Translate this page into:
Ameliorative effects of high-protein diet on hepatotoxic alterations in Swiss albino mice exposed to mobile phone radiation
*Corresponding author: Mausumi Sikdar (née Bhakta), Microbiology, Nutrition and Dietetics Laboratory, Department of Life Sciences, Presidency University, 86/1, College Street, Kolkata-700073, India. mausumi.dbs@presiuniv.ac.in
-
Received: ,
Accepted: ,
How to cite this article: Bhattacharya D, Biswas P, Gangopadhyay S, Bhakta MS. Ameliorative effects of high-protein diet on hepatotoxic alterations in swiss albino mice exposed to mobile phone radiation. Indian J Physiol Pharmacol 2020;64(4):258-64.
Abstract
Objectives:
Nowadays, mobile phones have become a prime necessity. At present, various advance networking connections such as 3G and 4G are available in the market to get a faster service. However, these mobile phones emit harmful electromagnetic radiations which have various ill effects on different physiological systems. The present study has been done to find out the extent of damage caused by mobile phone radiation on liver cells, and, whether high-protein diet (HPD) has any ameliorative effect on such changes.
Materials and Methods:
Male Swiss Albino mice were divided into four groups. Two groups of animals were exposed to mobile phone radiation at global system for mobile communications like frequencies at 1.8 GHz for 3 h continuously per day for 90 days. One of these radiation exposed groups received a normal diet (containing 5% casein) and the other exposed group received a HPD (supplemented with 20% casein). The control group animals received only a normal diet, while the animals of the remaining group received only a HPD. The serum biochemical and histological parameters of liver tissues and ladder assay of hepatic DNA were studied.
Results:
A significantly elevated level (P < 0.05) of aspartate aminotransferase, alanine transaminase, bilirubin and serum caspase-3 levels was observed in the group of animals exposed to electromagnetic radiation emitted from mobile phone. Histological changes were also observed in the experimental groups. Moreover, in genomic DNA ladder assay, fragmented DNA was observed in exposed group, which is a probable sign of cell apoptosis. Recovery symptoms were observed in the animals supplemented with high casein diet.
Conclusion:
From the present study, it may be concluded that electromagnetic radiation emitted from mobile phones has damaging effects on hepatocytes which may be ameliorated by HPD.
Keywords
Hepatic cells
Apoptosis
DNA damage
Electromagnetic radiation
DNA fragmentation
INTRODUCTION
The liver is considered as the principal detoxification site of the body. It either eliminates all toxic components or converts them to other non-toxic products, which are then eliminated from the body. As a result, when any harmful substance enters into the body or damages are caused due to radiation exposure, liver cells are prone to be damaged.[1]
Modern civilisation is dependent on various gadgets. We cannot even think of spending a single minute without using any electronic gadget. Some of these gadgets emit harmful ionising radiation, while others emit non-ionising radiation. Non-ionising radiations are those which do not carry enough energy per quantum to ionise any atom or molecule. However, when any biological system, especially animal body is exposed to these non-ionising or electromagnetic radiations, they cause various harmful effects on the different physiological systems.[2] The use of mobile phones has now become a prime necessity. The number of mobile phone users has increased up to 5 billion out of 7.8 billion people in the world.[3] People use mobile phones not only for making calls but also for various other necessary tasks such as social networking, banking transactions, online shopping and ticket booking. Mobile phones have become an indispensible tool in this pandemic time, both for adults and also for children. However, mobile phones, especially smartphones, emit non-ionising radiation that may be dangerous for the different physiological systems in humans and are responsible for ‘electropollution.’[4]
The International Agency for Research on Cancer of the World Health Organisation labelled cell phone radiation as ‘possibly carcinogenic to humans’ and put the cell phone radiation in the list of Group 2B agents, in a press report on May 31, 2011.[5]
Casein is considered to be a first class protein and almost all the essential amino acids are present in it. The isoelectric point of casein is 4.6 and the pH of milk is 6.6. In vitro studies done to evaluate the role of casein and casein derived peptides as an antioxidant agent indicates that casein and its peptides inhibit enzymatic and non-enzymatic lipid peroxidation. The antioxidant properties are not lost with dephosphorylation or proteolysis of proteins. Hence, casein can act as an anti-stress factor. However, there is no such evidence of casein acting as an antioxidant agent in vivo conditions.[6]
The present investigation has been done to study the effects of electromagnetic field exposure on hepatic cells of Swiss albino mice and whether casein has any in vivomodulatory effect on the changes caused by electromagnetic radiation.
