Translate this page into:
Protective effects of lycopene against adenine-induced chronic renal failure in rats
*Corresponding author: Sandip Patel, Department of Pharmacology, L.M. College of Pharmacy, Ahmedabad, Gujarat, India. patelsandozrx@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Gori P, Patel A, Solanki N, Shah U, Patel V, Patel S. Protective effects of lycopene against adenine-induced chronic renal failure in rats. Indian J Physiol Pharmacol 2021;65(2):74-85.
Abstract
Objectives:
Chronic renal failure (CRF) is a public health concern in both developed and developing countries. Therefore, there is still a need to look for secure and successful agents that can either minimise or prevent CRF from advancing to end-stage renal disorder. This study aimed to assess the effect of lycopene on adenine-induced CRF in the rat.
Materials and Methods:
Animals were divided into five groups (n = 6). Normal control group received normal vehicle, disease control group received orally adenine (50 mg/kg/day), L 100 group received orally lycopene (100 mg/kg/day) + adenine (50 mg/kg/day), L 200 group received orally lycopene (200 mg/kg/day) + adenine (50 mg/kg/day) and L 400 group received orally lycopene (400 mg/kg/day) + adenine (50 mg/kg/day) for 30 days.
Results:
Compared to the control group, the disease control group had decreased bodyweight, food intake and also increased the relative kidney weight and urine output. Adenine-treated group also significantly increased the blood urea nitrogen, serum creatinine, phosphorus, alkaline phosphatase, uric acid, magnesium and reduced the calcium, urine creatinine and urine urea nitrogen. Besides, adenine also gave a positive test of serum C-reactive protein and proteinuria. Histopathologically, adenine caused significant inflammatory changes to renal tissues compared with the normal control group. When administered concomitantly with adenine, lycopene alleviated all the measured adenine-induced physiological, biochemical and histological changes.
Conclusion:
We concluded from this analysis that oral lycopene administration could potentially mitigate the adverse effect of CRF that might be due to their antioxidant and free radical scavenging properties.
Keywords
Chronic renal failure
Lycopene
Adenine
Renal anaemia
Vascular calcification
INTRODUCTION
Chronic kidney disease (CKD) has grown into a serious public health concern and represented a major contributor to a disease burden in the world and affecting more than 200 million populations worldwide, and in India, age-adjusted end-stage renal diseases (ESRDs) incidence rate was 229/million populations as well as more than 100,000 new patients enlist annual renal replacement services.[1,2] CKD was tied to terrible health interventions and drastically reduced the quality of life for the patient. Incidence of morbidity and mortality is increasing year by year in the world and several factors that are responsible for the CKD advancement including obesity, high blood pressure, anaemia, vascular calcification and diabetes.[3] Till now, there is no single drug available which can be used to alleviate kidney function in CKD patients, and existing clinical strategies are mainly restricted to slow down disease progression to the ESRD condition where dialysis or kidney transplantation is the only treatment option.[4] Furthermore, it is firmly necessary to build new therapies or nutritional supplements, especially from natural remedies, either to mitigate the effect of the disease or perhaps to delay as well as accelerate the degradation in the function of the kidney. The critical alternative treatment for CKD was natural remedies. Natural remedies have become increasingly popular in the promotion of medical service and preventing disease and are being used as a traditional or unique treatment of CKD.
It is very well evidenced that oxidative stress, inflammation and apoptosis are involved in the pathophysiology of CKD and its associated complications.[5] The most common pathogenesis characteristic of CKD in ESRD is renal fibrosis. Renal fibrosis is characterised by high infiltration of inflammatory cells, injury of renal tubules, tubulointerstitial fibrosis of renal and degeneration of glomeruli.[6] These features frequently exist in both humans and animals. They are both significant mediators of the disease and have related effects in rat models of chronic renal failure (CRF).[7] Patients and research animals with CRF have elevated plasma concentrations of inflammatory mediators along with C-reactive protein (CRP), tumour necrosis factor (TNF), various cytokines as well as various nitrosative and oxidative stress indicators.[8,9] CRF animal models are essential for studying the fundamental biochemical, physiological and histological mechanisms that influence of CRF for developing and evaluating possible therapeutic substances. Adenine is metabolized to form 2,8 dihydroxyadenine which accumulates in to the renal tissues. Accumulated metabolite of adenine produces structural damages to tubulointerstitial tissue. Adenine induction of CRF in rodents is a common technique for mimic a functional abnormality in people with renal failure.[10,11]
Lycopene is a red-pigmented linear carotenoid and it is a more potent antioxidant naturally present in many fruits and vegetables.[12] It provides a high amount of antioxidants to help avoid various kinds of oxidant damage to tissues and cells. Dietary intake of tomato and tomato-based products is increasing the lycopene level in blood and reduced the oxidative damage of lipids, protein and DNA. Based on these properties, lycopene used as a curative and preventive treatment of various chronic diseases such as aging prevention, cancer prevention and anti-inflammation.[13,14] The past studies demonstrated the possible utility of certain natural products in animal CRF models, such as curcumin, soybean, Bryophyllum calycinum and Achyranthes asparagus, Arabic gum and lemongrass.[15-18] In the continuation of this analysis, we investigated possible potential beneficial effects of lycopene against adenine-induced CRF in rats by estimated various physiological parameters, serum and urinary renal function indices, CRP, anaemia indicator, proteinuria and histopathological parameters.
MATERIALS AND METHODS
Plant materials and chemicals
Adenine was bought from Sisco Research Laboratories Pvt. Ltd., Maharashtra, India. A gift sample of the lycopene was obtained from Medheal Pharmaceuticals, Ahmedabad, India. All chemicals and reagents used were AR grade. Diagnostic kits for the measurement of serum creatinine, calcium, uric acid, phosphorus, magnesium and alkaline phosphatase (ALP) kit were procured from the Coral Clinical System and Tulip Diagnostic Kits Pvt. Ltd., Goa, India. CRP latex test kit and AUTOSPAN Liquid Gold Urea kit were procured from ARKRAY Healthcare Pvt. Ltd., Gujarat, India.
Animals selection
Male Sprague Dawley rats were procured from Zydus Research Centre, Ahmedabad. Animals were housed in polypropylene rat cages as three animals/cage/sex and rice husk used as the bedding material. The animals were acclimatised to standard laboratory conditions for a minimum period of 1 week before commencement of treatment, temperature between the 25 ± 2°C and relative humidity 30–70%, and maintained on a 12 h light and 12 h dark cycle. Rat was fed laboratory rat pallet and pure drinking water supplied. The experimental protocol was approved by the Institutional Animal Ethics Committee (IAEC) and the care of animals was taken as per the guidance of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) (Protocol No: RPCP/ IAEC/2019-20/R1).
Acute oral toxicity study
The study was carried out using male Wistar rats according to the OECD guideline 423. A total of three male rats were used for the study that is, 2000 mg/kg with three animals in group
Experimental design
Induction of CRF
The animals were allowed to acclimatise for 1 week and fed normally before the experimental period. The rats were weighed and divided randomly by weight into a normal control group (6 rats) and adenine groups (40 rats). The rats in the adenine group were administered orally 50 mg/kg adenine (suspended in 0.5% NaCMC solution; 10 rats) once daily for 30 days. The rats in the normal control group were administered the same volume of vehicle (0.5% NaCMC solution). Further, adenine groups were divided in to four subgroups: A disease control group (n = 10), a low dose (100 mg/kg) lycopene group (L100; 10 rats), a medium dose (200 mg/kg) lycopene group (L200; 10 rats) and a high dose (400 mg/kg) lycopene group (L400; 10 rats). Lycopene was suspended in 0.5% NaCMC solution and administered with adenine simultaneously for 30 days. Food intake and body weight of animals were recorded every day during the study.
