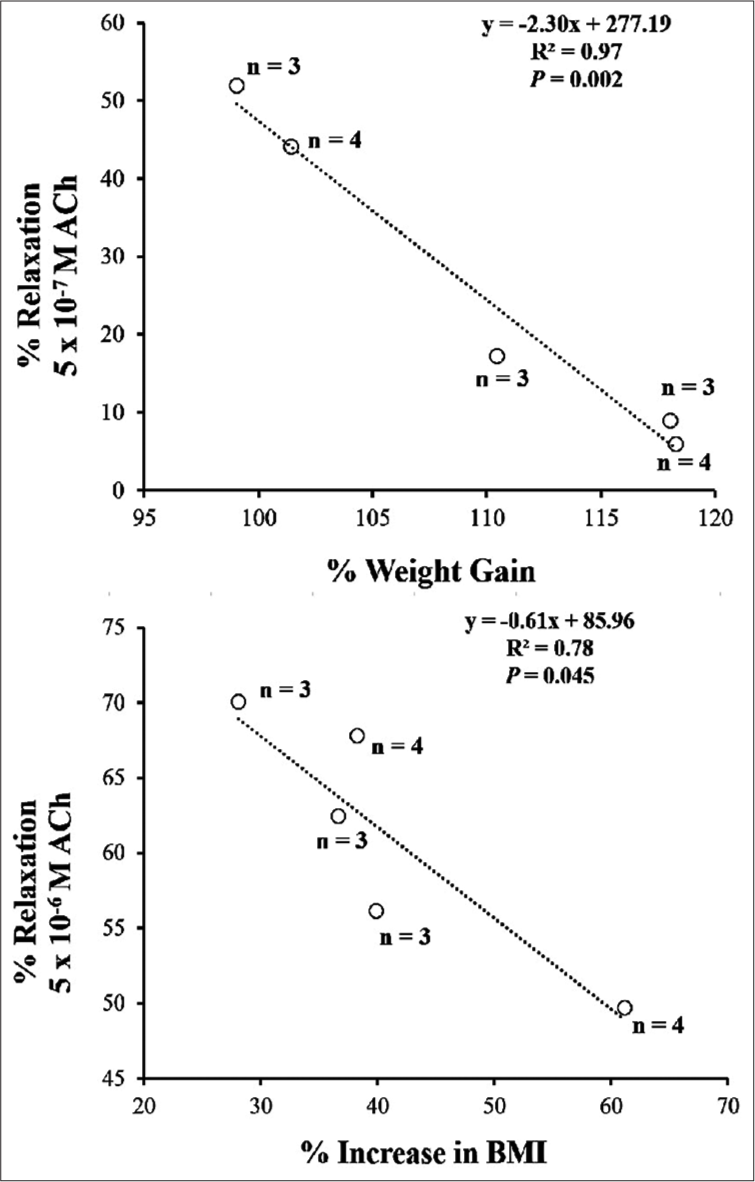
- Linear regression plots between vasorelaxation responses to acetylcholine and certain anthropometric measures of adiposity for the five high-fat diet rats. ‘n’ represents the number of aortic rings from each rat. ACh: acetylcholine; BMI: Body mass index.
[Figure 5] is a trace of the aortic ring vasorelaxant response to increasing concentrations of ACh upon precontraction by Phe (10−6 M) in an aortic ring from (a) HFD and (b) HFD + Curcumin rat. ACh showed a relaxant effect on Phe-induced contraction in a concentration-dependent manner in aortic rings from both groups of rats. In the ring from the HFD rat, even the high ACh concentration of 10−4 M did not cause complete (100%) relaxation, while in the ring from the HFD + Curcumin rat, a concentration of 5 × 10−7 M ACh could bring the tension to pre-constriction baseline level.