MATERIALS AND METHODS
Selection of animals
Twenty-four Swiss albino mice (Mus musculus) weighing 20 ± 10 g were used for the study. They were maintained in a 12 h light/dark cycle at 25–27°C. All animal experiments were performed according to the ethical guidelines suggested by the Institutional Animal Ethics Committee of Presidency University and Committee for the Purpose of Control and Supervision of Experiments on Animals, Ministry of Environment, Forest and Climate Change, Government of India.
Preparation of diets
Animals were divided into four groups as mentioned in the experimental design section and fed ad libitum on two series of formulated, isocaloric, in-house diets [Table 1] in accordance with their respective groups. One hundred grams of normal diet constituted of 5 g casein, 38.5 g wheat meal, 46.5 g chick pea and 5 g corn oil and its protein content contributed to 21.36% of the total energy, whereas 100 g of high-protein diet (HPD) contained 20 g casein, 39 g wheat meal, 31 g chick pea and 5 g corn oil and its protein content contributed 33.37% of the total energy.[7]
| % Mass | Composition | Normal diet | High-protein diet |
|---|---|---|---|
| Proteins | Wheat meal | 4.6585 | 4.719 |
| Chick peas | 10.4625 | 6.975 | |
| Casein | 5 | 20 | |
| Total | 20.12105 | 31.694 | |
| Carbohydrates | 53.7355 | 45.077 | |
| Lipids | 8.0725 | 7.275 | |
| Vitamins and minerals | 7.22 | 7.20 | |
| Energy | 367.37 kcal | 370.38 kcal |
Collection of serum
About 2 ml blood was drawn from the animals by cardiac puncture and serum was separated for biochemical assay.[8]
Histological study
The liver was carefully removed from the animals of all groups and fixed in buffered formal saline for further histological analysis, following haematoxylin-eosin (H/E) staining protocol.[9] The tissues were then dehydrated with graded alcohol, cleared in xylene before embedding in paraffin wax (melting point 56.0°C). Serial sections of approximately 4–5 μm thickness were cut in rotary microtome and then passed through xylene followed by absolute alcohol and water. The sections were stained with haematoxylin, dehydrated with graded alcohol, countered-stained with eosin, cleared in xylene and mounted in Canada balsam. The slides were left to air dry for 24 h before observation under the light microscope (Zeiss Axioscope A1, Thornwood, NY, USA).
Chemicals
Assay kits for the estimation of aspartate aminotransferase (AST), alanine aminotransferase (ALT) and bilirubin were bought from ARKRAY Healthcare Pvt. Ltd., Surat (India). Serum caspase-3 antibodies were bought from Merck, India. Commercially available fat free, low in vitamins casein was purchased from NICE Chemicals Private Ltd., Cochin, India.
Assessment of biochemical parameters
Transaminase (SGOT and SGPT) activities were estimated by the method of Reitman and Frankel and serum bilirubin was measured by Diazo method.[10,11]
Serum caspase-3 levels were measured by solid-phase, quantitative, sandwich enzyme-linked immunosorbent assay, using caspase-3 antibody from Merck, India.[12]
Study of genomic DNA (gDNA) by gel electrophoresis laddering assay
Fragmentation of gDNA, observed as the DNA ladder in gel electrophoresis, is indicative of DNA damage and cellular apoptosis according to Lu et al., 2002.[13] At first, the hepatic tissue was isolated and homogenised. It was washed overnight in PBS at a constant temperature of 37°C and then lysed in 0.5 ml lytic buffer (50 mM Tris-HCl, 10 mM EDTA, 0.5% Triton and 100 μg/ml protein kinase K, pH at 8.0). The lysate was incubated at 37°C in DNase-free RNase (100 μg/ ml) for 2 h. It was run by three sets of phenol/chloroform mixture (1:1 v/v) and was centrifuged at 15,000 rpm for 5 min at 4°C in chloroform. The partially extracted DNA was taken in double volume of ice cold 100% ethanol and 1/10 volume of 3 M sodium acetate. The mixture was then allowed to be precipitated at pH 5.2 for 1 h at −20°C. For further precipitation, it was again centrifuged at 15,000 rpm for 15 min at 4°C. The precipitated DNA pellet was washed in 70% ethanol and then air dried for further assay. The dried pellet was again dissolved in a mixture of 10 mM Tris-HCl and 1 mM EDTA at pH 8.0. This solution containing gDNA was then electrophoresed on 1.5% agarose gel and then stained with ethidium bromide. A 10,000 bp DNA ladder (Merck, India) was used as a marker of the molecular size of DNA and fragmented DNA was studied using UV illuminated gel imaging system (ChemiDoc, MP, Biorad).[14]