Sample collection
At the end of the experiment (30 days), all the animals were individually kept in metabolic cages for 24 h to collect urine for analysis. During the collection of the urine, animals provided free exposure to drinking water. After collection of the urine, 1–2 drops of concentrated hydrochloric acid were added and it was placed at 4°C for further examination of various urinary parameters such as proteinuria, urine urea nitrogen (UUN) and creatinine. Blood was collected through the retro-orbital method under the anaesthetic condition for complete blood count and serum biochemical analysis. The serum was isolated at 3000–4000 rpm for 10 min by centrifugation and it was placed at −20°C for the further examination of various serum biochemical parameters such as a creatinine, blood urea nitrogen (BUN), uric acid, calcium, phosphorus, magnesium, ALP and CRP. After collections of blood and urine animal were sacrificed using the high dose of anaesthetic agents and two kidneys were isolated. The kidneys were weighed and fixed for histopathological analysis in 10% neutral-buffered formalin.
Physiological analysis
Food intake and body weight of animals were recorded every day during the study, urine output and relative percentage kidney weight were measured at the end of the experiments.
Biochemical analysis
The concentrations of serum and urinary renal function parameters such as serum creatinine, uric acid, BUN, UUN and urine creatinine, serum vascular calcification components such as calcium, phosphorus, magnesium and ALP were estimated by spectrophotometer using a commercial kit, estimations of anaemia indicator such as red blood cells (RBCs), haemoglobin (HGB) and haematocrit (HCT) were measured using the CELL-DYN Ruby Haematology analyser. Estimations of serum CRP was done by qualitative rapid latex slide test using a commercial kit and CRP score was classified based on the following visual scale: Very strongly reactions; demonstrating wide agglutinated clusters on slide score = +4, strongly reaction; demonstrating wide agglutinated coarse on slide score = +3, moderate reactions; demonstrating moderate agglutination coarse on slide score = +2, weakly reactions; demonstrating small agglutinated finer on slide score = +1 and negative reactions; demonstrating smooth suspension on slide score = 0. Estimations of proteinuria were measured using the heat coagulation test and proteinuria score was classified based on the following visual scale: Clear solution; if a written sheet of paper is put in front of the urine column the test tube, handwritten letters can be read easily through the test tube score = 0, clear solution or mild turbidity; written letters can be read through the test tube very simply score = +1, moderate turbidity; written letters cannot be read through the test tube score = +2, severity turbidity or mild precipitation; none of this can be seen through the test tube score = +3 and precipitation or clots; none of this can be seen through the test tube score = +4.
Histopathology analysis
The kidney was fixed in 10% neutral formalin solution and then it was dehydrated using the ethanol solution followed by cleaning with xylene solution. Further, processed tissue was embedded in paraffin. Later, a 2 μm section of kidney tissue cut from the paraffin block by used the microtome and then it was stained with haematoxylin and eosin (H & E). All kidney part microscopic examinations were performed in a blinded pattern.
Statistical analysis
All the data were analysed by Window software with GraphPad Prism Version 8.0. Moreover, data are expressed as mean ± SEM (n = 6). One-way ANOVA or t-test performed to comparisons between the control group and different treatment groups and P < 0.05 considered to be statistically significant.
RESULTS
Acute oral toxicity study
In this study, single oral dose of lycopene at 2000 mg/kg, animals did not show any mortality and detectable signs of toxicity or behavioural changes and were found to be normal across the 14-day study period. Body weight was constant during the study period.
Effect of lycopene on physiological parameter in adenine-induced CRF in the rat