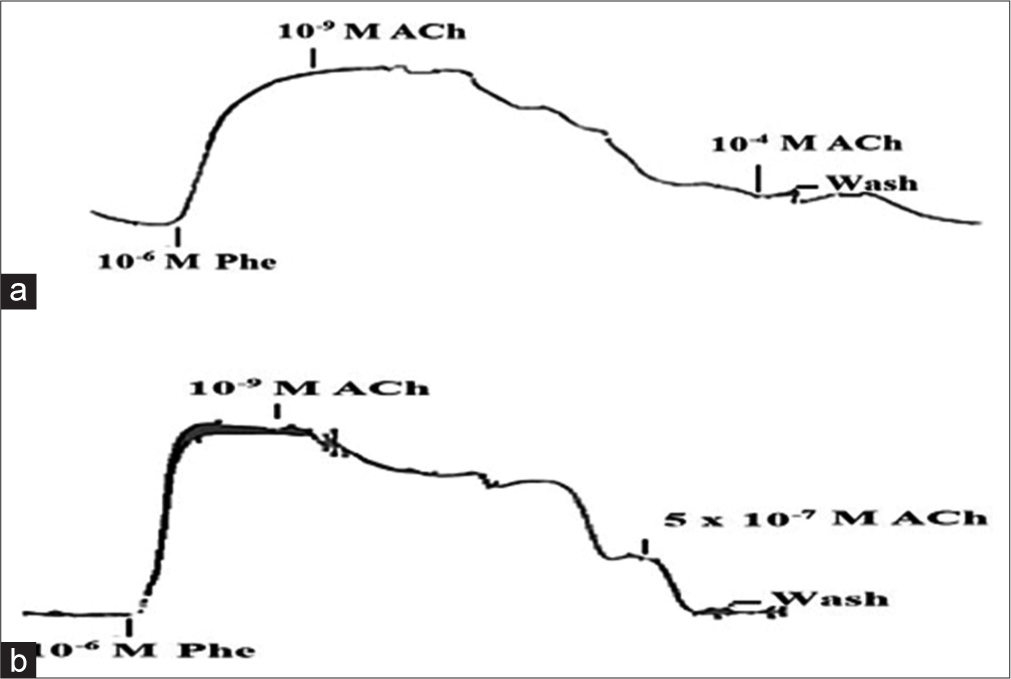
- An example of aortic ring vasorelaxant response to increasing concentrations of acetylcholine (ACh) upon precontraction by Phe (10-6 M) in (a) High-fat diet (HFD) and (b) HFD + Curcumin rats. M: Molar concentration; ACh: Acetylcholine; Phe: phenylephrine.
The concentration-response curves in [Figure 6a] show that vasorelaxation by ACh was greater for aortic rings from the HFD + Curcumin rats compared to those from HFD rats, but only reached a significant difference for ACh concentrations of 10−6 M or greater. The maximum vasorelaxation produced by ACh in HFD and HFD + curcumin rats was 66.41 ± 3.54% and 80.89 ± 5.19%, respectively. The ACh concentration that produced the greatest group difference in percent relaxation (20.61%) was 10−6 M. The EC50 values for ACh were the following: HFD: 1.44 ± 0.54 × 10−6 M; HFD + Curcumin: 9.05 ± 2.62 × 10−7 M. The influence of NO on relaxations to ACh was addressed by pre-treating some aortic rings from HFD and HFD + Curcumin rats with the NO synthase (NOS) inhibitor, L-NAME (10−4 M, 30 min incubation). L-NAME completely abolished the vasodilatory response in both groups of rats.