Study design
Mobile phone radiation exposure technique
Mice were exposed to global system for mobile communications like frequencies of 1.8 GHz emitted from mobile phone (Micromax Bharat 2; Q 402), connected with a power meter with specific absorption rate 1.5 W/kg for 3 h continuously per day for 3 months in the exposed groups. The cell phones were in calling mode and placed 14 cm perpendicularly on top of the cage made up of plexiglass. The animals were free to move in the cage during the exposure period.[15,16]
Experimental design
All animals were divided randomly into four groups (Group A-D), each group containing six animals. Animals of Group A were only fed normal diet and those of Group B were fed normal diet and were simultaneously exposed to mobile phone radiation. Animals of Group C were fed a HPD and were simultaneously exposed to the same dose of mobile phone radiation, while animals of Group D were fed only HPD and not exposed to any radiation. At the end of 90 days, animals were sacrificed and blood was drawn by cardiac puncture for biochemical assay and liver tissues were collected for histological study.
Statistical analysis
All data were expressed in terms of mean ± SEM (n= 6). One-way ANOVA was performed to determine the level of significance. P < 0.05 was considered as statistically significant.
RESULTS HPD
protects from the liver damage induced by mobile phone radiation
Hepatic damage was assessed by estimating AST and ALT activities and their values are represented in [Table 2]. Animals exposed to radiation and fed normal diet (Group B) showed significantly high levels (P< 0.05) of serum AST and ALT activities when compared to the control group (Group A). Animals treated with HPD along with electromagnetic radiation (Group C) showed significantly lower (P< 0.05) serum AST and ALT activities than the radiation exposed group fed with normal diet (Group B) and the signs of recovery are almost similar to those of the control group animals. Animals treated with only HPD (Group D) showed no significant (P> 0.05) change in the serum AST and ALT activities when compared to the control (Group A).
| Group of animals | AST activity (IU/L) | ALT activity (IU/L) | Bilirubin (mg/100 ml) | ||
|---|---|---|---|---|---|
| Total bilirubin | Conjugated bilirubin | Unconjugated bilirubin | |||
| Group A (no treatment) | 56.19±0.075 | 13.46±0.354 | 0.76±0.071 | 0.46±0.007 | 0.3±0.012 |
| Group B (normal diet + 1.8 GHz radiation) | 90.23±0.088* | 26.46±0.443* | 4.16±0.027* | 2.25±0.99* | 1.91±0.179* |
| Group C (high-protein diet + 1.8 GHz radiation) | 64.23±0.066*,# | 16.8±0.487*,# | 2.55±0.15*,# | 1.26±0.095*,# | 1.28±0.145*,# |
| Group D (high-protein diet) | 57.02±1.01 | 13.16±0.433 | 0.72±0.14 | 0.41±0.014 | 0.32±0.002 |
HPD prevents the rise in total, conjugated and unconjugated bilirubin on mobile phone radiation
Animals exposed to electromagnetic radiation emitted from mobile phone and fed normal diet (Group B) showed significantly higher amounts of total (P< 0.05), conjugated (P< 0.05) and unconjugated (P < 0.05) serum bilirubin [Table 2] compared with the control group, whereas animals treated with HPD and exposed to the same frequencies of electromagnetic radiation (Group C) showed serum bilirubin values tending toward normal [Table 2]. Hence, it may be said that signs of recovery are being observed in radiation exposed animals fed with HPD. On the other hand, animals treated with only HPD and not exposed to any radiation showed no alterations in the serum bilirubin value [Table 2].