At the end of the study period, animals received adenine alone, disease control group showed a significant decrease in body weight, food intake and a substantial increase in relative percentage kidney weight and urine output as compared with the normal control group (P < 0.01). Treatment with lycopene produced a significant elevation of body weight, food intake and reduction in relative percentage kidney weight and urine output in a dose-dependent system as compared with adenine-induced disease control group (P < 0.05) [Figure 1].

- Effect of lycopene on physiological parameter in adenine-induced chronic renal failure in the rat. (a) Body weight of animals, (b) Food intake, (c) Relative weight of left kidney, (d) Relative weight of right kidney, (e) Urine output. Columns and vertical bars depict mean±SEM (n=6). NC: Normal control, DC: Diseases control, L 100: 100 mg/kg Lycopene+Adenine, L 200: 200 mg/kg Lycopene+Adenine, L 400: 400 mg/kg Lycopene+Adenine. *P<0.05 compared with disease versus different treatment groups, **P<0.001 compared with disease versus different treatment groups, ***P<0.0001 compared with disease versus different treatment groups, ##P<0.001 compared with normal versus disease groups, ###P<0.0001 compared with normal versus disease groups.
Effect of lycopene on serum and urinary renal function indices in adenine-induced CRF in the rat
The consequence of treatment with adenine, lycopene at three different doses and adenine combination on serum and urinary renal function indices are shown in [Figure 2].
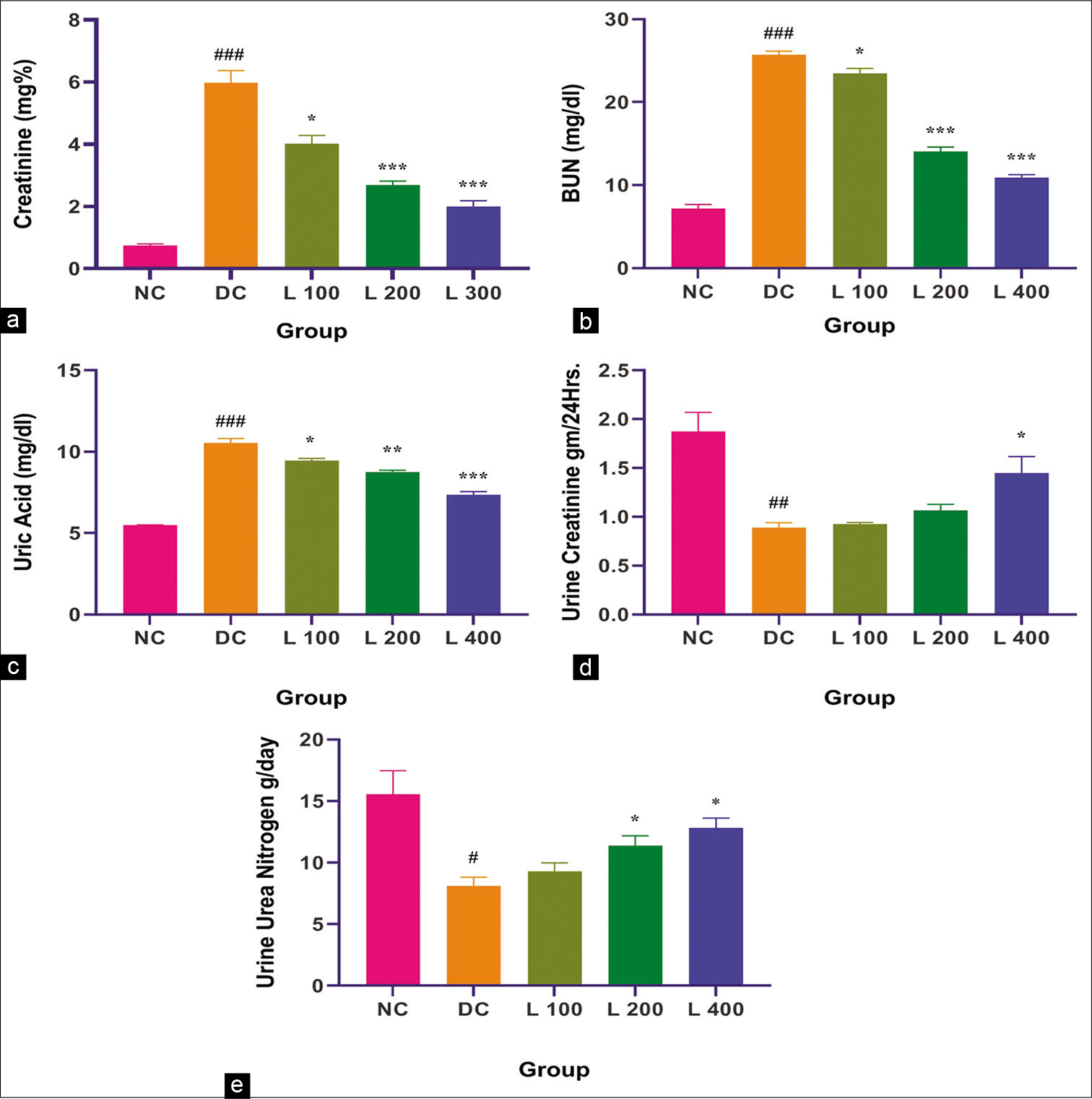
- Effect of lycopene on serum and urinary renal function indices in adenine-induced chronic renal failure in the rat. (a) Creatinine, (b) BUN, (c) Uric acid, (d) Urine creatinine, (e) UUN. Columns and vertical bars depict mean±SEM (n=6). NC: Normal control, DC: Diseases control, L 100: 100 mg/kg Lycopene+Adenine, L 200: 200 mg/kg Lycopene+Adenine, L 400: 400 mg/kg Lycopene+Adenine. *P<0.05 compared with disease versus different treatment groups, **P<0.001 compared with disease versus different treatment groups, ***P<0.0001 compared with disease versus different treatment groups, #P<0.05 compared with Normal versus disease groups, ##P<0.001 compared with normal versus disease groups, ###P<0.0001 compared with normal versus disease groups. BUN: Blood urea nitrogen, UUN: Urine urea nitrogen
Adenine administration causes significantly increased serum creatinine (5.98 mg%), uric acid (10.35 mg/dL), BUN (25.70 mg/dL) and a significant reduction in urine creatinine (0.89 mg%) and UUN (8.11 mg/dl) when compared with the control group and other lycopene-treated group (P < 0.05). In adenine-fed rats, lycopene significantly decreased the effect of adenine on serum creatinine (L100: 4.00 mg%, L200: 2.68 mg% and L400: 1.99 mg%), uric acid (L100: 9.45 mg/dl, L200: 8.75 mg/dl and L400: 7.37 mg/dl), BUN (L100: 23.45 mg/dl, L200: 14.02 mg/dl and L400: 10.90 mg/dl), urine creatinine (L100: 0.92 mg%, L200: 1.06 mg% and L400: 1.44 mg%) and UUN (L100: 9.29 mg/dl, L200: 11.38 mg/dl and L400: 12.80 mg/dl) (P < 0.05) in a dose-dependent system.
Effect of lycopene on serum vascular calcification parameters in adenine-induced CRF in the rat
The consequence of treatment with adenine, lycopene at three different doses and adenine combination on serum vascular calcification parameters are shown in [Figure 3]. Adenine administration causes a significant decrease in serum calcium and significantly increased phosphorus, magnesium and ALP when compared with the control group and other lycopene-treated group (P < 0.05). In adenine-fed rats, lycopene significantly decreased the outcome of adenine on calcium, phosphorus, magnesium and ALP (P < 0.05) in a dose-dependent system.
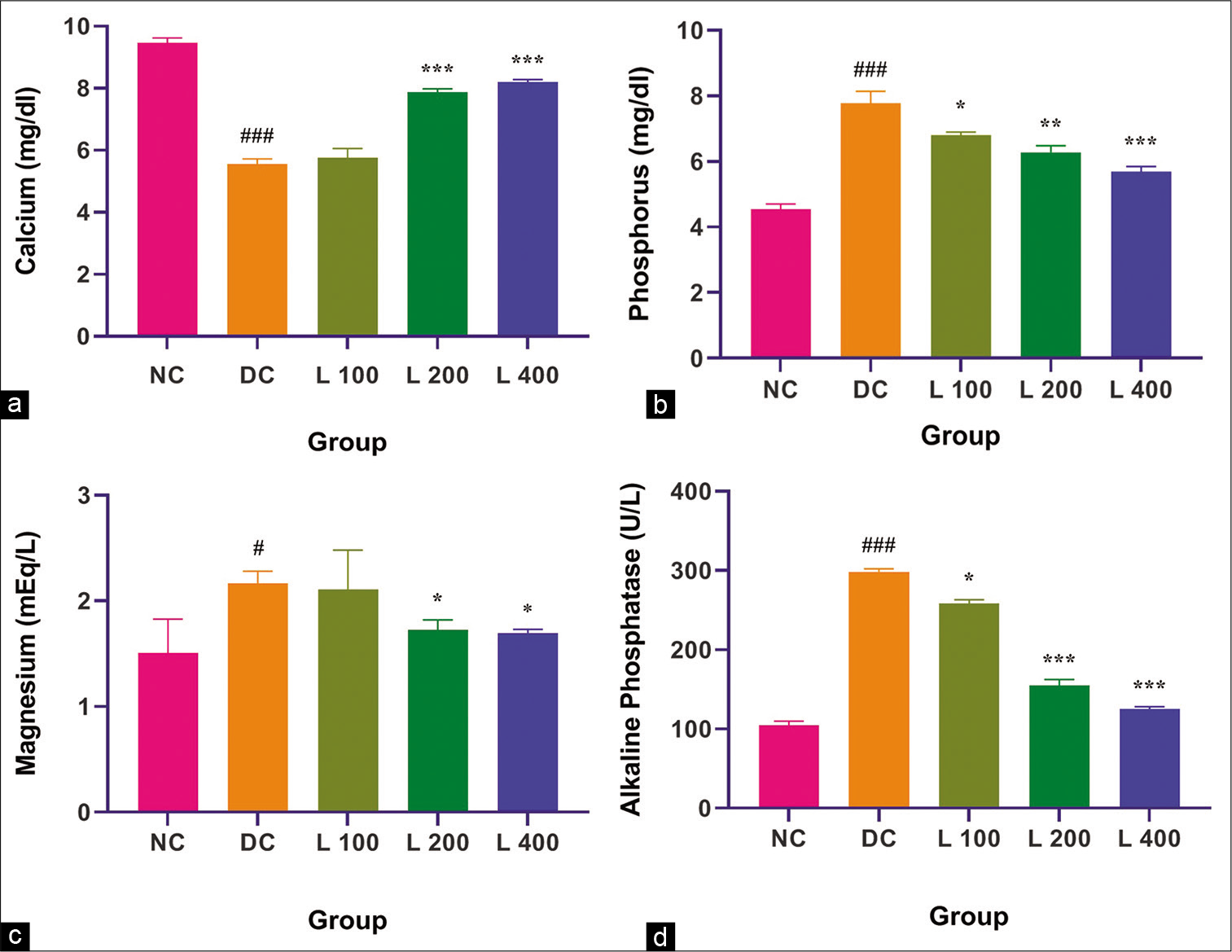
- Effect of lycopene on serum vascular calcification parameters in adenine-induced chronic renal failure in the rat. (a) Calcium, (b) Phosphorus, (c) Magnesium, (d) Alkaline phosphatase. Columns and vertical bars depict mean±SEM (n=6). NC: Normal control, DC: Diseases control, L 100: 100 mg/kg Lycopene+Adenine, L 200: 200 mg/kg Lycopene+Adenine, L 400: 400 mg/kg Lycopene+Adenine. *P<0.05 compared with disease versus different treatment groups, **P<0.001 compared with disease versus different treatment groups, ***P<0.0001 compared with disease versus different treatment groups, #P<0.05 compared with normal versus disease groups, ###P<0.0001 compared with normal versus disease groups.