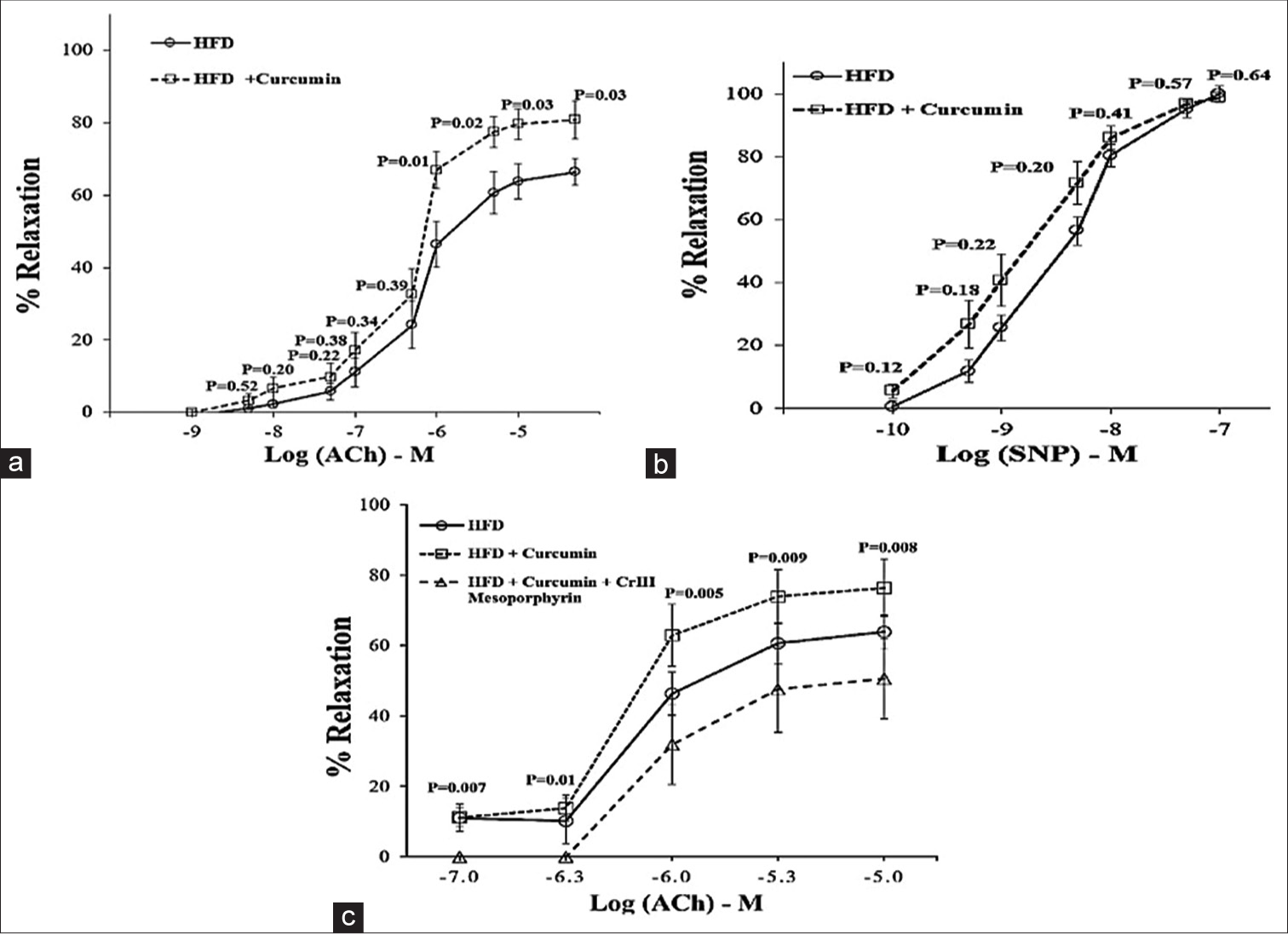
- (a) % Relaxation from Phe (10-6 M)-induced contraction by addition of cumulative concentrations of acetylcholine (ACh) to aortic rings from high-fat diet (HFD) and HFD + Curcumin rats. HFD: n = 17; HFD + Curcumin: n = 18. (b) Effect of increasing sodium nitroprusside (SNP) concentrations on % relaxations from phenylephrine (Phe) (10-6 M)-induced tension in aortic rings from either HFD or HFD + Curcumin rats. P-value with unpaired t-test. (c) Effect of chromium (Cr) III Mesoporphyrin incubation on vasorelaxation responses to ACh in HFD rats supplemented with curcumin. n = 6 aortic rings for the HFD + Curcumin aortic rings. P-value with Paired t-test for comparing HFD + Curcumin with HFD + Curcumin + Cr III Mesoporphyrin responses. Each point is the mean + standard error of ‘n’ observations.