HPD defends against histoarchitectural changes of liver due to mobile phone radiation
The histological structure of liver tissue of control animals with normal diet (Group A) showed prominent central vein and hepatic lobules [Figure 1 and 2a]. Dead cell debris obstructed the central veins and severely distorted hepatic cells were observed in the radiation exposed animal group fed normal diet (Group B), as depicted in [Figure 1b].

- The haematoxylin and eosin stained hepatic tissue sections from various groups of mice at low magnification (100×). The hepatic parenchyma, lobules and the sinusoidal spaces and central vein are maintained in normal state in control liver (a), whereas hepatic parenchyma is disorganised and morphologically damaged with indistinct boundaries of hepatic lobules, cellular occlusion of the lumen of the central vein, disoriented and increased sinusoidal spaces on mobile phone irradiation (b) as indicated by the black arrows. HPD supplementation helped in sustaining the normal morphology of the hepatic parenchyma even on exposure to electromagnetic radiation from the mobile phone (c). Liver tissues of mice, kept on HPD only, demonstrated the features relatable to that of the control group (d).
![H/E stained hepatic tissue sections from different groups of mice at high magnification (400×). Normal features are observed in case of control liver (a) The central veins are enlarged in the liver of the radiation exposed group (b) with karyorrhexis and karyolysis as the predominant features in hepatic cells when compared to that of control. HPD prevented the cellular changes with reduced the occurrence of karyorrhexis and karyolysis and minimal obstruction in the central vein, induced by electromagnetic radiation on the liver (c). Hepatic morphology of animals, maintained on HPD only, was similar to that of control (d). [Thick arrow: cellular occlusion of central vein; thin arrows: hepatic tissue damages]](/content/114/2020/64/4/img/IJPP-64-258-g002.png)
- H/E stained hepatic tissue sections from different groups of mice at high magnification (400×). Normal features are observed in case of control liver (a) The central veins are enlarged in the liver of the radiation exposed group (b) with karyorrhexis and karyolysis as the predominant features in hepatic cells when compared to that of control. HPD prevented the cellular changes with reduced the occurrence of karyorrhexis and karyolysis and minimal obstruction in the central vein, induced by electromagnetic radiation on the liver (c). Hepatic morphology of animals, maintained on HPD only, was similar to that of control (d). [Thick arrow: cellular occlusion of central vein; thin arrows: hepatic tissue damages]
Data are presented as mean±SEM. Values bearing superscripts are significantly different in the with respect to the control group and Group D (*) and Group B (#) (P<0.05). AST: Aspartate aminotransferase, ALT: Alanine aminotransferase
Furthermore, on radiation exposure, hepatic lobules lost their normal shape with undifferentiated boundaries. The hepatic cells were arranged irregularly instead of their characteristic radial arrangements, and sinusoidal spaces increased and were disorganised [Figure 1 and 2b]. The incidences of karyorrhexis and karyolysis in hepatic cells were noteworthy observations in radiation exposed liver [Figure 2b]. On the other hand, HPD supplementation prevented the cellular alterations induced by electromagnetic radiation of the same frequencies on the liver to a considerable extent [Figure 1 and2c], specifically the obstruction in the central veins was minimal and they maintained their proper shape in comparison with the animals exposed to electromagnetic radiation and fed on normal diet (Group B). The hepatic lobules retained their cytoarchitecture, however, karyorrhexis and karyolysis were minimised to some extent when compared to the control liver. The histological characteristics of the hepatic sections of animals fed on HPD (Group D) resembled those of the control animals [Figure 1 and2d].
HPD downplays the upsurge in serum caspase-3 activity on mobile phone radiation
Animals irradiated with electromagnetic radiation from mobile phone and fed on normal diet (Group B), exhibited higher serum caspase-3 activity (P< 0.05) with respect to the control group of animals (Group A) [Figure 3]. Supplementation of diet with 20% casein in animals exposed to electromagnetic irradiation (Group C), lowered the serum caspase-3 activity (P< 0.05) to a considerable extent compared to irradiated animals fed on normal diet (Group B). In the group of animals fed only on HPD (Group D), the serum caspase-3 activity was at par with the controls (P> 0.05) [Figure 3].