Effect of lycopene on CRP in adenine-induced CRF in the rat
The effects of treatment with adenine and lycopene + adenine on CRP score are shown in [Figure 4]. Adenine administration causes significantly increased CRP score when compared with the control group and other lycopene-treated group (P < 0.05). In adenine-fed rats, lycopene significantly decreased the outcome of adenine on the CRP score (P < 0.05) in a dose-dependent system.
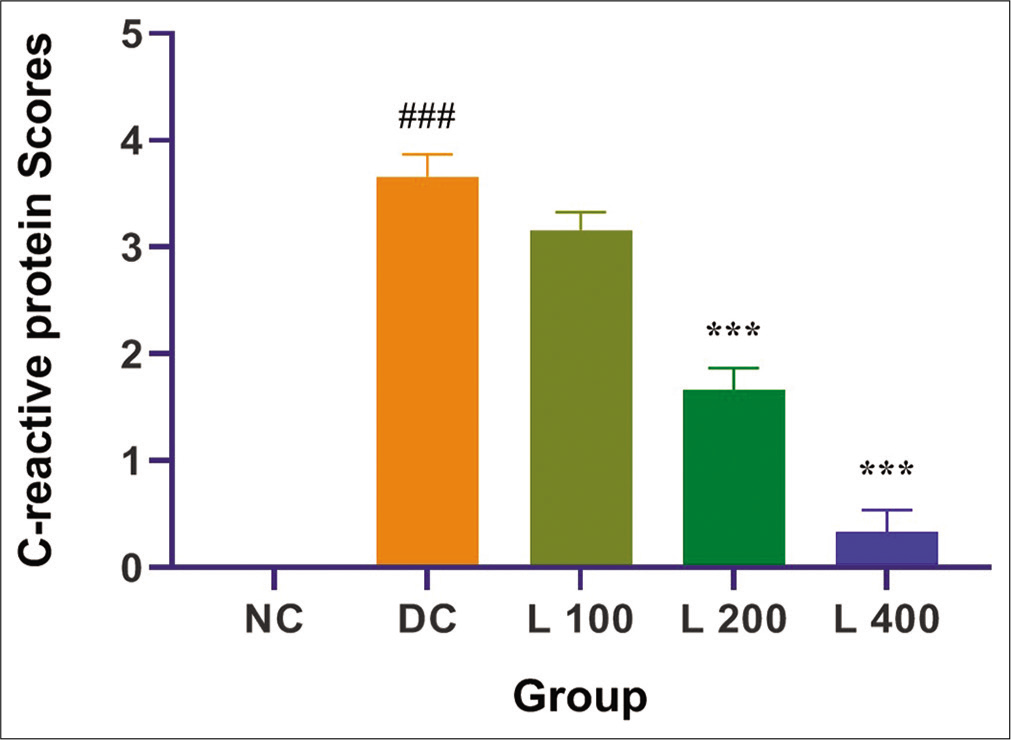
- Effect of lycopene on C-reactive protein scores in adenine-induced chronic renal failure in rat. Columns and vertical bars depict mean±SEM (n=6). NC: Normal control, DC: Diseases control, L 100: 100 mg/kg Lycopene+Adenine, L 200: 200 mg/kg Lycopene+Adenine, L 400: 400 mg/kg Lycopene+Adenine. ***P<0.0001 compared with disease versus different treatment groups, ###P<0.0001 compared with normal versus disease groups.
Effect of lycopene on anaemia indicator in adenine-induced CRF in the rat
Adenine, administered alone, caused significantly reduced anaemia indicators such as RBC, HGB and HCT when compared with the control group and other lycopene-treated group (P < 0.05). Lycopene various doses significantly decreased the adenine impact on anaemia indicator and it shows in [Figure 5].
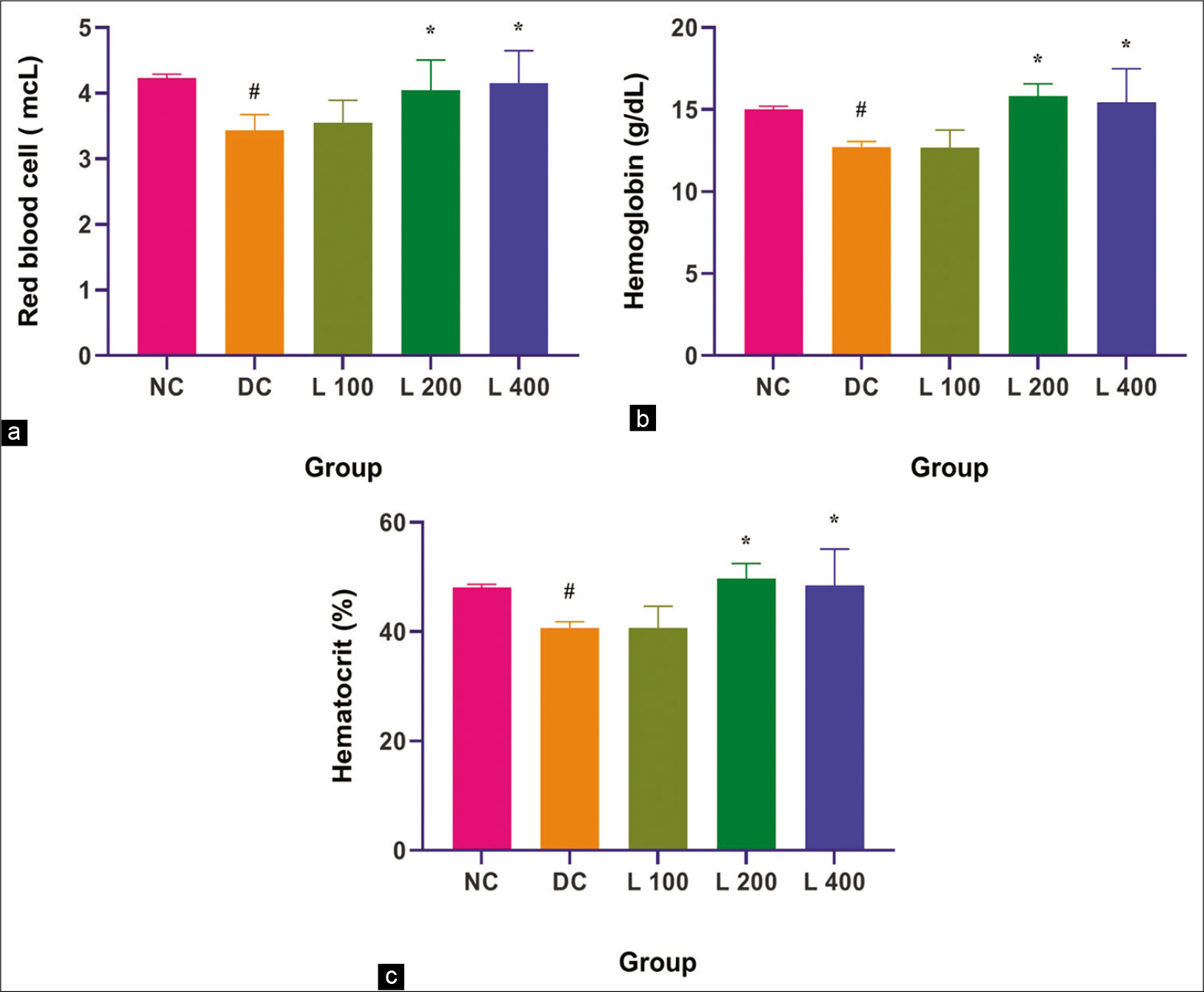
- Effect of lycopene on anaemia indicator in adenine-induced chronic renal failure in rat. (a) Red blood cells, (b) Haemoglobin, (c) Haematocrit. Columns and vertical bars depict mean±SEM (n=6). NC: Normal control, DC: Diseases control, L 100: 100 mg/kg Lycopene+Adenine, L 200: 200 mg/kg Lycopene+Adenine, L 400: 400 mg/kg Lycopene+Adenine. *P<0.05 compared with disease versus different treatment groups, #P<0.05 compared with normal versus disease groups.