7 aortic rings from 4 HFD rats (a maximum of 2 rings from each rat), exhibited a relaxing response to SNP in a concentration-dependent manner and comparable to that of 7 rings from 4 HFD + Curcumin rats [Figure 6b] for all SNP concentrations, indicating that curcumin did not affect the endothelium-independent relaxation in aortic rings from HFD rats. Incubation of 6 HFD + Curcumin aortic rings (3 rats with 2 rings from each rat) with chromium mesoporphyrin chloride (10−4 M for 60 min), a competitive inhibitor of HO-1, significantly reduced the ACh-induced vasorelaxations to levels even lower than those for HFD rats [Figure 6c]. The EC50 values for ACh were the following: HFD: 2.96 ± 1.36 × 10−6 M; HFD + Curcumin: 1.35 ± 0.51 × 10−6 M; HFD + Curcumin + Cr III Mesoporphyrin: 7.15 ± 3.62 × 10−6 M.
DISCUSSION
To the best of our knowledge, the current study is one of a few that investigated the role of oral supplementation of a phytosome curcumin formulation in anthropometric modulation and its effect on endothelial function in HFD-induced rats. The originality of the study is that because the curcumin was given as an oral supplement for many weeks, rather than being added to the organ bath, myographic investigations on aortic vasoreactivity may capture more realistic differences in endothelial function between HFD rats and HFD-Cur rats.
We investigated the effect of a HFD for 20 weeks on some anthropometric measures of adiposity in rats at the end of the feeding regimen, with the purpose of proving that this diet used could induce some degree of adiposity. More importantly, we explored whether a dietary curcumin phytosome formulation could reduce these adiposity measures and influence rat aortic responses to some vasoactive substances.
With the HFD rats having a weight gain relative to the initial weight of approximately 18% more than the ND rats, this model was not as extremely obese as some of the genetically obese rodent models. The weight gain difference between ND and HFD rats is more consistent with subjects that are overweight.[28] Nevertheless, the significantly greater increase in weight, BMI, and Lee adiposity index after 20 weeks of feeding on the HFD, compared to the ND rats indicates that the HFD was effective in inducing some degree of adiposity. Even though daily food consumption was not measured, the consistently higher weekly body weights in HFD rats, compared to ND rats, indicate a more sizeable weekly energy intake, most probably due to the greater amount of digestible energy from fat in the HFD in comparison to the ND. This is consistent with another study reporting that several weeks into a regimen of a diet with a high fat content can lead to increased adiposity in rodents.[29]
Even though the rats on the HFD were not categorised as obese at the end of the 20-week feeding regimen, a negative consequence of the observed increase in adiposity on endothelial function was demonstrated by the poorer vasorelaxation responses to ACh with greater adiposity measures related to the percentage of weight gain and increase in BMI. Such findings indicate adiposity-dependent impairment of endothelium-dependent vasorelaxation to ACh and are in line with those reported by,[10] where even 6 weeks of HFD could induce endothelial dysfunction. However, in the current study, the vasorelaxation response was not associated with an increase in abdominal circumference, a measure of central obesity in rats.