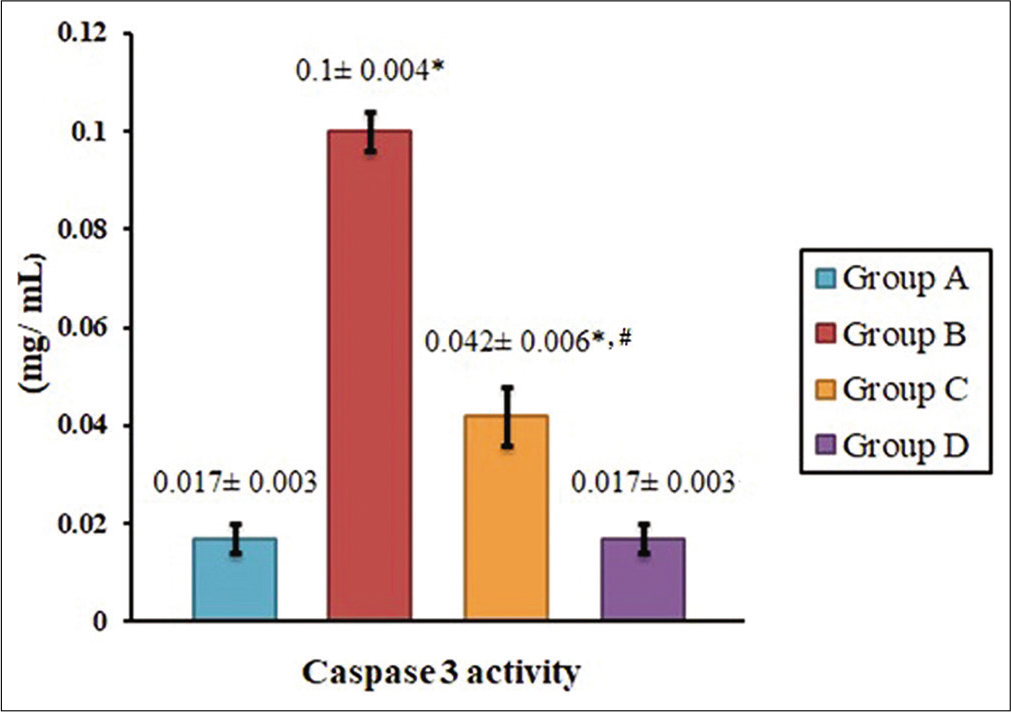
- Inhibitory role of high-protein diet on serum caspase-3 activity induced by electromagnetic radiation in mice. Data are presented as mean ± SEM. Values bearing superscripts are significantly different with respect to the control group and Group D (*) and Group B (#) (P < 0.05).
HPD inhibits the fragmentation and loss of hepatic gDNA due to mobile phone radiation
The image of gel electrophoresis of hepatic DNA depicts the extent of fragmentation of the gDNA. The gDNA band of the Group A hepatic cells exhibited a conspicuous band (>10,000 bp) and negligible fragmentation [Figure 4]. However, gDNA from the hepatic cells of mobile phone irradiated animals (Group B), displayed faint band at >10,000 bp position and the fragments were prominent between 750 bp and 500 bp position [Figure 4]. gDNA of hepatic cells from animals, exposed to mobile phone radiation and fed on HPD (Group C), retained their band at >10,000 bp position although minimum laddering characteristics was observed. The gDNA from hepatic cells of Group D (fed only on HPD) animals was comparable to that of Group A [Figure 4].
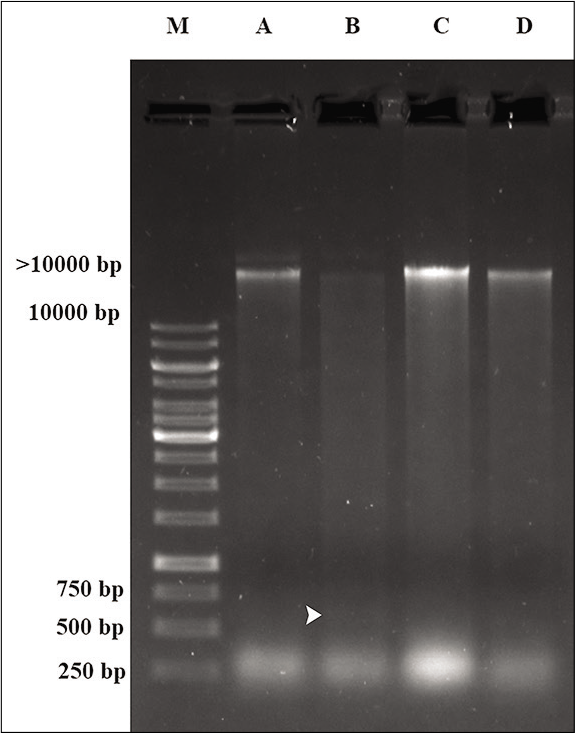
- The image of hepatic DNA gel electrophoresis showing genomic DNA damage induced by mobile phone radiation and its protection by HPD supplementation. Prominent gDNA bands are visible at >10,000 bp positions for the Groups A, C and D in contrast to faint bands observed at that position along with fragments (white arrowhead) noticed at position between 500 and 750 bp for gDNA of the hepatic cells of mobile phone irradiated animals (Group B) (M: Marker lane).