Effect of lycopene on proteinuria in adenine-induced CRF in the rat
The effects of treatment with adenine and lycopene + adenine on proteinuria score are shown in [Figure 6]. Adenine administration causes significantly increased proteinuria score when compared with the control group and other lycopene-treated group (P < 0.05). In adenine-fed rats, lycopene significantly reduced the effect of adenine on proteinuria (P < 0.05) in a dose-dependent manner.
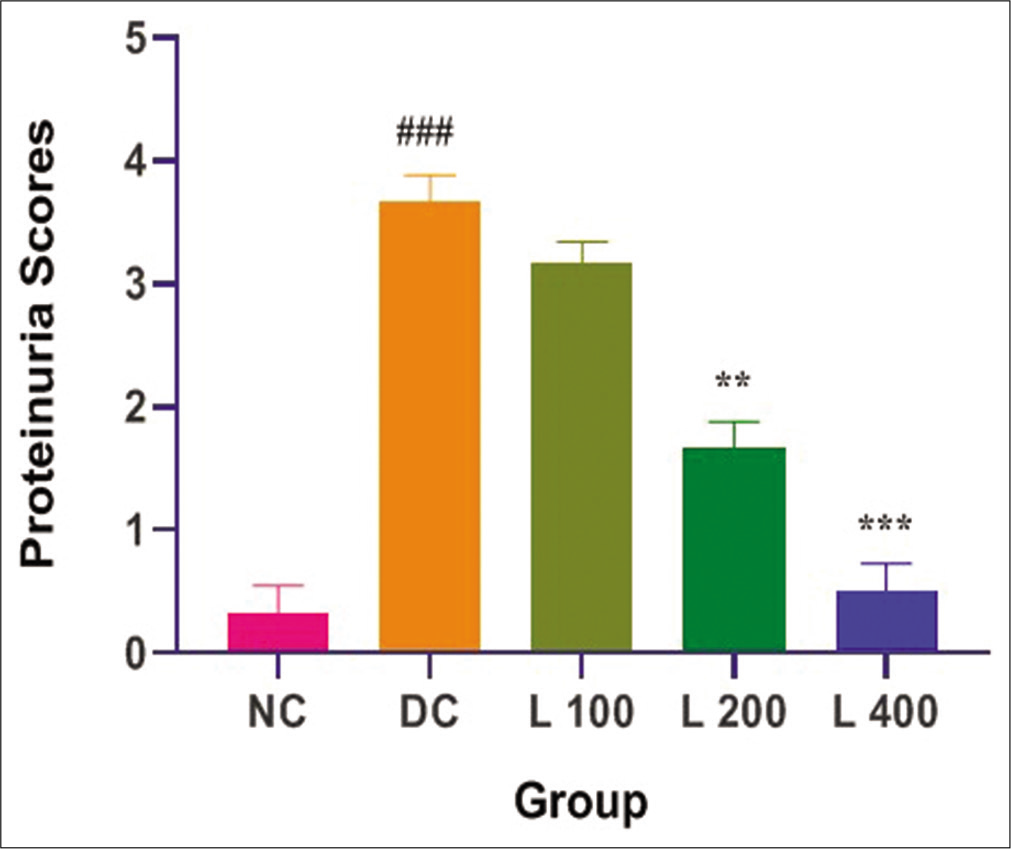
- Effect of lycopene on proteinuria scores in adenine-induced chronic renal failure in rat. Columns and vertical bars depict mean±SEM (n=6). NC: Normal control, DC: Diseases control, L 100: 100 mg/kg Lycopene+Adenine, L 200: 200 mg/kg Lycopene+Adenine, L 400: 400 mg/kg Lycopene+Adenine.**P<0.001 compared with disease versus different treatment groups, ***P<0.0001 compared with disease versus different treatment groups, ###P<0.0001 compared with normal versus disease groups.
Effect of lycopene on histopathology of kidney in adenine-induced CRF in the rat
The gross morphology of the kidney of the rat normal control group showed normal appearance in shape, size and colour. However, the kidney of the adenine-fed diseases control group demonstrated enlarged discoloration kidneys, swelling and deformity. Administration lycopene at different doses along with adenine protected structural integrity of the kidney compared with that kidney of the disease control group [Figure 7].
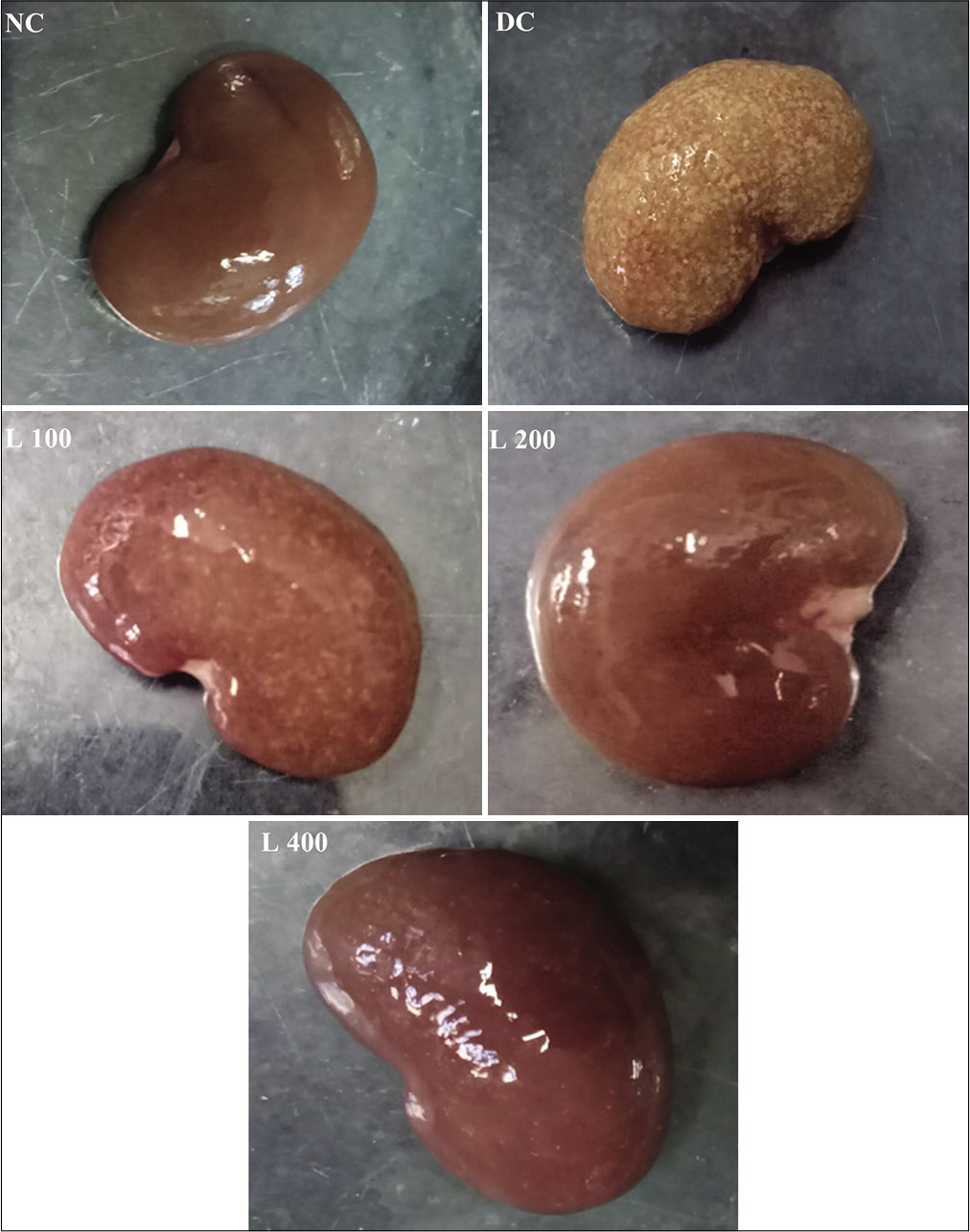
- Effect of lycopene on gross morphology of kidney in adenine-induced chronic renal failure in rat. NC: Normal control, DC: Diseases control, L 100: 100 mg/kg Lycopene+Adenine, L 200: 200 mg/kg Lycopene+Adenine, L 400: 400 mg/kg Lycopene+Adenine.