Our finding that phytosomal curcumin supplemented with HFD rats could reduce some anthropometric measures of adiposity to normal values is in line with some clinical studies,[30] and specifically with those[31,32] reporting that a phytosomal curcumin formulation (Meriva-SF) significantly improves anthropometric indices in obese individuals. Even for overweight individuals, a bioavailable form of curcumin has been reported to improve weight management.[33]
Despite the chemical instability of curcumin at intestinal pH values, its low water solubility, poor bioavailability, and quick conjugation and excretion,[34,35] complexation of curcumin with phospholipids provides a delivery format that can induce its optimal absorption and tissue distribution.[18,19] Indeed, animal studies have reported a more than 20-fold increase in the absorption of curcumin from Meriva as compared to unformulated natural curcumin.[20] This formulation has also accumulated significant clinical documentation of efficacy for the management and treatment of osteoarthritis[21] and diabetic microangiopathy/retinopathy,[22] for reduction of neuropathic pain,[36] and as a complementary therapy, improving the efficacy of anti-cancer drugs.[37]
A possible mechanism of curcumin’s effect of reducing anthropometric measurements could be related to the modulation of appetite, but we cannot be certain in the current study, as the amount of daily food consumption was not measured. This was added as one limitation of the study. So far, the more commonly accepted theories on curcumin’s weight loss potential is related to its effect in attenuating adipogenesis,[38,39] increasing metabolic rate and energy expenditure,[40] reducing oxidative stress[41] and increasing insulin sensitivity.[42,43] A more recent study on high-fat-diet mice reported the involvement of gut microbiota in curcumin’s effect of improving insulin sensitivity.[44]
The current results from myographic investigations indicate a lack of effect of curcumin on contractile responses in the rat thoracic aorta, whether they were non-receptor or receptor mediated, by KCl or Phe, respectively. The absence of group significant difference in the contractile response to Phe at most concentrations, but one (5 × 10−8 M), indicates that overall, curcumin supplementation did not significantly affect the response of the aortic ring to Phe, nor the percentage of contraction to maximum Phe concentration (10−5 M), relative to KCl contraction. Our results disagree with the only myographic study on thoracic aorta in C57BL/6 mice,[45] in which 6-week dietary curcumin supplementation induced a significantly increased vascular contractility to KCl or Phe. The discrepancy in findings between the two studies could be related to differences in the levels of adiposity of the animals used, or to differences in mechanical and pharmacological properties of mouse and rat aorta.[46]
In the current study, L-NAME increased the pressor effect of Phe for both groups of rats, suggesting that normally NO release suppresses the contraction in the aorta. Despite the absence of a statistically significant difference between the two groups of rats, L-NAME enhanced the contraction to Phe to a greater extent in HFD + Curcumin rats in comparison to those from HFD rats. As increased responses to Phe after the addition of L-NAME are taken as a measure of basal NO release,[47,48] this suggests that curcumin phytosome supplementation can probably increase basal NO release in the aorta from rats placed on a HFD.
There has been extensive literature on the effect of dietary curcumin on endothelial function in humans, usually measured noninvasively with high-resolution ultrasound by assessing flow-mediated dilation (FMD) at the brachial artery, with a meta-analysis study[49] of several randomised clinical trials indicating a significant effect of curcumin preparations to increase the FMD compared to placebo and thus endothelial function. Such findings have also been confirmed by animal ex vivo myographic studies on other vascular beds, such as cerebral arteries in ageing[50] or resistance mesenteric vessels in Rett syndrome.[12]
As far as we know, no myographic studies have examined the effect of oral curcumin supplementation on aortic vasodilatory responses in aortic rings from rats placed on an HFD. Our results point to improved vasodilatory response to ACh in adipose HFD-induced rats supplemented with curcumin phytosome, in comparison to those not supplemented, especially at concentrations equal to or greater than 10−6 M.
Abolishment of vasodilatory responses to ACh by L-NAME confirms NO mediation of ACh-induced dilation in aortic rings from both groups, and comparable SNP-induced dilation in HFD and HFD + Curcumin rats suggests that vascular smooth muscle responsiveness to NO was not affected by curcumin, and proves the endothelial-specific effects of curcumin. Since responses to ACh are considered a measure of stimulated NO release,[47] the greater vasodilatory responses in curcumin-supplemented rats indicate that dietary curcumin phytosome may increase stimulated NO release in HFD-induced adiposity. This is expected since it is established that curcumin exerts such a protective role on the endothelium by stabilising the activity of NOS, which is required for the synthesis of endothelial NO[49,51] that indirectly exerts a relaxant effect in the vascular smooth muscle cells.[52]