DISCUSSION
The significance of the biological effects of mobile phone radiation is enhanced due to its continuous exposure in the modern human population. In this study, short-term mobile phone radiofrequency radiations at a relatively high intensity (1.8 GHz) are used since this is the 4G frequency currently available in India from different mobile network operators. The duration of radiation exposure, chosen in this study, is modified on the basis of two different studies,[16,17] where 0.9 GHz frequency and similar experimental setups were used. Although 3 h/day of mobile phone radiation exposure for 60 days caused fatal manifestations in different haematological parameters in mice,[16] 4 h/day exposure to cellular phone radiation for 60 days displayed no toxic alterations in the mice kidneys.[17] Since, a novel frequency, higher than 0.9 GHz, is used to mimic the real-life conditions, mice were irradiated using common mobile handset for a duration of 3 h/day for a period of 90 days.
Casein-enriched diet (20% of total mass) has shown to be effective in improving the adrenogonadal function after treatment with GnRH antagonist.[18] The efficacy of HPD enriched with casein has already been established as a prospective formulation to ameliorate altered haematological functions on electromagnetic irradiation.[16] The protein content of normal and HPDs, used in this study, contributes to 21.36% and 33.37% of total energy, respectively, which are much lower than the threshold level of toxic dose.
Results of biochemical estimations in this study indicate an elevation in serum AST, ALT and bilirubin values in the animals exposed to electromagnetic radiation and receiving thee normal diet (Group B). Elevated serum AST and ALT levels are associated with pathological conditions such as myocardial infarction, liver disorders like viral hepatitis resulting in cell necrosis, obstructive jaundice or severe haemolytic disorders.[19] In alcoholic hepatitis, a moderate increase of serum AST and ALT levels may be seen.[20]
This study reveals that both conjugated and unconjugated bilirubin levels were increased in animals exposed to electromagnetic radiation and receiving normal diet (Group B). As a result, the total bilirubin level had also increased. Conjugated bilirubin levels may be increased due to severe bile duct obstruction, resulting in obstructive jaundice, hepatic cell death, etc.[21] The serum level of unconjugated bilirubin increases during severe haemolytic anaemia.[22] Previous observation by Bhattacharya et al. indicated that the animals exposed to mobile phone radiation suffered from low R.B.C. count.[16] From the present study, it may be concluded that the low R.B.C. count is due to severe haemolysis, which is reflected by the elevated serum unconjugated bilirubin levels.
From the histological studies, it is observed that, in the experimental group, in which the animals were exposed to electromagnetic radiation along with normal diet (Group B), the central hepatic vein is extremely occluded and the hepatic lobules were also distorted in shape, which are similar to the histological structure of liver in rats with severe obstructive jaundice.[23] Due to the distortion of hepatic cells, the stress marker enzymes (AST and ALT) may be increased and the obstruction of the hepatic central vein caused the significant increment of the serum bilirubin level. Various simulation studies indicate that exposure to electromagnetic radiation in 1.8 GHz or more frequencies may cause oxidative stress through its thermal effect on hepatic cell damage, causing severe apoptosis.[24] The hepatic parenchymal cells may also be subjected to oxidative stress injury in the liver. Moreover, Kupffer cells, hepatic stellate cells and endothelial cells are potentially more exposed and sensitive to oxidative stress-related molecules.[25] The biliary duct may be occluded by these apoptotic cells, which is reflected by symptoms similar to the obstructive jaundice. The presence of karyorrhexis and karyolysis nature of the nucleus indicates that there was higher frequency of occurrence of apoptosis in the hepatic cells.[26]
Caspases are the crucial mediators of apoptosis. Among them, caspase-3 is a frequently activated death protease, catalysing the specific cleavage of many key cellular proteins. Caspase-3 is indispensable for apoptotic chromatin condensation and DNA fragmentation in cells.[27] As a result, from the elevated level of serum caspase-3 activities, it may be concluded that there is a significant increase in cellular apoptosis.