Kidney section stained with H and E from normal control group [Figure 8a] showed normal histology of renal tissue such as the normal architecture of renal distal convoluted tubule, renal proximal convoluted tubule and renal glomeruli. Kidney section from the disease control group [Figure 8b] showed a larger area of renal injury and irregular architecture of renal tissue such as loss of brush border of proximal tubules, inflammatory cells infiltrations, adenine crystal deposition, a large number of renal tubule dilation, tubular atrophy, thickening of bowman capsules and degeneration of glomeruli. Kidney section from a low dose of lycopene (100 mg/kg) along with adenine group [Figure 8c] showed a moderate area of renal injury and some irregular architecture of renal tissue such as moderate renal tubule dilation, degeneration of glomeruli, thickening of bowman capsules, inflammatory cells infiltrations, adenine crystal deposition and also show some part of the renal in tubular atrophy and also showed a very small change in the histological presentation of the kidney segment compared with the diseases control group. Kidney section from the middle dose of lycopene (200 mg/kg) along with adenine group [Figure 8d] showed mild area of renal injury and some irregular architecture of renal tissue such as mild degeneration of glomeruli, thickening of bowman capsules, inflammatory cells infiltrations, adenine crystal deposition and also showed a significant enhancement of the histological analysis of the kidney segment compared with the diseases control group. Kidney section from the high dose of lycopene (400 mg/kg) along with adenine group [Figure 8e] showed very mild area of renal injury and normal architecture of renal tissue such as mild inflammation cells infiltrations, adenine crystal deposition and also showed a significant enhancement of the histological assessment of the kidney segment compared with the diseases control group.
![Effect of lycopene on kidney histopathological changes (H and E staining [×10]) in adenine-induced chronic renal failure in rat. (a) Normal control, (b) diseases control, (c) 100 mg/kg Lycopene+Adenine, (d) 200 mg/kg Lycopene+Adenine, (e) 400 mg/ kg Lycopene+Adenine. *: Degeneration of glomeruli; #: Dilated tubule; †: Thickening of bowman capsules; ←: Tubular atrophy; ←: Adenine crystal deposition; ←: Brush border loss of proximal tubule; ←: Inflammatory cells infiltration.](/content/114/2021/65/2/img/IJPP-65-074-g008.png)
- Effect of lycopene on kidney histopathological changes (H and E staining [×10]) in adenine-induced chronic renal failure in rat. (a) Normal control, (b) diseases control, (c) 100 mg/kg Lycopene+Adenine, (d) 200 mg/kg Lycopene+Adenine, (e) 400 mg/ kg Lycopene+Adenine. *: Degeneration of glomeruli; #: Dilated tubule; †: Thickening of bowman capsules; ←: Tubular atrophy; ←: Adenine crystal deposition; ←: Brush border loss of proximal tubule; ←: Inflammatory cells infiltration.
DISCUSSION
In the present study, we developed a CRF animal model by oral administration of adenine (50 mg/kg) for 30 days in male SD rats. Biochemical and histological examination confirmed the induction of CRF. We observed that CRF was associated with reduced weight gain, kidney weight gain, along with impaired renal function and histological dimension features. Treatment with the lycopene improved renal function and improvised biochemical and histological renal results in rats with CRF induced with adenine. Lycopene tends to be an effective regulator of experimentally induced CRF thus being a significant therapeutic substance for CRF.
Long-term adenine feeding caused metabolic abnormalities in rats that were similar to CRF in humans. Azotaemia, the accumulation of uremic toxins, metabolic abnormalities of amino acids and electrolytes and hormonal imbalances were among the effects. The pathological findings in the kidneys of these experimental rats indicated proximal tubule lesions, some distal tubule lesions and glomeruli lesions. The rats with significant disturbances had contracted kidneys.[19]
Administration of adenine at the dose of 50 mg/kg substantially reduced body weight, food intake, increase relative percentage kidney weight and urine output, which was reported in the previous study.[8,20-22] In this study, adenine raises the weight of the kidney relative to the bodyweight that also suggests kidney enlargement. Simultaneous treatment of lycopene with adenine ameliorated changes in relative kidney weight associated with adenine-induced renal damage. The increase in relative kidney weight in CRF is primarily due to the transforming growth factor-beta (TGF-β)/fibrotic reaction, which increases in kidney weight.[22] The final body weight and food intake of the disease control group were slightly lower than in the normal control group, L100, L 200 and L 400 group. In addition, CRF resulted in impaired triglyceride-lipoprotein metabolism that decreased fatty acid delivery to adipose tissue and restricted energy storage ability, this leads to weight loss, food intake decrease and cachexia in CRF.[1,23] Further, the kidney is lost its ability to concentrate urine and impaired thrust mechanism in CRF which supports the finding of increased water intake in animals of the disease control group.[23] However, lycopene at the dose of 200 and 400 mg/kg treatment improved water consumption which attributes to its defensive impact on renal injury and thus maintains the capacity of kidneys to concentrate the urine that is apparent from the reduced urinary output of rats treated with lycopene.
BUN, UUN and serum creatinine, urine creatinine level are standard clinical indexes for assessing the role of glomerular filtration, the capacity of renal tubules to reabsorb and staging of CRF.[24-27] Treatment with adenine significantly impaired kidney functions by affecting biochemical parameters of serum and urine as evident from increased levels of serum creatinine, BUN and reduced levels of serum calcium, urinary creatinine and UUN which are consistent with the findings reported in the previous study.[8,20-22,28-30]
In the present investigation, lycopene treatment improved kidney functions as evident from the reduced levels of serum creatinine, BUN, uric acid, ALP, magnesium and increased levels of serum calcium, urinary creatinine and UUN levels. These results suggested improved renal function and glomerular filtration rate by the treatment of lycopene. The effect is attributed to enhanced toxin metabolism by lycopene which alleviates azotaemia and thereby suppresses damage to kidney function.