The attenuation of vasorelaxation response to ACh in curcumin-supplemented HFD rats to HFD levels by the heme oxygenase 1 (HO-1) inhibitor, Cr III-Mesoporphyrin, indicates that Cr III-Mesoporphyrin offset the protective role of curcumin and that HO-1 may partly be involved in curcumin phytosome enhancement of vasorelaxation in the aorta of HFD rats. Indeed, previous studies have established that curcumin is a potent inducer of HO-1 in vascular endothelial cells,[53,54] and there is accumulating evidence suggesting that HO-1 can improve vascular function, at least in part, by enhancing eNOS activation[55] and compensating for the loss of NO bioavailability.[56]
CONCLUSION
In this study, the mean arterial blood pressure, and levels of metabolic parameters such as serum insulin, cholesterol, triglyceride, and glucose were not measured in the rats. It would have been preferable to evaluate how phytosomal curcumin supplementation would influence such parameters along the 20 weeks of an HFD feeding regimen. Furthermore, the consumption of food by each rat was not monitored. Despite these limitations, this study demonstrates the following: Rats fed on an HFD develop increased adiposity after 20 weeks of this feeding regimen, and the greater adiposity, the worse the endothelial function. Oral supplementation of curcumin in phytosomal format did reduce some anthropometric measures of adiposity in rats placed on the HFD regimen. It may have also enhanced the basal release of NO in the aorta of HFD rats. Curcumin phytosome did not modulate endothelium-independent dilations; however, it enhanced endothelium-dependent dilations in the aorta from HFD rats, probably indicating to increase in stimulated NO release. Such an effect of curcumin may be partly mediated by HO-1. All these findings suggest a possible role for curcumin phytosome supplementation in enhancing endothelial function in rats with increased adiposity, partly by increasing NO release. Such improvement of vasorelaxation response would indicate that even while being on an HFD, phytosomal curcumin supplementation may exert protective effects on the arterial system, allowing for better vasodilatory responses, and in turn reducing the risk of elevation of systemic arterial blood pressure above normal.
Author contributions
Dr. Rima Abdul Razzak participated in the conception, and design of the article, from the acquisition of data to bibliographic search, analysis, writing, illustration, data interpretation, and critical discussion. Mohammad Nadir Khan participated in the dissection of the aortic tissue, mounting of the aortic rings, and preparation of solutions, chemicals, and drugs. Amar Marwani participated in animal care, anthropometric measurements, and feeding the rats according to instructions.
Ethical approval
Animal experiments conform to internationally accepted standards and have been approved by the Research and Ethics Committee at the College of Medicine in our university. (Ethical approval No. E03-PI-11/19 September, 2020).
Declaration of patient consent
No human subjects (patients) were included in this study.
Conflicts of interest
The authors declare that there is no conflict of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship
This work was supported by a grant from the Arabian Gulf University (Grant number. G02/AGU 11/19).
References
- Human obesity and endothelium-dependent responsiveness. Br J Pharmacol. 2012;165:561-73.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular endothelial dysfunction and pharmacological treatment. World J Cardiol. 2015;7:719-41.
- [CrossRef] [PubMed] [Google Scholar]
- Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest. 2004;114:1752-61.
- [CrossRef] [PubMed] [Google Scholar]
- Obesity and body fat distribution induce endothelial dysfunction by oxidative stress: Protective effect of vitamin C. Diabetes. 2001;50:159-65.
- [CrossRef] [PubMed] [Google Scholar]
- Obesity, atherosclerosis and the vascular endothelium: Mechanisms of reduced nitric oxide bioavailability in obese humans. Int J Obes Relat Metab Disord. 2002;26:754-64.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of mechanisms involved in impaired vascular reactivity between high sucrose and high fat diets in rats. Nutr Metab (Lond). 2010;7:48.
- [CrossRef] [PubMed] [Google Scholar]
- High-fat feeding reduces endothelium-dependent vasodilation in rats: Differential mechanisms for saturated and unsaturated fatty acids? Clin Exp Pharmacol Physiol. 2006;33:708-13.
- [CrossRef] [PubMed] [Google Scholar]
- Differential vascular dysfunction in response to diets of differing macronutrient composition: A phenomenonological study. Nutr Metab (Lond). 2007;4:15.
- [CrossRef] [PubMed] [Google Scholar]
- High-fat diet-induced reduction in nitric oxide-dependent arteriolar dilation in rats: Role of xanthine oxidase-derived superoxide anion. Am J Physiol Heart Circ Physiol. 2006;291:H2107-15.
- [CrossRef] [PubMed] [Google Scholar]
- Endothelial dysfunction and inflammation precede elevations in blood pressure induced by a high-fat diet. Arq Bras Cardiol. 2018;110:558-67.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin increases vasodilatory effect of cilostazol in diabetic rat aorta. Indian J Exp Biol. 2012;50:128-32.