The damaging effect of electromagnetic radiation emitted from mobile phone on DNA has been reported in several previous studies.[28,29] DNA fragmentation is directly related to cell apoptosis and reactive oxygen species (ROS) generation.[30] The presence of fragmented DNA in hepatic cells in our study clearly indicates the occurrence of severe hepatic cell apoptosis. According to Kesari et al., regular and long-term use of microwave devices (mobile phone) at domestic level can have negative impact on various biological systems. They also suggested that increased ROS may play an important role by enhancing the effect of electromagnetic radiation.[31] Apoptosis plays a crucial role for development and tissue homeostasis in living organisms and it is carefully regulated and balanced by pro-apoptotic and anti-apoptotic proteins.[32]
Due to the supplementation of casein in the diet, the pathological symptoms are significantly subsided (Group C animals). As casein acts as an anti-stress factor and due to the presence of amino acids such as glutamic acid (20.2%), lysine (7.4%), aspartic acid (6.4%) and arginine (3.7%), it induces cell proliferation, which is responsible for the replacement of apoptotic cells with newly formed cells.[33,34] Thus, the serum bilirubin level has been significantly decreased. The casein-derived peptides have free radical scavenging and metal chelating properties, through which the stress marker enzymes may be significantly decreased.[35]
CONCLUSION
The present study entails that the electromagnetic radiation emitted from mobile phones induce apoptotic changes in hepatic cells, which can be ameliorated by HPD fortified with casein. HPD is enriched with bioactive peptides[18] which are essential to undertake its protective functions and reduce the hepatic cell damage caused by mobile phone radiation.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Authors acknowledge the Faculty Research and Professional Development grant of Presidency University and the Department of Higher Education, Science and Technology and Biotechnology (Biotech.), Government of West Bengal (77 [Sanc.] - BT/ST/P/S&T/IG-39/2016) grant to the corresponding author for financial assistance.
Conflicts of interest
There are no conflicts of interest.
References
- Renal and hepato protective effects of green tea (Camellia sinensis) extract on Wistar rats treated with sodium oxalate. Int J Pharm Bio Sci. 2016;7:740-6.
- [CrossRef] [Google Scholar]
- Radiation effects of mobile phones and tablets on the skin: A systematic review. Adv Med. 2018;1:1-5.
- [CrossRef] [PubMed] [Google Scholar]
- Mobile phone radiation: Physiological and pathophysiologcal considerations. Indian J Physiol Pharmacol. 2015;59:125-35.
- [Google Scholar]
- Available from: http://www.auspi.in/news/EMF-Information-Paper_30-07-2014.pdf [Last accessed on 2014 Nov 02]
- [Google Scholar]
- Caseins and casein hydrolysates. Antioxidative properties and relevance to lipoxygenase inhibition. J Agric Food Chem. 2001;49:295-302.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of a high protein diet on cognition and brain metabolism in cirrhotic rats. Physiol Behav. 2015;149:220-8.
- [CrossRef] [PubMed] [Google Scholar]
- Blood sample collection in small laboratory animals. J Pharmacol Pharmacother. 2010;1:87-93.
- [CrossRef] [PubMed] [Google Scholar]
- The history of histology: A brief survey of sources. Hist Sci. 1977;15:77-101.
- [CrossRef] [Google Scholar]
- A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am J Clin Pathol. 1957;28:56-63.
- [CrossRef] [PubMed] [Google Scholar]
- Determination of serum bilirubin by the diazo method using the diazotized 3-nitroaniline reacting readily with the photoproducts of bilirubin. Jap J Clin Chem. 1994;23:158-63.
- [Google Scholar]
- Molecular mechanisms of caspase regulation during apoptosis. Nat Rev Mol Cell Biol. 2004;5:897-907.