Uric acid takes an important role in heart and kidney injury in adenine-induced chronic kidney failure which is consistent with our findings. In our study, adenine caused elevation in uric acid levels which is manifested in CRF. However, treatment with lycopene ameliorated the levels of uric acid which is attributed to its capacity to inhibit xanthine oxidase enzyme. Earlier studies reported having xanthine oxidase inhibitory effect of lycopene in skeletal muscle tissues Moreover, allopurinol, the xanthine oxidase inhibitor reversed the adenine-induced CKD.[31]
CKD also involves vascular calcification, a metabolic disorder manifested as hyperphosphataemia, hypocalcaemia, hypermagnesaemia and elevated levels of ALP.[32,33] Vascular calcification represents a greater risk factor for CKD in haemodialysis patients and is linked with an increased risk of abnormal cardiovascular events such as atherosclerosis, ischaemic heart disease and vascular stiffening.[34] Administration of adenine for 30 days increased serum phosphate, magnesium and ALP levels and reduced serum calcium concentrations. Treatment with lycopene improved indicators of vascular calcification suggesting a protective role in the associated complication of CRF.
Anaemia is a general manifestation of chronic renal diseases correlated with severe cardiovascular morbidity and mortality. Adenine is reported to establish renal anaemia in a rodent model of chronic kidney failure which is evident from reduced RBC, HBG and HCT levels.[35] In our study, adenine administration substantially reduced RBC, HGB and HCT levels whereas lycopene treatment improved anaemic indicators, thereby alleviating manifestations of adenine-induced renal anaemia. Lycopene is known to have anti-anaemic effects in diabetic conditions. The effects may be attributed to the renoprotective effect of lycopene thereby improving erythropoietin levels which accelerates erythropoiesis.[36]
The process of CRP seems to be an acute reactant that enhanced inflammation and infection and it has often been used as a diagnostic tool for such situations in CRF. Treatment with adenine substantially increased serum CRP, which was previously reported.[18,32] The present study proposed that a positive serum CRP test was observed with adenine-treated animals, whereas lycopene at higher dose demonstrated a negative serum CRP test.
The rise proteinuria level in the urine is mainly due to damage to glomerular membranes in the basement suggesting proteinuria.[37] Collected urine samples over 24 h of adenine-treated animals demonstrated a positive heat coagulation test indicating the condition of proteinuria, whereas concurrent treatment with lycopene at higher doses in ameliorated the conditions of proteinuria in adenine-treated animals.
The morphological observations indicated structural damage of the renal tubules with adenine which was consistent with the previous findings.[17,38] Histological analysis of kidney of adenine-treated animals displayed the larger area with renal injury and irregular architecture of renal tissue such as a loss of brush border of proximal tubules, inflammatory cells infiltrations, adenine crystal deposition, a large number of renal tubule dilation, tubular atrophy and thickening of bowman capsules and degeneration of glomeruli. Excessive aggregation of adenine crystals in the renal tubules was known to damage the renal tubules and induction of inflammatory responses. However, microarchitectural damage to renal tissue caused by orally gavage adenine is significantly attenuated by oral gavage the lycopene which is further supported by improvement in the biochemical parameters of serum and urine. As per the result, lycopene 200 mg/kg and 400 mg/kg dose more effective dose in adenine-induced renal damage as evidenced by the improved in physiological, biochemical and histological examination.
Adenine-induced CRF is associated with increased expression of TGF-β, changes in apoptotic, oxidative stress and inflammatory factors such as nuclear factor kappa B (NF-kB) and TNF-alpha (TNF-α).[39] Lycopene is known to exert nephroprotective actions in various animal models of nephrotoxicity mainly through suppression of TGF-β, apoptotic markers such as Bax, Bcl2, Caspase-3, Caspase-9,[40] NF-kB[41-43] and TNF-α,[44-47] cytokines such as IL-6, IL-1β and oxidative stress. Lycopene decreased the level of glutathione peroxidase, decline content of GSH throughout kidney tissue as well as blocked the increase in lipid peroxides in the kidney tissue and restoring mitochondrial dysfunction.[48] Therefore, it is apparent from the previous studies that lycopene may be alleviated adenine-induced CRF through the aforementioned mechanisms.
In addition, lycopene is effective for renal antioxidant enzymes and it shown protective effect against STZ-induced diabetic nephropathy. Lycopene may be used as an adjuvant medication to reduce the difficulties associated with cisplatin-induced nephrotoxicity among cancer patients.[49,50] Based on this preclinical study, lycopene will be used as alternative or dietary treatment option for CKD patient and their comorbid condition.
CONCLUSION
The results of the present investigation suggested that oral administration of lycopene for 30 days significantly ameliorated adenine-induced renal damage as evidenced by the improved in physiological, serum and urine renal function indices, anaemia indicator, anti-inflammatory activity, modulation of vascular calcification and histological examination. The possible mechanisms of this action may be attributed to their antioxidant and cytoprotective properties. Further investigation will require to identify the molecular level mechanism action of lycopene in CRF.
Acknowledgments
This work was supported by the Department of Pharmacology, Ramanbhai Patel College of Pharmacy, Charotar University of Science and Technology, Changa-Anand. The authors thank to Ramanbhai Patel College of Pharmacy for providing the necessary facilities and equipments.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- The renoprotective effect of the dipeptidyl peptidase-4 inhibitor sitagliptin on adenine-induced kidney disease in rats. Biomed Pharmacother. 2019;110:667-76.
- [CrossRef] [Google Scholar]
- Epidemiology and risk factors of chronic kidney disease in India-results from the SEEK (Screening and Early Evaluation of Kidney Disease) study. BMC Nephrol. 2013;14:114.
- [CrossRef] [Google Scholar]
- Effect of aqueous extract and anthocyanins of calyces of Hibiscus sabdariffa (Malvaceae) in rats with adenine-induced chronic kidney disease. J Pharm Pharmacol. 2017;69:1219-29.
- [CrossRef] [Google Scholar]
- Protective effects of Salvia miltiorrhiza on adenine-induced chronic renal failure by regulating the metabolic profiling and modulating the NADPH oxidase/ROS/ERK and TGF-β/Smad signaling pathways. J Ethnopharmacol. 2018;212:153-65.
- [CrossRef] [Google Scholar]
- The balance of powers: Redox regulation of fibrogenic pathways in kidney injury. Redox Biol. 2015;6:495-504.
- [CrossRef] [Google Scholar]
- Natural products for the prevention and treatment of kidney disease. Phytomedicine. 2018;50:50-60.
- [CrossRef] [Google Scholar]
- Renoprotective effects of novel interleukin-1 receptor-associated kinase 4 inhibitor AS2444697 through anti-inflammatory action in 5/6 nephrectomized rats. Naunyn Schmiedebergs Arch Pharmacol. 2014;387:909-19.
- [CrossRef] [Google Scholar]
- The effect of sildenafil on rats with adenine-induced chronic kidney disease. Biomed Pharmacother. 2018;108:391-402.
- [CrossRef] [Google Scholar]
- Renal and myocardial histopathology and morphometry in rats with adenine-induced chronic renal failure: Influence of gum acacia. Cell Physiol Biochem. 2014;34:818-28.
- [CrossRef] [Google Scholar]
- Adenine acts in the kidney as a signaling factor and causes salt-and water-losing nephropathy: Early mechanism of adenine-induced renal injury. Am J Physiol Renal Physiol. 2019;316:F743-57.