- [Google Scholar]
- Vascular dysfunction in a mouse model of Rett syndrome and effects of curcumin treatment. PLoS One. 2013;8:e64863.
- [CrossRef] [PubMed] [Google Scholar]
- Dietary curcumin ameliorates aging-related cerebrovascular dysfunction through the AMPK/uncoupling protein 2 pathway. Cell Physiol Biochem. 2013;32:1167-77.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin blocks homocysteine-induced endothelial dysfunction in porcine coronary arteries. J Vasc Surg. 2004;40:1216-22.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin ameliorates high glucose-induced acute vascular endothelial dysfunction in rat thoracic aorta. Clin Exp Pharmacol Physiol. 2009;36:1177-82.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin attenuates fructose-induced vascular dysfunction of isolated rat thoracic aorta rings. Pharm Biol. 2014;52:972-7.
- [CrossRef] [PubMed] [Google Scholar]
- The curcumin-induced vasorelaxation in rat superior mesenteric arteries. Ann Vasc Surg. 2018;48:233-40.
- [CrossRef] [PubMed] [Google Scholar]
- Phytosomal curcumin: A review of pharmacokinetic, experimental and clinical studies. Biomed Pharmacother. 2017;85:102-12.
- [CrossRef] [PubMed] [Google Scholar]
- Formulation of more efficacious curcumin delivery systems using colloid science: Enhanced solubility, stability, and bioavailability. Molecules. 2020;25:2791.
- [CrossRef] [PubMed] [Google Scholar]
- Comparative absorption of a standardized curcuminoid mixture and its lecithin formulation. J Nat Prod. 2011;74:664-9.
- [CrossRef] [PubMed] [Google Scholar]
- Efficacy and safety of Meriva®, a curcuminphosphatidylcholine complex, during extended administration in osteoarthritis patients. Altern Med Rev. 2010;15:337-44.
- [Google Scholar]
- Meriva®, a lecithinized curcumin delivery system, in diabetic microangiopathy and retinopathy. Panminerva Med. 2012;54:11-6.
- [Google Scholar]
- Reduction of delayed onset muscle soreness by a novel curcumin delivery system (Meriva®): A randomised, placebo-controlled trial. J Int Soc Sports Nutr. 2014;11:31.
- [CrossRef] [PubMed] [Google Scholar]
- Dose translation between laboratory animals and human in preclinical and clinical phases of drug development. Drug Dev Res. 2018;79:373-82.
- [CrossRef] [PubMed] [Google Scholar]
- Determination of the surface area of the white rat with its application to the expression of metabolic results. Am J Physiol Legacy Content. 1929;89:24-33.
- [CrossRef] [Google Scholar]
- Potassium channels in regulation of vascular smooth muscle contraction and growth. Adv Pharmacol. 2017;78:89-144.
- [CrossRef] [PubMed] [Google Scholar]
- Evolving mechanisms of vascular smooth muscle contraction highlight key targets in vascular disease. Biochem Pharmacol. 2018;153:91-122.
- [CrossRef] [PubMed] [Google Scholar]
- Diet-induced obesity in Sprague-Dawley rats causes microvascular and neural dysfunction. Diabetes Metab Res Rev. 2010;26:306-18.
- [CrossRef] [PubMed] [Google Scholar]
- High-fat diet-induced obesity Rat model: A comparison between Wistar and Sprague-Dawley Rat. Adipocyte. 2015;5:11-21.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of curcumin supplementation on weight loss and anthropometric indices: An umbrella review and updated meta-analyses of randomized controlled trials. Am J Clin Nutr. 2023;117:1005-16.
- [CrossRef] [PubMed] [Google Scholar]
- The effect of curcumin supplemsentation on anthropometric indices in overweight and obese individuals: A systematic review of randomized controlled trials. Adv Exp Med Biol. 2021;1291:121-37.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin-the nutraceutical with pleiotropic effects? Which cardiometabolic subjects might benefit the most? Front Nutr. 2022;9:865497.