- [CrossRef] [PubMed] [Google Scholar]
- Innate direct anticancer effector function of human immature dendritic cells II Role of TNF, lymphotoxin-α1β2, Fas ligand, and TNF-related apoptosis-inducing ligand. J Immunol. 2002;168:1831-9.
- [CrossRef] [PubMed] [Google Scholar]
- Modulation of hepatotoxicity, DNA fragmentation and gene expression of Solanum nigrum leaves extract in rats treated with silver nanoparticles. J Appl Pharm Sci. 2017;7:25-35.
- [Google Scholar]
- Microwave exposure affecting reproductive system in male rats. Appl Biochem Biotechnol. 2010;2:416-28.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of electromagnetic radiation exposure on haematological parameters of Swiss albino mice and their modulation by high protein diet. Biomedicine. 2016;36:121-7.
- [Google Scholar]
- Histological and biochemical studies of mice kidney after exposure to mobile phone radiation. J Bas Res Med Sci. 2016;3:45-51.
- [CrossRef] [Google Scholar]
- Effect of casein diet on gonadotropin releasing hormone antagonist induced changes in adrenal gonadal functions in male rats. Indian J Exp Biol. 2001;39:1249-53.
- [Google Scholar]
- Incidence and outcome of ischemic hepatitis complicating septic shock. Hepatol Res. 2009;39:700-5.
- [CrossRef] [PubMed] [Google Scholar]
- The ratio of aspartate aminotransferase to alanine aminotransferase: Potential value in differentiating non-alcoholic steatohepatitis from alcoholic liver disease. Am J Gastroenterol. 1999;94:1018-22.
- [CrossRef] [PubMed] [Google Scholar]
- Liver enzyme alteration: A guide for clinicians. CMAJ. 2005;172:367-79.
- [CrossRef] [PubMed] [Google Scholar]
- Unconjugated hyperbilirubinemia in commercial pilot: A clinical dilemma. Indian J Aerospace Med. 2009;53:53-7.
- [Google Scholar]
- Histone preconditioning protects against obstructive jaundice-induced liver injury in rats. Exp Ther Med. 2014;8:15-20.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of isothermal radiofrequency radiation on cytolytic T lymphocytes. FASEB J. 1996;10:913-9.
- [CrossRef] [PubMed] [Google Scholar]
- Biochemical mechanisms in drug-induced liver injury: Certainties and doubts. World J Gastroenterol. 2009;15:4865-76.
- [CrossRef] [PubMed] [Google Scholar]
- Apoptosis: A review of programmed cell death. Toxicol Pathol. 2007;35:495-516.
- [CrossRef] [PubMed] [Google Scholar]
- Starfish apaf-1 activates effector caspase-3/9 upon apoptosis of aged eggs. Sci Rep. 2018;8:1-14.
- [CrossRef] [PubMed] [Google Scholar]
- Pathophysiology of cell phone radiation: Oxidative stress and carcinogenesis with focus on male reproductive system. Reprod Biol Endocrinol. 2009;7:114-22.
- [CrossRef] [PubMed] [Google Scholar]
- 2.45 GHz microwave irradiation-induced oxidative stress affects implantation or pregnancy in mice Mus musculus. Appl Biochem Biotechnol. 2013;169:1727-51.
- [CrossRef] [PubMed] [Google Scholar]
- Detection of reactive oxygen species (ROS) and apoptosis in human fragmented embryos. Hum Reprod. 1998;13:998-1002.
- [CrossRef] [PubMed] [Google Scholar]
- Cell phone radiation exposure on brain and associated biological systems. Indian J Exp Biol. 2013;51:187-200.
- [Google Scholar]
- Protective effects of zinc on 2. 45 GHz electromagnetic radiation-induced oxidative stress and apoptosis in HEK293 cells. Biol Trace Elem Res. 2020;194:368-78.
- [CrossRef] [PubMed] [Google Scholar]
- The voluminosity of bovine casein micelles and some of its implications. J Dairy Res. 1979;46:317-23.
- [CrossRef] [PubMed] [Google Scholar]
- Structure and stability of bovine casein micelles. Adv Protein Chem. 1992;43:63-151.
- [CrossRef] [Google Scholar]
- The effect of amino acids and amino acid derivatives on cell proliferation. Acta Biomed Ateneo Parmense. 1993;64:139-45.
- [Google Scholar]