- [CrossRef] [Google Scholar]
- Zhen-wu-tang ameliorates adenine-induced chronic renal failure in rats: Regulation of the canonical Wnt4/beta-catenin signaling in the kidneys. J Ethnopharmacol. 2018;219:81-90.
- [CrossRef] [Google Scholar]
- Protective effect of lycopene against ochratoxin A induced renal oxidative stress and apoptosis in rats. Exp Toxicol Pathol. 2013;65:853-61.
- [CrossRef] [Google Scholar]
- A systematic review on lycopene and its beneficial effects. Biomed Pharmacol J. 2017;10:2113-20.
- [CrossRef] [Google Scholar]
- Curcumin ameliorates kidney function and oxidative stress in experimental chronic kidney disease. Basic Clin Pharmacol Toxicol. 2018;122:65-73.
- [CrossRef] [Google Scholar]
- Ameliorate effects of soybean soluble polysaccharide on adenine-induced chronic renal failure in mice. Int J Biol Macromol. 2020;149:158-64.
- [CrossRef] [Google Scholar]
- Effect of herbal extracts of bryophyllum calycinum and achyranthes aspera on serum biochemical profile of rats having adenine induced chronic kidney disease. Indian J Vet Sci Biotechnol. 2019;15:1-5.
- [CrossRef] [Google Scholar]
- Ameliorating effect of gum arabic and lemongrass on chronic kidney disease induced experimentally in rats. Bull Natl Res Cent. 2019;43:47.
- [CrossRef] [Google Scholar]
- Animal model of adenine-induced chronic renal failure in rats. Nephron. 1986;44:230-4.
- [CrossRef] [Google Scholar]
- Endothelin A receptor blocker and calcimimetic in the adenine rat model of chronic renal insufficiency. BMC Nephrol. 2017;18:323.
- [CrossRef] [Google Scholar]
- Betulinic acid attenuates renal fibrosis in rat chronic kidney disease model. Biomed Pharmacother. 2017;89:796-804.
- [CrossRef] [Google Scholar]
- Effects of the SGLT-2 inhibitor canagliflozin on adenine-induced chronic kidney disease in rats. Cell Physiol Biochem. 2019;52:27-39.
- [CrossRef] [Google Scholar]
- Chronic treatment of curcumin improves hepatic lipid metabolism and alleviates the renal damage in adenine-induced chronic kidney disease in Sprague-Dawley rats. BMC Nephrol. 2019;20:431.
- [CrossRef] [Google Scholar]
- Protection effect of intracellular melanin from Lachnum YM156 and Haikunshenxi capsule combination on adenine-induced chronic renal failure in mice. Medchemcomm. 2017;8:917-23.
- [CrossRef] [Google Scholar]
- The effect of activated charcoal on adenine-induced chronic renal failure in rats. Food Chem Toxicol. 2014;65:321-8.
- [CrossRef] [Google Scholar]
- Hydrogen sulfide ameliorates chronic renal failure in rats by inhibiting apoptosis and inflammation through ROS/MAPK and NF-kB signaling pathways. Sci Rep. 2017;7:455.
- [CrossRef] [Google Scholar]
- Adenine-induced chronic kidney disease in rats. Nephrology (Carlton). 2018;23:5-11.
- [CrossRef] [Google Scholar]
- Ameliorative effect of ursolic acid on renal fibrosis in adenine-induced chronic kidney disease in rats. Biomed Pharmacother. 2018;101:972-80.
- [CrossRef] [Google Scholar]
- The flavonoid rutin improves kidney and heart structure and function in an adenine-induced rat model of chronic kidney disease. J Funct Foods. 2017;33:85-93.
- [CrossRef] [Google Scholar]
- An integrated lipidomics and phenotype study reveals protective effect and biochemical mechanism of traditionally used Alisma orientale Juzepzuk in chronic kidney disease. Front Pharmacol. 2018;9:53.
- [CrossRef] [Google Scholar]
- Lycopene supplementation attenuated xanthine oxidase and myeloperoxidase activities in skeletal muscle tissues of rats after exhaustive exercise. Br J Nutr. 2005;94:595-601.
- [CrossRef] [Google Scholar]
- Magnesium citrate protects against vascular calcification in an adenine-induced chronic renal failure rat model. J Cardiovasc Pharmacol. 2018;72:270-6.
- [CrossRef] [Google Scholar]
- Effect of cross-linked chitosan iron (III) on vascular calcification in uremic rats. Exp Biol Med. 2018;243:796-802.
- [CrossRef] [Google Scholar]
- Endogenously elevated bilirubin modulates kidney function and protects from circulating oxidative stress in a rat model of adenine-induced kidney failure. Sci Rep. 2015;5:15482.
- [CrossRef] [Google Scholar]
- A novel approach to adenine-induced chronic kidney disease associated anemia in rodents. PLoS One. 2018;13:e0192531.
- [CrossRef] [Google Scholar]
- Lycopene improves on basic hematological and immunological parameters in diabetes mellitus. BMC Res Notes. 2019;12:805.
- [CrossRef] [Google Scholar]
- Renal disease pathophysiology and treatment: Contributions from the rat. Dis Model Mech. 2016;9:1419-33.
- [CrossRef] [Google Scholar]
- Prevention of injury by resveratrol in a rat model of adenine-induced chronic kidney disease. Trop J Pharm Res. 2017;16:2027-32.
- [CrossRef] [Google Scholar]
- Expression of connective tissue growth factor is increased in injured myocardium associated with protein kinase C β2 activation and diabetes. Diabetes. 2002;51:2709-18.
- [CrossRef] [Google Scholar]
- Role of apoptosis in the pathogenesis of acute renal failure. Curr Opin Nephrol Hypertens. 2002;11:301-8.
- [CrossRef] [Google Scholar]
- Lycopene inhibits matrix metalloproteinase-9 expression and down-regulates the binding activity of nuclear factor-kappa B and stimulatory protein-1. J Nutr Biochem. 2007;18:449-56.
- [CrossRef] [Google Scholar]
- Lycopene attenuates inflammation and apoptosis in post-myocardial infarction remodeling by inhibiting the nuclear factor-kB signaling pathway. Mol Med Rep. 2015;11:374-8.
- [CrossRef] [Google Scholar]
- Lycopene reduced gene expression of steroid targets and inflammatory markers in normal rat prostate. FASEB J. 2004;24:272-4.
- [CrossRef] [Google Scholar]
- Protective effect of lycopene on deltamethrin-induced histological and ultrastructural changes in kidney tissue of rats. Toxicol Ind Health. 2014;30:160-73.
- [CrossRef] [Google Scholar]
- Lycopene supplementation reduces TNF-α via RAGE in the kidney of obese rats. Nutr Diabetes. 2014;4:e142.
- [CrossRef] [Google Scholar]
- Lycopene inhibits caspase-3 activity and reduces oxidative organ damage in a rat model of thermal injury. Burns. 2012;38:861-71.
- [CrossRef] [Google Scholar]
- Lycopene attenuates diabetes-associated cognitive decline in rats. Life Sci. 2008;83:128-34.
- [CrossRef] [Google Scholar]
- Protective effect of lycopene against nephrotoxicity induced by cyclosporine in rats. life Sci. 2013;10:1850-6.
- [Google Scholar]
- Lycopene and its potential role in diabetic nephropathy induced in rats. Benha Vet Med J. 2018;34:26-41.
- [CrossRef] [Google Scholar]
- Ameliorative effect of lycopene effect on cisplatin-induced nephropathy in patients. J Nephropathol. 2017;6:144-9.
- [CrossRef] [Google Scholar]