- [CrossRef] [PubMed] [Google Scholar]
- Potential role of bioavailable curcumin in weight loss and omental adipose tissue decrease: Preliminary data of a randomized, controlled trial in overweight people with metabolic syndrome. Preliminary study. Eur Rev Med Pharmacol Sci. 2015;19:4195-202.
- [Google Scholar]
- Beyond yellow curry: Assessing commercial curcumin absorption technologies. J Am Coll Nutr. 2015;34:347-58.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin: A review of its effects on human health. Foods. 2017;6:92.
- [CrossRef] [PubMed] [Google Scholar]
- Safety and efficacy of an add-on therapy with curcumin phytosome and piperine and/or lipoic acid in subjects with a diagnosis of peripheral neuropathy treated with dexibuprofen [Erratum] J Pain Res. 2013;6:641.
- [CrossRef] [PubMed] [Google Scholar]
- Phytosome complex of curcumin as complementary therapy of advanced pancreatic cancer improves safety and efficacy of gemcitabine: Results of a prospective phase II trial. Pharmacol Res. 2018;132:72-9.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin rescues high fat diet-induced obesity and insulin sensitivity in mice through regulating SREBP pathway. Toxicol Appl Pharmacol. 2016;304:99-109.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin attenuates adipogenesis by inducing preadipocyte apoptosis and inhibiting adipocyte differentiation. Nutrients. 2019;11:2307.
- [CrossRef] [PubMed] [Google Scholar]
- Chronic gastrointestinal inflammation induces anxiety-like behavior and alters central nervous system biochemistry in mice. Gastroenterology. 2010;139:2102-12.e1.
- [CrossRef] [PubMed] [Google Scholar]
- Oxidative stress, plant natural antioxidants, and obesity. Int J Mol Sci. 2021;22:1786.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin and insulin resistance-Molecular targets and clinical evidences. Biofactors. 2016;42:561-80.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of curcumin consumption on human chronic diseases: A narrative review of the most recent clinical data. Phytother Res. 2018;32:957-75.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin improves insulin sensitivity in high-fat diet-fed mice through gut microbiota. Nutr Metab (Lond). 2022;19:76.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin enhances vascular contractility via induction of myocardin in mouse smooth muscle cells. Acta Pharmacol Sin. 2017;38:1329-39.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular reactivity of isolated thoracic aorta of the C57BL/6J mouse. J Pharmacol Exp Ther. 2000;294:598-604.
- [Google Scholar]
- Effects of nitric oxide and superoxide on relaxation in human artery and vein. Atherosclerosis. 1997;133:77-86.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular smooth muscle contraction is an independent regulator of endothelial nitric oxide production. Scand Cardiovasc J. 1999;33:33-78.
- [CrossRef] [Google Scholar]
- Curcumin preparations can improve flow-mediated dilation and endothelial function: A meta-analysis. Complement Med Res. 2020;27:272-81.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin supplementation improves vascular endothelial function in healthy middle-aged and older adults by increasing nitric oxide bioavailability and reducing oxidative stress. Aging (Albany NY). 2017;9:187-208.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular nitric oxide: Beyond eNOS. J Pharmacol Sci. 2015;129:83-94.
- [CrossRef] [PubMed] [Google Scholar]
- Curcumin, an antioxidant and anti-inflammatory agent, induces heme oxygenase-1 and protects endothelial cells against oxidative stress. Free Radic Biol Med. 2000;28:1303-12.
- [CrossRef] [PubMed] [Google Scholar]
- Potential therapeutic effects of natural heme oxygenase-1 inducers in cardiovascular diseases. Antioxid Redox Signal. 2013;18:507-21.
- [CrossRef] [PubMed] [Google Scholar]
- Heme oxygenase-1 ameliorates oxidative stress-induced endothelial senescence via regulating endothelial nitric oxide synthase activation and coupling. Aging (Albany NY). 2018;10:1722-44.
- [CrossRef] [PubMed] [Google Scholar]
- Role of heme oxygenase in preserving vascular bioactive NO. Nitric Oxide. 2010;23:251-7.
- [CrossRef] [PubMed] [Google Scholar]






